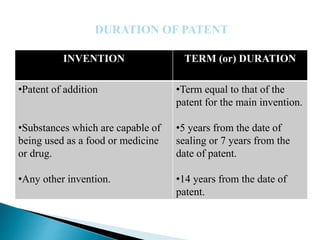
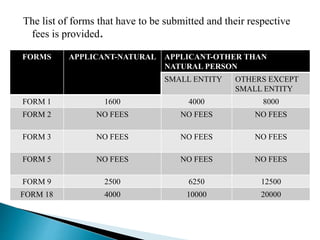
The document provides information on different types of patent applications in India. It discusses provisional applications, which are filed before an invention is finalized to claim priority. Non-provisional or ordinary applications are filed when priority is not claimed. Convention applications allow claiming priority based on applications filed in convention countries. PCT international applications can be filed to seek protection in multiple countries simultaneously. National phase applications must then be filed in each designated country. The document also discusses patent of addition applications for modifications of existing inventions, and divisional applications which divide one application into multiple applications.