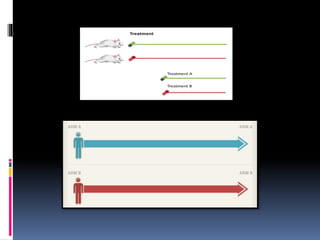
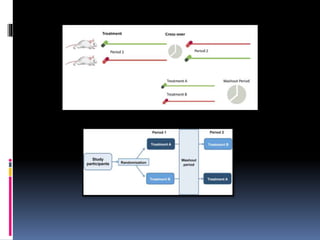
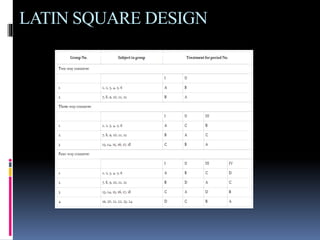
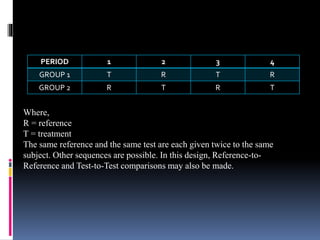
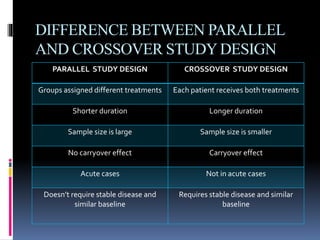
The document discusses cross-over study designs in pharmacokinetic studies, emphasizing the importance of minimizing experimental variables and avoiding biases in bioavailability studies. It compares parallel and crossover designs, noting that crossover designs reduce intersubject variability and allow participants to act as their own controls. Various statistical concepts, such as randomization and replication, are integral to study design, and specific designs like Latin square and balanced incomplete block design are highlighted for their effectiveness in clinical trials.