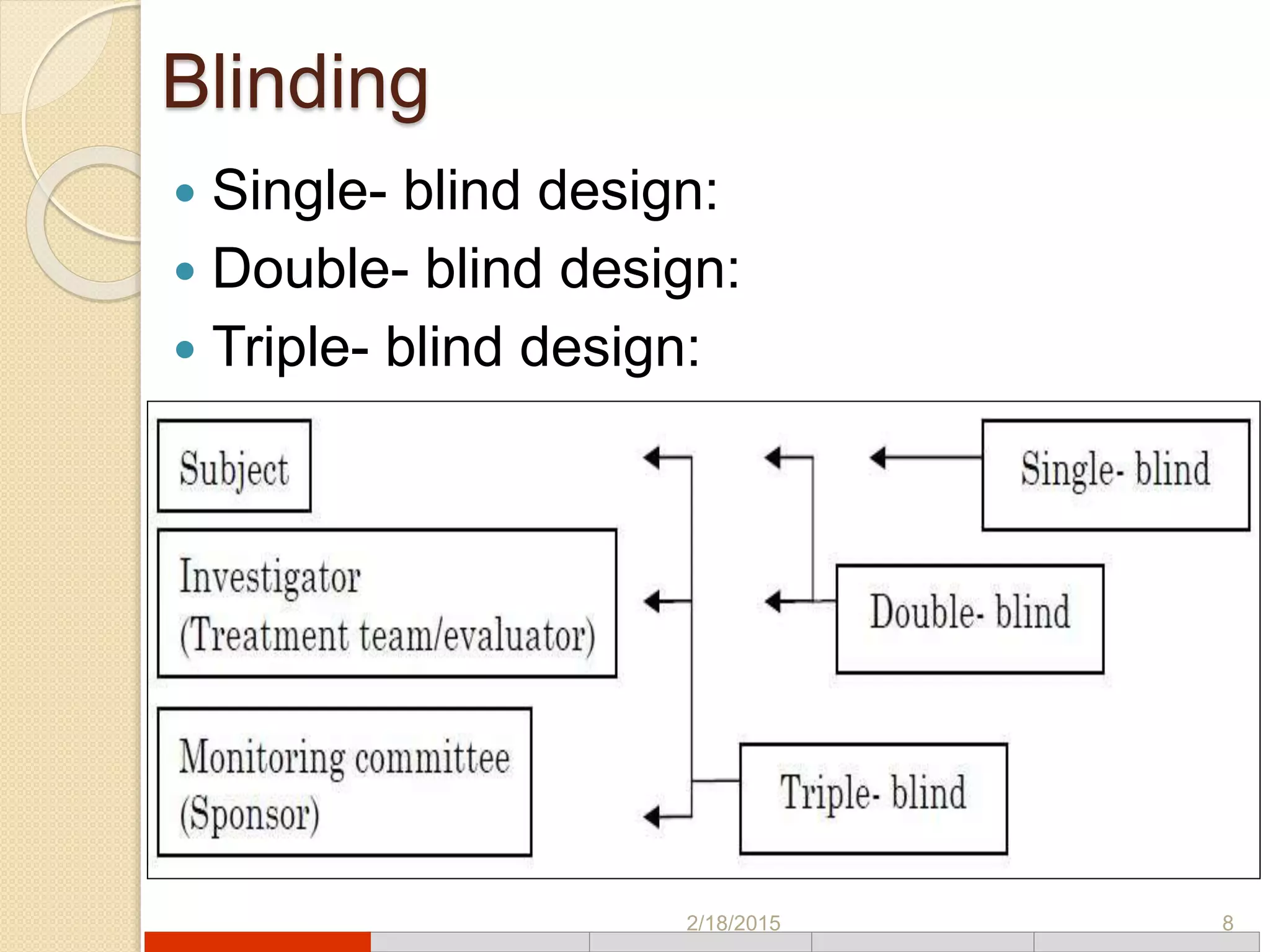
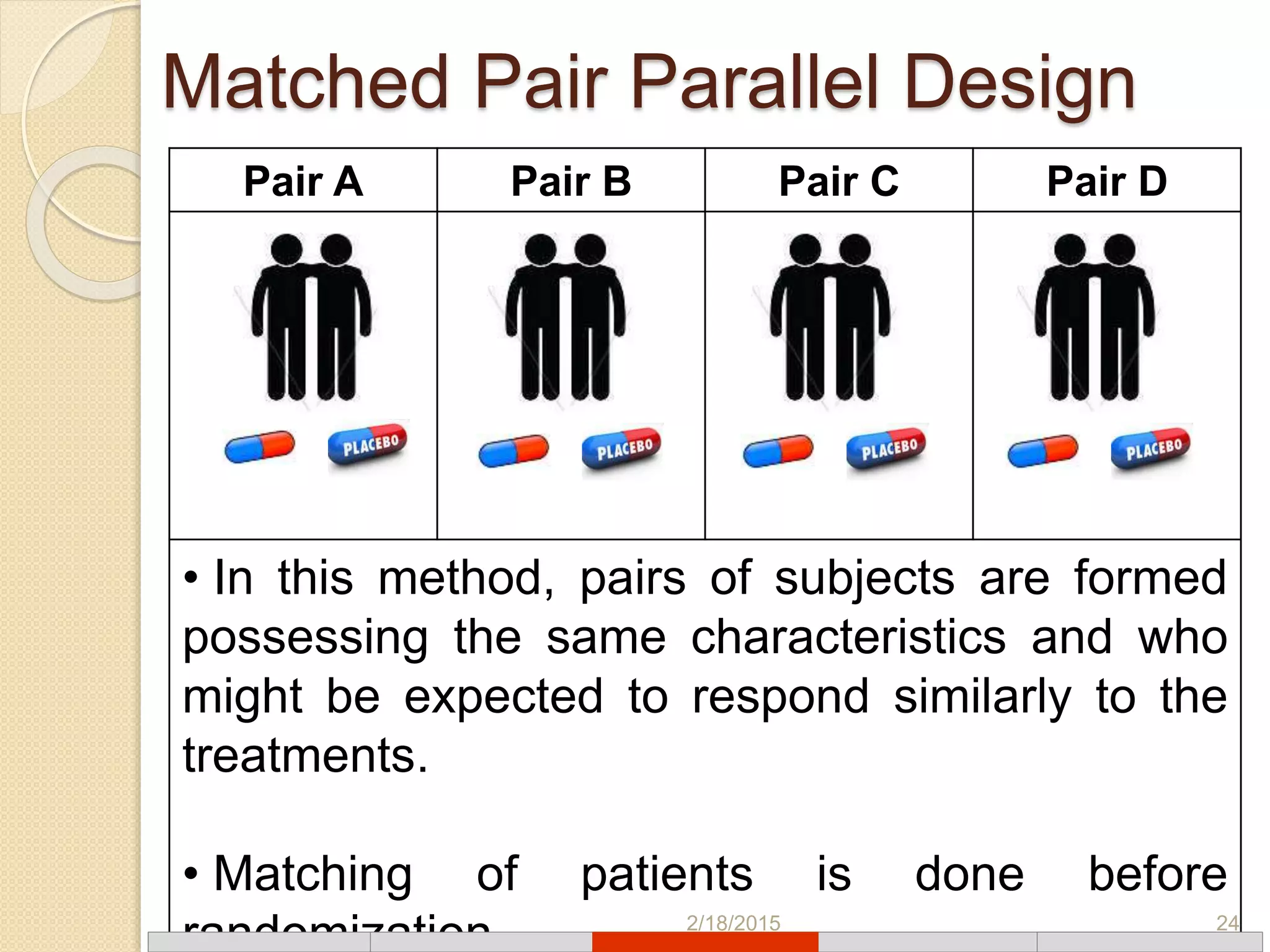
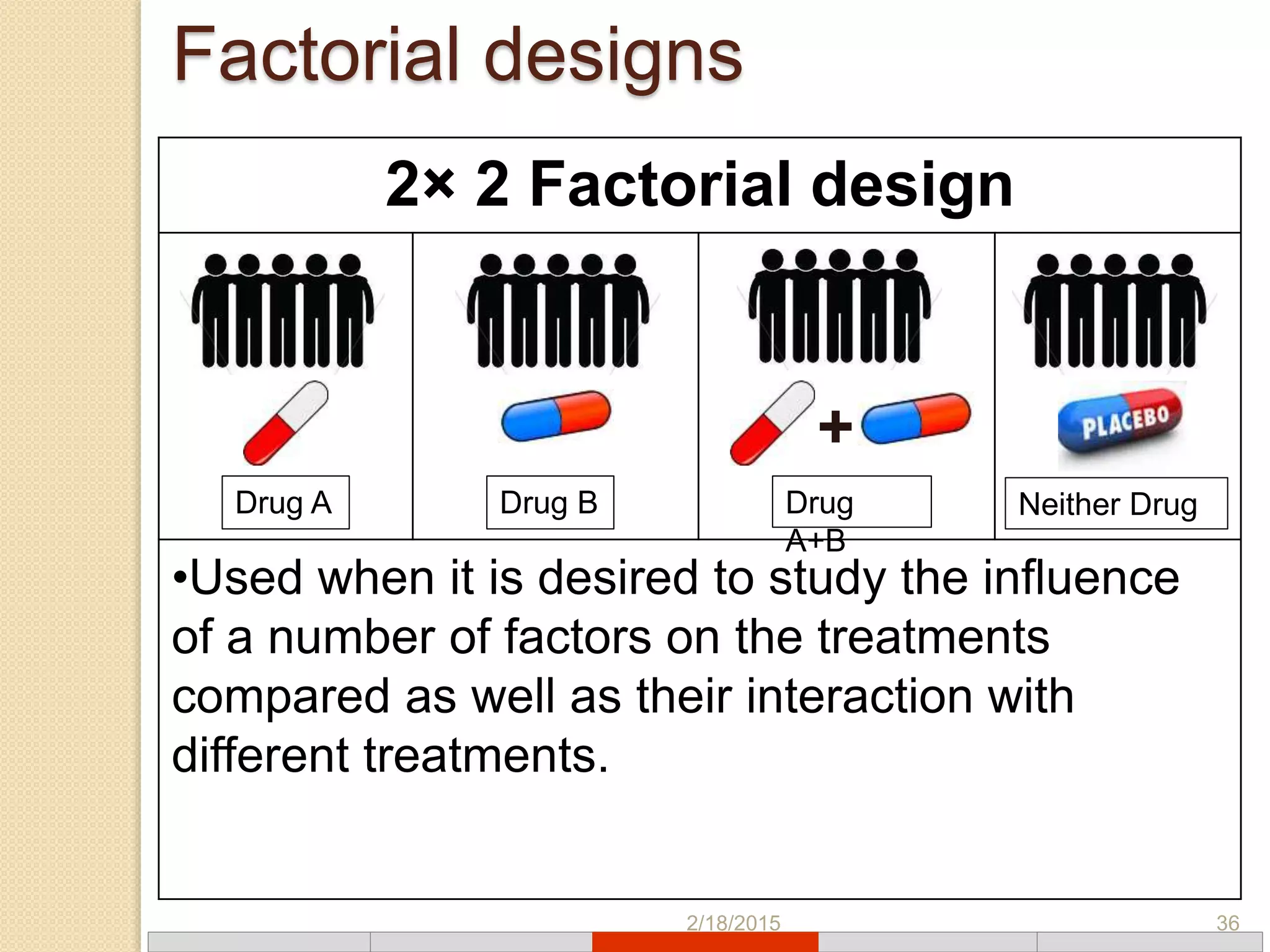
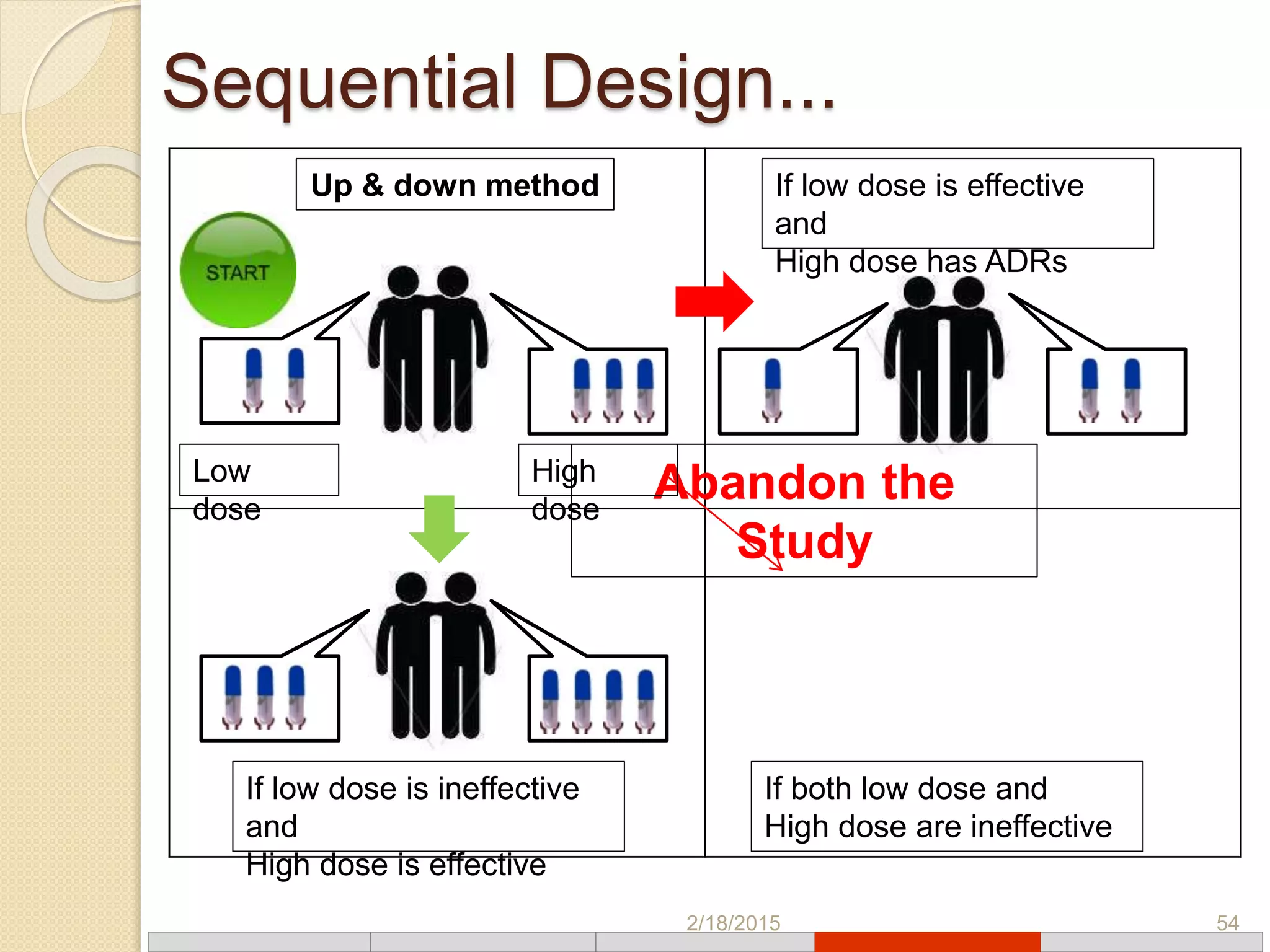
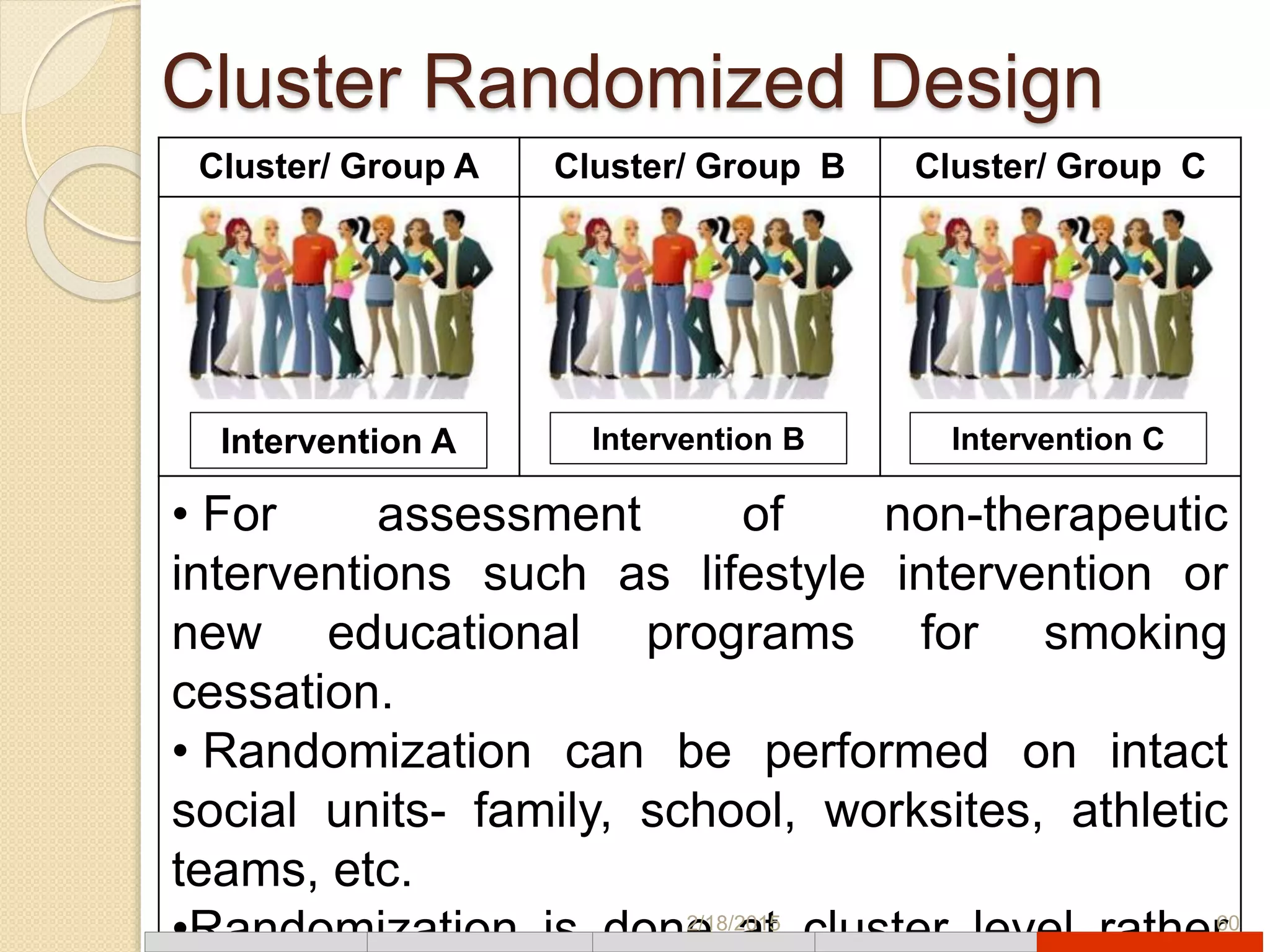
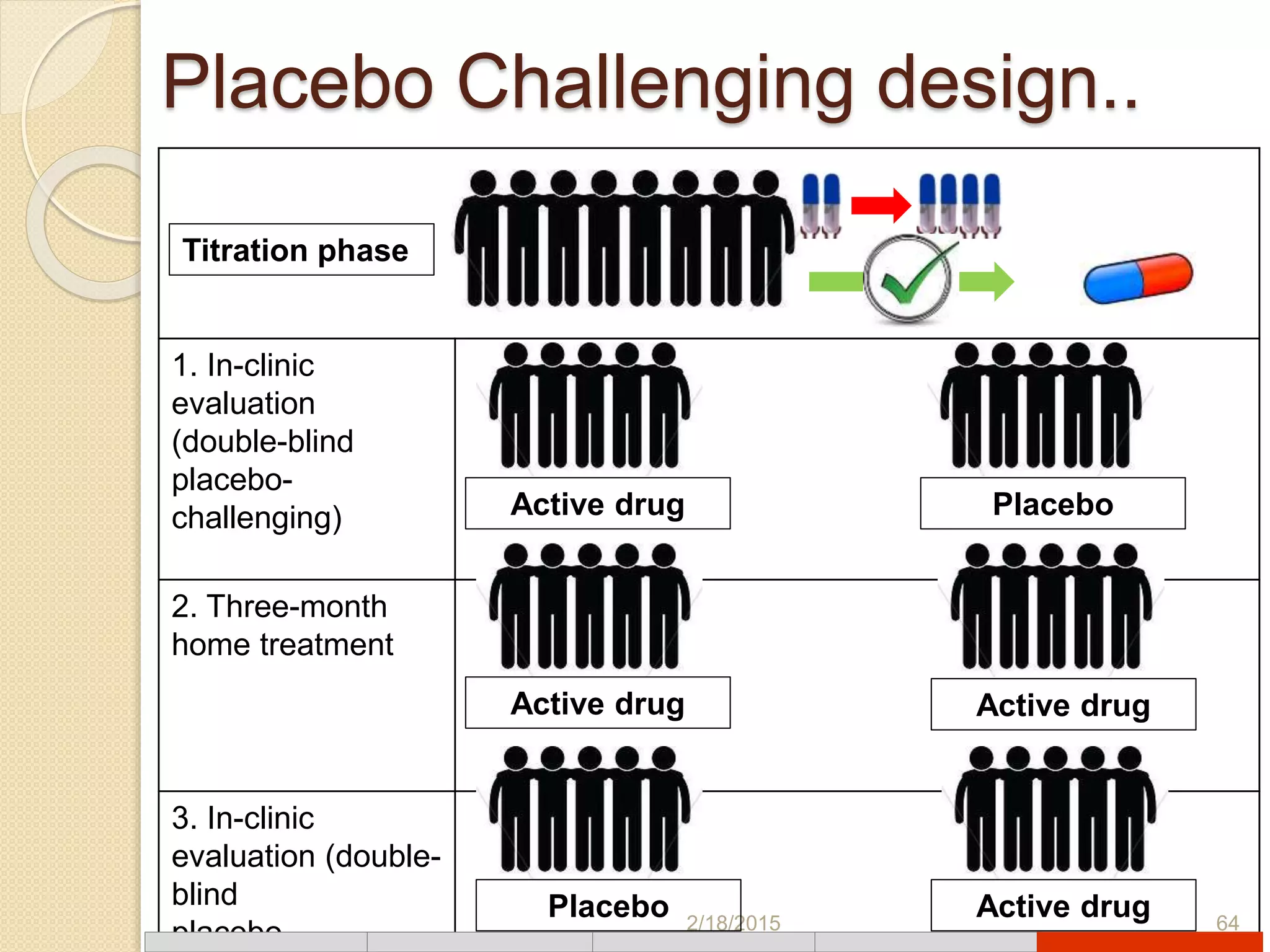
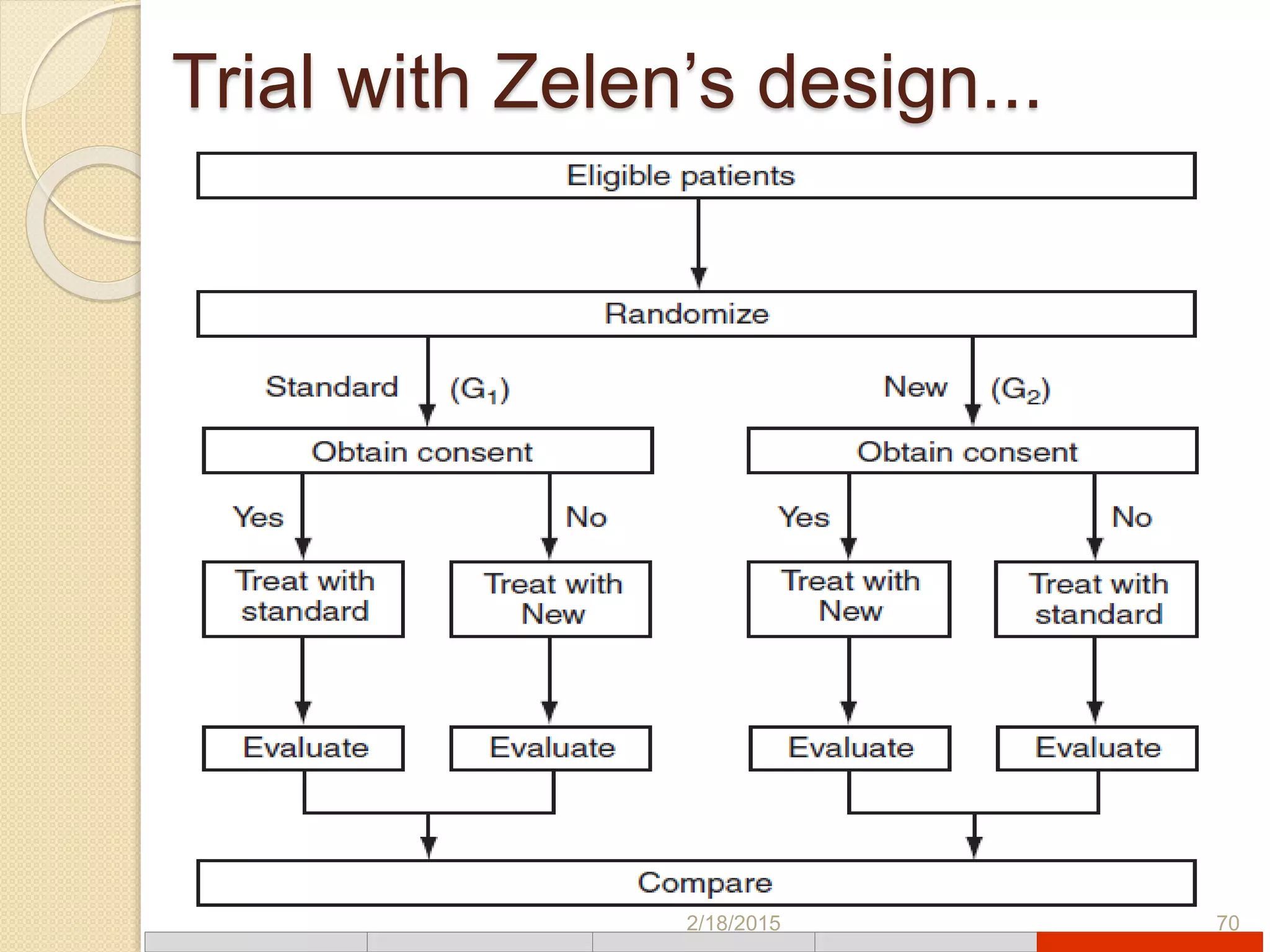
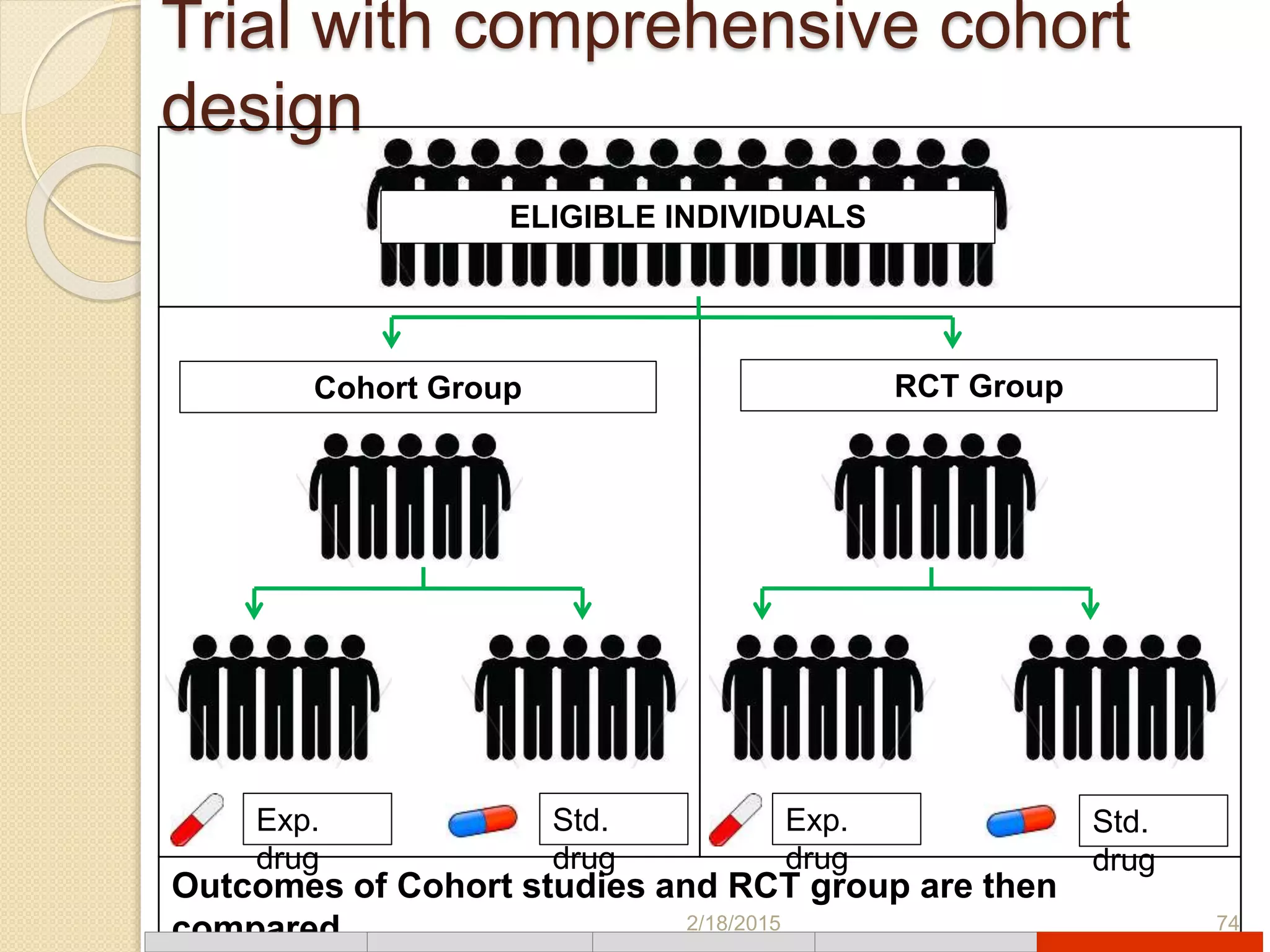
This document discusses various types of clinical trial designs. It begins by defining clinical trials and describing key elements like the PICO framework. It then covers ways to reduce bias through randomization and blinding. The document categorizes clinical trials based on factors like number of centers, control groups, randomization, and blinding. It provides details on traditional study designs like parallel group designs and crossover designs. It also discusses special designs for small populations and miscellaneous designs. Overall, the document provides an overview of different clinical trial designs, methods to reduce bias, and ways to categorize trial types.