Combined sci c5 syllabus
•Download as PPT, PDF•
1 like•305 views
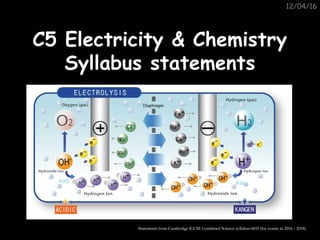
Electrolysis is the process by which ionic compounds break up into simpler substances when electricity is passed through them. Key terms include electrode, electrolyte, anode, and cathode. Students should be able to describe electrolysis in terms of the ions present and reactions at the electrodes. They should also be able to predict the products of electrolysis for molten lead(II) bromide, aqueous copper chloride, and other specified binary compounds in their molten states.
Report
Share
Report
Share
Recommended
Topic 5 syllabus statements

This document outlines statements from the Cambridge IGCSE Chemistry syllabus regarding electricity and chemistry. It describes key concepts such as defining electrolysis, describing the products and observations of electrolysis for various electrolytes, stating that metals form at the cathode and non-metals at the anode, relating products to electrolytes and electrodes, describing electrolysis in terms of ions and reactions, constructing half-equations, and predicting electrolysis products. It also describes electroplating, uses of materials in cables, the transfer of charge during electrolysis, producing electricity from cells, and manufacturing processes for aluminum and chlorine/hydrogen.
4s4 electrolysis( Part 1)

The document discusses key concepts in electrochemistry including electrolytes, conductors, electrolysis, and cations and anions. It defines an electrolyte as a substance that can conduct electricity due to the presence of freely moving ions. Conductors are able to conduct electricity but do not undergo chemical reactions. Electrolysis is the process of using a direct electric current to drive nonspontaneous chemical reactions through an electrolyte. During electrolysis, cations migrate to the cathode and anions migrate to the anode.
Electrolysis of molten compounds

Electrolysis is a process where an electrolyte is broken down into its constituent elements when electricity passes through. An electrolytic cell converts electrical energy to chemical energy using a battery, electrolyte, and two electrodes. During electrolysis, anions move toward the anode and cations move toward the cathode, where they are discharged by gaining or losing electrons.
C5 syllabus statements

This document outlines the key topics and concepts covered in a syllabus for a C5 Electricity & Chemistry course. It includes 9 points that describe electrolysis processes, products, and principles. Specifically, it covers how electrolysis breaks ionic compounds into simpler substances through the chemical effects of electricity. It also describes the use of electrodes, electrolytes, anodes and cathodes and how they relate to the reactions and products in different electrolysis examples, including molten salts, aqueous solutions, and metal refining. Finally, it mentions electroplating of metals and the industrial production of aluminum, chlorine, hydrogen and sodium hydroxide.
Electrolysis part 2: molten compound

1) Electrolysis of molten compounds involves passing an electric current through a molten compound, causing its ions to migrate to the electrodes.
2) During electrolysis of molten lead(II) bromide (PbBr2), the Pb2+ ions migrate to the cathode, where they gain electrons and deposit as metallic lead. Meanwhile, the Br- ions migrate to the anode, where they lose electrons and form a bromine gas product.
3) The overall reaction is the decomposition of PbBr2 into lead metal and bromine gas.
Electrolysis molten substances

Electrolytes are substances that can conduct electricity in the molten or liquid state and undergo chemical changes. Electrolysis is a process where electrolytes are broken down into their constituent elements by passing electricity through them. During electrolysis, ions migrate to the oppositely charged electrodes. At the anode, ions lose electrons and form gases or dissolve. At the cathode, ions gain electrons and form solid elements. Examples of electrolysis of molten lead(II) bromide and lead(II) oxide are described through their half reactions at the anode and cathode and overall reactions.
IGCSE Electricity

The document provides information about electrolysis, including:
1) Electrolysis is the chemical effect of electricity on ionic compounds, causing them to break up into simpler substances like elements.
2) During electrolysis, ions move to electrodes of opposite charge where chemical reactions occur - non-metals form at the anode and metals or hydrogen form at the cathode.
3) Examples of electrolysis include molten lead(II) bromide producing lead at the cathode and bromine at the anode, and aqueous copper(II) chloride producing copper at the cathode and chlorine at the anode.
Electricity & Chemistry

The document discusses electrolysis and the principles behind it. It explains that electrolysis involves passing electricity through an electrolyte, which causes chemical decomposition. Ions migrate to the electrodes and are discharged. Metals are formed at the cathode by reduction, while non-metals or oxygen form at the anode by oxidation. It provides examples of electrolysis such as molten salts like NaCl and aqueous solutions like copper sulfate. Factors affecting ion discharge are also discussed.
Recommended
Topic 5 syllabus statements

This document outlines statements from the Cambridge IGCSE Chemistry syllabus regarding electricity and chemistry. It describes key concepts such as defining electrolysis, describing the products and observations of electrolysis for various electrolytes, stating that metals form at the cathode and non-metals at the anode, relating products to electrolytes and electrodes, describing electrolysis in terms of ions and reactions, constructing half-equations, and predicting electrolysis products. It also describes electroplating, uses of materials in cables, the transfer of charge during electrolysis, producing electricity from cells, and manufacturing processes for aluminum and chlorine/hydrogen.
4s4 electrolysis( Part 1)

The document discusses key concepts in electrochemistry including electrolytes, conductors, electrolysis, and cations and anions. It defines an electrolyte as a substance that can conduct electricity due to the presence of freely moving ions. Conductors are able to conduct electricity but do not undergo chemical reactions. Electrolysis is the process of using a direct electric current to drive nonspontaneous chemical reactions through an electrolyte. During electrolysis, cations migrate to the cathode and anions migrate to the anode.
Electrolysis of molten compounds

Electrolysis is a process where an electrolyte is broken down into its constituent elements when electricity passes through. An electrolytic cell converts electrical energy to chemical energy using a battery, electrolyte, and two electrodes. During electrolysis, anions move toward the anode and cations move toward the cathode, where they are discharged by gaining or losing electrons.
C5 syllabus statements

This document outlines the key topics and concepts covered in a syllabus for a C5 Electricity & Chemistry course. It includes 9 points that describe electrolysis processes, products, and principles. Specifically, it covers how electrolysis breaks ionic compounds into simpler substances through the chemical effects of electricity. It also describes the use of electrodes, electrolytes, anodes and cathodes and how they relate to the reactions and products in different electrolysis examples, including molten salts, aqueous solutions, and metal refining. Finally, it mentions electroplating of metals and the industrial production of aluminum, chlorine, hydrogen and sodium hydroxide.
Electrolysis part 2: molten compound

1) Electrolysis of molten compounds involves passing an electric current through a molten compound, causing its ions to migrate to the electrodes.
2) During electrolysis of molten lead(II) bromide (PbBr2), the Pb2+ ions migrate to the cathode, where they gain electrons and deposit as metallic lead. Meanwhile, the Br- ions migrate to the anode, where they lose electrons and form a bromine gas product.
3) The overall reaction is the decomposition of PbBr2 into lead metal and bromine gas.
Electrolysis molten substances

Electrolytes are substances that can conduct electricity in the molten or liquid state and undergo chemical changes. Electrolysis is a process where electrolytes are broken down into their constituent elements by passing electricity through them. During electrolysis, ions migrate to the oppositely charged electrodes. At the anode, ions lose electrons and form gases or dissolve. At the cathode, ions gain electrons and form solid elements. Examples of electrolysis of molten lead(II) bromide and lead(II) oxide are described through their half reactions at the anode and cathode and overall reactions.
IGCSE Electricity

The document provides information about electrolysis, including:
1) Electrolysis is the chemical effect of electricity on ionic compounds, causing them to break up into simpler substances like elements.
2) During electrolysis, ions move to electrodes of opposite charge where chemical reactions occur - non-metals form at the anode and metals or hydrogen form at the cathode.
3) Examples of electrolysis include molten lead(II) bromide producing lead at the cathode and bromine at the anode, and aqueous copper(II) chloride producing copper at the cathode and chlorine at the anode.
Electricity & Chemistry

The document discusses electrolysis and the principles behind it. It explains that electrolysis involves passing electricity through an electrolyte, which causes chemical decomposition. Ions migrate to the electrodes and are discharged. Metals are formed at the cathode by reduction, while non-metals or oxygen form at the anode by oxidation. It provides examples of electrolysis such as molten salts like NaCl and aqueous solutions like copper sulfate. Factors affecting ion discharge are also discussed.
Electrolysis term 2 lss ace

The document discusses electrolysis, including its uses, basics, history, experiments conducted, and potential dangers. It describes two electrolysis experiments conducted at home - electrolysis of sodium chloride solution using copper electrodes, and electrolysis of sodium bicarbonate solution. Both experiments resulted in oxidation at the anode and reduction at the cathode. Potentially dangerous gases like chlorine and hydrogen were produced.
Electrolytic cells

Introduction to and mathematics of electrolytic cells, including electrolysis of water, sodium chloride solution and liquid sodium chloride.
Chapter 6 Electrochemistry

The document defines electrolytes as substances that conduct electricity in molten or aqueous solutions through freely moving ions. Electrolytes undergo chemical changes during electrolysis, which is the process of breaking down compounds into elements using an electric current. Non-electrolytes do not conduct electricity in any state and contain covalent molecules with no freely moving ions. Examples are given to classify substances as electrolytes or non-electrolytes based on their state and ability to conduct electricity.
Electrolysis revision

This document provides information about electrolysis. It discusses:
1. How electrolysis can be used to split up ionic compounds by melting them or dissolving them in water. Positive ions move to the negative cathode and negative ions move to the positive anode.
2. The rules for what products are formed at the electrodes during electrolysis of molten ionic compounds and ionic solutions. Hydrogen, metals, halogens, oxygen may be produced depending on the ions present.
3. Industrial applications of electrolysis including producing chlorine and sodium hydroxide from salt water, and purifying copper.
Electricity and chemistry class 10

This document discusses the electrolysis of brine solution (concentrated NaCl) to produce sodium hydroxide and chlorine gas. During electrolysis, sodium and hydrogen ions move to the cathode while chlorine and hydroxide ions move to the anode. At the cathode, only hydrogen ions are discharged to form hydrogen gas. At the anode, chlorine ions are discharged to form chlorine gas, leaving behind a solution of sodium hydroxide. A diaphragm cell is used to separately collect the chlorine gas and sodium hydroxide solution produced. Sodium hydroxide has uses including in soap production and paper making. Chlorine gas has uses as a bleaching agent and to produce hydrochlor
6.3 (a) electrolysis of an aqueous solution

The document discusses the electrolysis of aqueous solutions. It explains that during electrolysis, only one ion is selectively discharged at each electrode based on its position in the electrochemical series, the nature of the electrode, and the concentration of ions. The ion discharged at the anode depends on which is easier to oxidize, while the ion discharged at the cathode depends on which is easier to reduce. Different products are formed depending on these factors and the specific electrolyte used.
Experiment Form 4 Chapter 6 Electrochemistry

Experiment 6.8 investigates displacement reactions between metals and their salt solutions to construct an electrochemical series. The procedure involves placing different metal strips (magnesium, zinc, lead, copper) into separate salt solutions (magnesium nitrate, zinc nitrate, lead nitrate, copper nitrate). Observations are made to check for any color changes in the solutions, solid deposits on the metals, and metal dissolution. It is hypothesized that the greater the number of metals that can displace a metal from its salt solution, the higher its position will be in the electrochemical series.
Chapter 6 electrochemistry

The document discusses electrochemistry and electrolysis. It defines electrolytes and non-electrolytes, and explains how electrolytes can conduct electricity in molten or aqueous states through the movement of ions. Examples are given of electrolysis processes and how electrolysis can be used for metal extraction, purification, and electroplating.
Chapter 19.4 : Electrochemistry

The document provides an overview of key concepts in electrochemistry including:
1) The components and operation of electrochemical cells including voltaic cells like batteries and fuel cells as well as electrolytic cells.
2) Half-reactions, electrode potentials, and using these to determine spontaneity of redox reactions.
3) Processes like corrosion, electroplating, electrolysis of water, and recharging of batteries that involve redox reactions driven by electrical energy.
Electrochemistry

Includes a discussion of Voltaic and electrolytic cells, the Nernst equation and the relationship between electrochemical processes, chemical equilibrium and free energy.
**More good stuff available at:
www.wsautter.com
and
http://www.youtube.com/results?search_query=wnsautter&aq=f
Factors affecting electrolysis

Electrolysis is the process of using direct electrical current to cause a non-spontaneous chemical reaction. During electrolysis, ions conduct electricity and move towards the electrode that attracts them. The key factors that determine the products of electrolysis include:
1) The type of electrolyte, whether it is a molten salt or aqueous solution, determines which ions are present and can be reduced or oxidized.
2) An ion's position in the electrochemical series indicates its tendency to gain or lose electrons. Ions lower in the series are more readily reduced or oxidized.
3) The concentration of the electrolyte solution can allow ions higher in the electrochemical series to also be reduced or
Electrolysis

Electrolysis is an electrochemical process where an electric current is passed through an ionic substance like molten salt or electrolyte solution, causing a non-spontaneous chemical reaction to occur. During electrolysis, ions migrate to the electrodes where they undergo oxidation or reduction reactions. Cations migrate to the cathode and gain electrons through reduction. Anions migrate to the anode and lose electrons through oxidation. The document defines key terms related to electrolysis like electrodes, electrolyte, ions, and provides examples of the half-reactions and overall reaction that occur during the electrolysis of molten sodium chloride.
Electrolysis

Electrolysis is the decomposition of a substance by an electric current, where electrolytes carry current as ions in solution. During electrolysis, ions move to the electrodes and undergo oxidation or reduction reactions. At the cathode, electrons are gained and reduction occurs. At the anode, electrons are lost and oxidation occurs. The amount of substance deposited or gas produced can be calculated using Faraday's law, relating current, time, and moles of electrons in the electrode reactions.
Topic 7 Concept Electrolysis

The document describes the electrolysis of a copper chloride solution using a voltage of 6V. During the process, chlorine gas is produced at one electrode while solid copper forms at the other electrode, as electrons are involved in the transfer of copper and chlorine between electrode reactions. Key reactions include the formation of chlorine gas at the anode from chloride ions and copper metal at the cathode from copper ions gaining electrons.
ENGINEERING CHEMISTRY- Solved Model question paper,2017-18

This document contains the solved question paper for Engineering Chemistry. It discusses several topics:
1. The derivation of the Nernst equation for single electrode potential and its relationship to Gibbs free energy.
2. Concentration cells and calculating concentrations from cell potential.
3. The construction and working of a methanol-oxygen fuel cell.
4. The construction, working, and applications of lithium-ion batteries.
5. Key battery characteristics like cell potential, capacity, and cycle life.
6. The construction and advantages of a calomel reference electrode.
Electrolysis 

The three main factors that affect the electrolysis of an aqueous solution are:
1) The position of ions in the electrochemical series, which determines which ions will be reduced or oxidized at the electrodes.
2) The concentration of ions or electrolytes in the solution. Higher concentrations result in greater rates of reaction.
3) The type of electrodes used, as certain electrodes may dissolve and alter the composition of the solution during electrolysis.
Electrolysis molten sodium chloride

The document summarizes the process of electrolysis for two types of electrolytes: molten sodium chloride and aqueous electrolytes. For molten sodium chloride, sodium and chloride ions move to the cathode and anode, respectively, where they are discharged. Sodium forms at the cathode while chlorine gas forms at the anode. For aqueous electrolytes like dilute sulfuric acid, hydrogen, sulfate, and hydroxide ions are present and migrate to the electrodes where hydrogen gas forms at the cathode and oxygen gas forms at the anode.
Electrolysis chemist

Electrolysis is the process of using electricity to cause a non-spontaneous chemical reaction. During electrolysis, ions migrate towards the electrodes and undergo oxidation or reduction reactions. At the cathode, positively charged ions are reduced and neutral atoms are formed. At the anode, negatively charged ions are oxidized and neutral atoms or molecules are formed. The products of electrolysis depend on factors like the ions present in the electrolyte, their concentration and position in the reactivity series, and the nature of the electrodes. Electrolysis has applications in purifying and extracting metals, and in electroplating.
ELECTROCHEMSTRY POWER POINT

Electrolysis is the process of using electric current to cause non-spontaneous chemical changes. During electrolysis, ions are discharged at the electrodes. The key factors that determine which ions are discharged include the position of ions in the electrochemical series, concentration of ions, and type of electrode. Electrolysis has important industrial applications such as electroplating, metal purification, and metal extraction.
Introduction to electrolysis(older)

This document provides definitions and explanations related to electrolysis. It defines key terms like electrolyte, electrode, anode, and cathode. It explains that electrolysis involves the decomposition of ionic compounds through the movement of ions when a current is passed. Molten salts and those dissolved in solutions conduct electricity because the ions are free to move. Simple experiments are described to demonstrate electrolysis, including the electrolysis of molten lead(II) bromide. Equations are given for the reactions at the anode and cathode.
Combined sci c4 syllabus

The document outlines 7 statements from the Cambridge IGCSE Combined Science syllabus regarding C4 Stoichiometry. It covers writing formulas for simple compounds using element symbols, deducing formulas from relative atom numbers or models, determining formulas for ionic compounds from ion charges, constructing and using word and symbolic equations including ionic equations, and deducing balanced chemical reactions from relevant information.
Combined sci c6 syllabus

Chemical reactions can be exothermic or endothermic depending on whether they release or absorb energy in the form of heat. Exothermic reactions release energy as heat while endothermic reactions absorb heat. The document outlines syllabus statements relating to the energetics of chemical reactions, specifically defining exothermic and endothermic changes and how they relate to the transformation between chemical and thermal energy.
More Related Content
What's hot
Electrolysis term 2 lss ace

The document discusses electrolysis, including its uses, basics, history, experiments conducted, and potential dangers. It describes two electrolysis experiments conducted at home - electrolysis of sodium chloride solution using copper electrodes, and electrolysis of sodium bicarbonate solution. Both experiments resulted in oxidation at the anode and reduction at the cathode. Potentially dangerous gases like chlorine and hydrogen were produced.
Electrolytic cells

Introduction to and mathematics of electrolytic cells, including electrolysis of water, sodium chloride solution and liquid sodium chloride.
Chapter 6 Electrochemistry

The document defines electrolytes as substances that conduct electricity in molten or aqueous solutions through freely moving ions. Electrolytes undergo chemical changes during electrolysis, which is the process of breaking down compounds into elements using an electric current. Non-electrolytes do not conduct electricity in any state and contain covalent molecules with no freely moving ions. Examples are given to classify substances as electrolytes or non-electrolytes based on their state and ability to conduct electricity.
Electrolysis revision

This document provides information about electrolysis. It discusses:
1. How electrolysis can be used to split up ionic compounds by melting them or dissolving them in water. Positive ions move to the negative cathode and negative ions move to the positive anode.
2. The rules for what products are formed at the electrodes during electrolysis of molten ionic compounds and ionic solutions. Hydrogen, metals, halogens, oxygen may be produced depending on the ions present.
3. Industrial applications of electrolysis including producing chlorine and sodium hydroxide from salt water, and purifying copper.
Electricity and chemistry class 10

This document discusses the electrolysis of brine solution (concentrated NaCl) to produce sodium hydroxide and chlorine gas. During electrolysis, sodium and hydrogen ions move to the cathode while chlorine and hydroxide ions move to the anode. At the cathode, only hydrogen ions are discharged to form hydrogen gas. At the anode, chlorine ions are discharged to form chlorine gas, leaving behind a solution of sodium hydroxide. A diaphragm cell is used to separately collect the chlorine gas and sodium hydroxide solution produced. Sodium hydroxide has uses including in soap production and paper making. Chlorine gas has uses as a bleaching agent and to produce hydrochlor
6.3 (a) electrolysis of an aqueous solution

The document discusses the electrolysis of aqueous solutions. It explains that during electrolysis, only one ion is selectively discharged at each electrode based on its position in the electrochemical series, the nature of the electrode, and the concentration of ions. The ion discharged at the anode depends on which is easier to oxidize, while the ion discharged at the cathode depends on which is easier to reduce. Different products are formed depending on these factors and the specific electrolyte used.
Experiment Form 4 Chapter 6 Electrochemistry

Experiment 6.8 investigates displacement reactions between metals and their salt solutions to construct an electrochemical series. The procedure involves placing different metal strips (magnesium, zinc, lead, copper) into separate salt solutions (magnesium nitrate, zinc nitrate, lead nitrate, copper nitrate). Observations are made to check for any color changes in the solutions, solid deposits on the metals, and metal dissolution. It is hypothesized that the greater the number of metals that can displace a metal from its salt solution, the higher its position will be in the electrochemical series.
Chapter 6 electrochemistry

The document discusses electrochemistry and electrolysis. It defines electrolytes and non-electrolytes, and explains how electrolytes can conduct electricity in molten or aqueous states through the movement of ions. Examples are given of electrolysis processes and how electrolysis can be used for metal extraction, purification, and electroplating.
Chapter 19.4 : Electrochemistry

The document provides an overview of key concepts in electrochemistry including:
1) The components and operation of electrochemical cells including voltaic cells like batteries and fuel cells as well as electrolytic cells.
2) Half-reactions, electrode potentials, and using these to determine spontaneity of redox reactions.
3) Processes like corrosion, electroplating, electrolysis of water, and recharging of batteries that involve redox reactions driven by electrical energy.
Electrochemistry

Includes a discussion of Voltaic and electrolytic cells, the Nernst equation and the relationship between electrochemical processes, chemical equilibrium and free energy.
**More good stuff available at:
www.wsautter.com
and
http://www.youtube.com/results?search_query=wnsautter&aq=f
Factors affecting electrolysis

Electrolysis is the process of using direct electrical current to cause a non-spontaneous chemical reaction. During electrolysis, ions conduct electricity and move towards the electrode that attracts them. The key factors that determine the products of electrolysis include:
1) The type of electrolyte, whether it is a molten salt or aqueous solution, determines which ions are present and can be reduced or oxidized.
2) An ion's position in the electrochemical series indicates its tendency to gain or lose electrons. Ions lower in the series are more readily reduced or oxidized.
3) The concentration of the electrolyte solution can allow ions higher in the electrochemical series to also be reduced or
Electrolysis

Electrolysis is an electrochemical process where an electric current is passed through an ionic substance like molten salt or electrolyte solution, causing a non-spontaneous chemical reaction to occur. During electrolysis, ions migrate to the electrodes where they undergo oxidation or reduction reactions. Cations migrate to the cathode and gain electrons through reduction. Anions migrate to the anode and lose electrons through oxidation. The document defines key terms related to electrolysis like electrodes, electrolyte, ions, and provides examples of the half-reactions and overall reaction that occur during the electrolysis of molten sodium chloride.
Electrolysis

Electrolysis is the decomposition of a substance by an electric current, where electrolytes carry current as ions in solution. During electrolysis, ions move to the electrodes and undergo oxidation or reduction reactions. At the cathode, electrons are gained and reduction occurs. At the anode, electrons are lost and oxidation occurs. The amount of substance deposited or gas produced can be calculated using Faraday's law, relating current, time, and moles of electrons in the electrode reactions.
Topic 7 Concept Electrolysis

The document describes the electrolysis of a copper chloride solution using a voltage of 6V. During the process, chlorine gas is produced at one electrode while solid copper forms at the other electrode, as electrons are involved in the transfer of copper and chlorine between electrode reactions. Key reactions include the formation of chlorine gas at the anode from chloride ions and copper metal at the cathode from copper ions gaining electrons.
ENGINEERING CHEMISTRY- Solved Model question paper,2017-18

This document contains the solved question paper for Engineering Chemistry. It discusses several topics:
1. The derivation of the Nernst equation for single electrode potential and its relationship to Gibbs free energy.
2. Concentration cells and calculating concentrations from cell potential.
3. The construction and working of a methanol-oxygen fuel cell.
4. The construction, working, and applications of lithium-ion batteries.
5. Key battery characteristics like cell potential, capacity, and cycle life.
6. The construction and advantages of a calomel reference electrode.
Electrolysis 

The three main factors that affect the electrolysis of an aqueous solution are:
1) The position of ions in the electrochemical series, which determines which ions will be reduced or oxidized at the electrodes.
2) The concentration of ions or electrolytes in the solution. Higher concentrations result in greater rates of reaction.
3) The type of electrodes used, as certain electrodes may dissolve and alter the composition of the solution during electrolysis.
Electrolysis molten sodium chloride

The document summarizes the process of electrolysis for two types of electrolytes: molten sodium chloride and aqueous electrolytes. For molten sodium chloride, sodium and chloride ions move to the cathode and anode, respectively, where they are discharged. Sodium forms at the cathode while chlorine gas forms at the anode. For aqueous electrolytes like dilute sulfuric acid, hydrogen, sulfate, and hydroxide ions are present and migrate to the electrodes where hydrogen gas forms at the cathode and oxygen gas forms at the anode.
Electrolysis chemist

Electrolysis is the process of using electricity to cause a non-spontaneous chemical reaction. During electrolysis, ions migrate towards the electrodes and undergo oxidation or reduction reactions. At the cathode, positively charged ions are reduced and neutral atoms are formed. At the anode, negatively charged ions are oxidized and neutral atoms or molecules are formed. The products of electrolysis depend on factors like the ions present in the electrolyte, their concentration and position in the reactivity series, and the nature of the electrodes. Electrolysis has applications in purifying and extracting metals, and in electroplating.
ELECTROCHEMSTRY POWER POINT

Electrolysis is the process of using electric current to cause non-spontaneous chemical changes. During electrolysis, ions are discharged at the electrodes. The key factors that determine which ions are discharged include the position of ions in the electrochemical series, concentration of ions, and type of electrode. Electrolysis has important industrial applications such as electroplating, metal purification, and metal extraction.
Introduction to electrolysis(older)

This document provides definitions and explanations related to electrolysis. It defines key terms like electrolyte, electrode, anode, and cathode. It explains that electrolysis involves the decomposition of ionic compounds through the movement of ions when a current is passed. Molten salts and those dissolved in solutions conduct electricity because the ions are free to move. Simple experiments are described to demonstrate electrolysis, including the electrolysis of molten lead(II) bromide. Equations are given for the reactions at the anode and cathode.
What's hot (20)
ENGINEERING CHEMISTRY- Solved Model question paper,2017-18

ENGINEERING CHEMISTRY- Solved Model question paper,2017-18
Viewers also liked
Combined sci c4 syllabus

The document outlines 7 statements from the Cambridge IGCSE Combined Science syllabus regarding C4 Stoichiometry. It covers writing formulas for simple compounds using element symbols, deducing formulas from relative atom numbers or models, determining formulas for ionic compounds from ion charges, constructing and using word and symbolic equations including ionic equations, and deducing balanced chemical reactions from relevant information.
Combined sci c6 syllabus

Chemical reactions can be exothermic or endothermic depending on whether they release or absorb energy in the form of heat. Exothermic reactions release energy as heat while endothermic reactions absorb heat. The document outlines syllabus statements relating to the energetics of chemical reactions, specifically defining exothermic and endothermic changes and how they relate to the transformation between chemical and thermal energy.
Reactions & Formulas

This is an example of a combustion reaction. Propane (C3H8) is burning in oxygen gas (O2), producing carbon dioxide (CO2) and water vapor (H2O) as products. The balanced chemical equation is:
C3H8 + 5O2 → 3CO2 + 4H2O
Combined sci c8 syllabus

This document outlines the syllabus statements from the Cambridge IGCSE Combined Science syllabus 0653 for exams from 2016 to 2018 regarding acids, bases and salts. It describes the key properties of acids and bases, including pH and characteristic reactions. It also covers the preparation, separation and purification of salts, methods for identifying common cations and anions using specific tests, and the identification of several common gases.
Topic 3 syllabus statements

This document outlines statements from the Cambridge IGCSE Chemistry syllabus regarding atomic structure, elements, compounds, and bonding. It defines key terms like atomic number, mass number, isotopes, ions, ionic bonds, covalent bonds, and metallic bonding. It describes the electronic structure of atoms and how different types of bonds are formed between elements. It also addresses the properties and structures of ionic compounds, covalent molecules, macromolecules, metals, and alloys.
Energy change during chemical rcts

1. Exothermic reactions release heat energy to their surroundings, causing an increase in temperature. Reactions like burning fuels and explosions are exothermic.
2. Endothermic reactions absorb heat energy from their surroundings, causing a decrease in temperature. Dissolving salts in water is an example of an endothermic process.
3. Whether a reaction is exothermic or endothermic depends on whether more energy is released or absorbed during bond breaking and forming. Exothermic reactions release more energy than they absorb.
Combined sci c3 syllabus

This document contains statements from the Cambridge IGCSE Combined Science syllabus 0653 covering topics on atoms, elements, compounds, mixtures, atomic structure, the periodic table, ions, ionic bonds, molecules, and covalent bonds. Specifically, it outlines key concepts students should understand, such as the differences between physical and chemical changes, elements, compounds and mixtures. It also describes atomic structure, the periodic table, ion formation, ionic bonding, covalent bonding and drawing dot-and-cross diagrams to represent covalent molecular structures.
4 ecology syllabus statements

1) The document discusses key concepts in ecology including species, communities, ecosystems, energy flow and carbon cycling. It provides definitions and explanations of these core topics.
2) A central idea is that the continued survival of living organisms relies on sustainable communities and ecosystems, and that ecosystems require continuous supplies of energy and carbon to sustain life processes.
3) The document gives guidance on classifying species and on quantitative representations of energy flow between trophic levels using pyramids.
Atomic structure & bonding

1. The document discusses atomic structure and bonding. It describes the structure of atoms in terms of electrons and the nucleus containing protons and neutrons. It also discusses the arrangement of electrons in shells and the significance of noble gas structures and valency electrons.
2. The document then covers the periodic table and periodic trends. It defines properties such as atomic number, mass number, and isotopes. It explains how the periodic table is arranged based on these properties and how elements in the same group have similar properties.
3. The types of bonding are described including ionic bonding between metals and non-metals which forms ionic lattices, and covalent bonding between non-metals which forms molecules by sharing electron
The periodic table and identification of ions

The document summarizes key concepts about the periodic table, identification of ions and gases. It describes trends in properties for groups I and VII, such as how reactivity increases down group I due to electron shielding. Tests are outlined to identify aqueous cations, anions and common gases. Uses of transition metals and noble gases are briefly mentioned.
Reactions of acids

The document is about acids and alkalis and their reactions. It discusses how acids and alkalis react to form salts and water through a neutralization reaction. It provides examples of reactions between acids and bases like metals, metal oxides, and carbonates. The document also covers acid rain, how it is formed from air pollution, and its environmental effects. It describes methods to reduce acid rain by limiting the emissions of sulfur dioxide and nitrogen oxides.
4.2 energy flow

Here is a 6 mark outline of how energy passes through ecosystems:
1. Light energy from the sun is captured by producers like plants through photosynthesis.
2. This light energy is converted to chemical energy stored in carbohydrates.
3. Primary consumers eat producers and obtain this chemical energy.
4. At each trophic level as organisms eat one another, only about 10% of the energy is transferred between trophic levels with the rest lost as heat or through feces and waste.
5. This energy loss at each level limits the length of food chains and biomass at higher trophic levels due to decreasing energy availability.
6. Heat energy produced during respiration cannot be reused by organisms and
The Chi-Squared Test

The document discusses the benefits of exercise for mental health. Regular physical activity can help reduce anxiety and depression and improve mood and cognitive functioning. Exercise causes chemical changes in the brain that may help boost feelings of calmness, happiness and focus.
Acids, Bases and Salts (Chemistry 'O' level)

Chemistry 'O' level syllabus: Acids, Bases & Salts
Please download to access the animations that come with the slides. Some of the animations help to clear away hidden boxes.
Slides include interactive learning elements!!!
Contact me if you want more slides or to custom made for you at:
Email: fazzydoo@gmail.com
Facebook: Faiz Abdullah (look at picture)
Twitter: @fazzydoo
Viewers also liked (14)
Similar to Combined sci c5 syllabus
Electrical properties of solids

This document discusses the electrical properties of solid inorganic materials. It begins by defining solid electrolytes as crystalline solids that conduct electricity via the movement of ions. Some key solid electrolyte materials discussed include silver iodide (AgI), sodium beta-alumina, and lithium cobalt oxide (LiCoO2). Applications of solid electrolytes mentioned include use in solid oxide fuel cells, lithium-ion batteries, oxygen gas sensors, and as separators in electrochemical cells.
F.Sc. Part 1 Chemistry.Ch.10.Test (Malik Xufyan)

The document contains information about chemistry test series books published by Malik Xufyan of JIAS Academy, Jhang Institute for Advanced Studies. It lists the titles of 9 books covering chemistry courses from 9th class to F.Sc. Part II, which are available in both chapter-wise and board paper-wise test series formats. It also provides the contact information of the publisher.
Electrochemistry 

This document provides an overview of electrochemistry. It discusses key topics like what electrochemistry is, the history and founders of electrochemistry, oxidation-reduction reactions, balancing redox equations, standard electrode potential, the Nernst equation, batteries, corrosion, electrolysis, Faraday's laws of electrolysis, and more. The document serves as a high-level introduction to many fundamental concepts in electrochemistry.
Excited state and quantum numbers

This document provides information on electron configurations and quantum numbers:
- It defines the principal quantum number n, which describes the main energy level of an electron. It also defines the subsidiary quantum number l, which describes the shape of an orbital, and the magnetic quantum number m, which describes the orientation of an orbital in space.
- It discusses the four quantum numbers that describe each electron in an atom: n, l, m, and spin quantum number s.
- It provides examples of writing the four quantum numbers for the last electron in different atoms like Li, Cl, B, O, and Ar.
- It includes sample exam questions that test understanding of electron configurations, ion formations, bonding definitions
Electrochemistry

This document provides an overview of electrochemistry. It begins by defining electrochemistry as the study of chemical reactions at the interface of an electrode and electrolyte involving the interaction of electrical and chemical changes. The document then discusses the history and founders of electrochemistry, including Faraday's two laws of electrolysis. It explains key concepts such as oxidation-reduction reactions, balancing redox equations, and the Nernst equation. The document also covers applications including batteries, corrosion, electrolysis, and branches of electrochemistry like bioelectrochemistry and nanoelectrochemistry.
Unit_I_Electrochemistry.ppt

Introduction – cells – types - representation of galvanic cell - electrode potential - Nernst equation (derivation of cell EMF) - calculation of cell EMF from single electrode potential - reference electrode: construction, working and applications of standard hydrogen electrode, standard calomel electrode - glass electrode – EMF series and its applications - potentiometric titrations (redox) - conductometric titrations - mixture of weak and strong acid vs strong base.
pk-chourasia-electrochemistry-1.pptx

Here are the answers to the questions:
1. The salt bridge completes the circuit by allowing the flow of ions between the half-cells without physical mixing of the solutions. The ions from the salt bridge maintain electroneutrality in each half-cell.
2. Electrode potential is the tendency of an element to gain or lose electrons when it is placed in contact with its ions. It is measured in volts.
3. In an electrochemical cell, the anode is negative because oxidation occurs at the anode where electrons are lost. The cathode is positive because reduction occurs at the cathode where electrons are gained. Electrons always flow from the negative to the positive electrode.
4. The factors required
Electrochemistry and batteries

Galvanic cell, Nernst equation, Calomel electrode, Quinhydrone electrode, Glass electrode, Dry cell, Lead Acid battery, Li ion cell, Methanol oxygen fuel cell
electrolysis multiple choice

This document is a chemistry exam question paper focusing on electricity and chemistry. It contains 44 multiple choice questions testing understanding of key concepts such as:
- Substances used in electrical cables and why
- Products of electrolysis of various solutions and molten salts
- Electroplating and electrorefining processes
- Use of electrolysis in industrial processes like metal extraction
Electrode material for battery in automobile

Title: Advancements in Electrode Materials for Automotive Batteries: A Comprehensive Review
Abstract:
The automotive industry is rapidly transitioning towards electric propulsion systems to mitigate environmental impacts and reduce dependency on fossil fuels. Central to this shift are advancements in battery technology, particularly in electrode materials, which play a critical role in determining battery performance, energy density, and lifespan. This comprehensive review explores the latest developments in electrode materials for automotive batteries, encompassing lithium-ion, solid-state, and beyond lithium-ion technologies. We delve into the fundamental principles governing electrode material selection, discuss current challenges, and analyze emerging trends such as silicon-based anodes, sulfur cathodes, and solid electrolytes. Through an extensive examination of recent research and commercial developments, we provide insights into the future direction of electrode materials for automotive batteries, highlighting key areas for further research and innovation.
1. Introduction:
- Overview of the importance of electrode materials in automotive batteries
- Transition towards electric vehicles (EVs) and the role of batteries
- Purpose and scope of the review
2. Fundamentals of Battery Electrodes:
- Electrochemical principles underlying battery operation
- Role of electrodes in battery performance
- Requirements for automotive applications: energy density, power density, longevity, and safety
3. Lithium-Ion Batteries:
- Overview of lithium-ion battery architecture
- Current electrode materials: graphite anodes, lithium cobalt oxide (LCO), lithium iron phosphate (LFP), etc.
- Challenges and limitations: capacity degradation, safety concerns, resource availability
- Recent advancements in electrode materials for lithium-ion batteries
4. Beyond Lithium-Ion Batteries:
- Need for higher energy density and sustainability
- Emerging alternatives: lithium-sulfur (Li-S), lithium-air (Li-O2), sodium-ion (Na-ion), potassium-ion (K-ion) batteries
- Electrode materials for non-lithium systems: sulfur cathodes, sodium-ion anodes, etc.
- Comparative analysis of different beyond lithium-ion technologies
5. Silicon-Based Anodes:
- Potential of silicon as a high-capacity anode material
- Challenges: volume expansion, cycling stability, Coulombic efficiency
- Strategies to mitigate silicon anode limitations: nanostructuring, alloying, coatings
- Progress in commercialization and integration into automotive batteries
6. Solid-State Batteries:
- Advantages of solid-state electrolytes over liquid electrolytes
- Materials for solid-state electrolytes: sulfides, oxides, polymers
- Solid-state electrode materials: lithium metal, sulfides, etc.
- Recent breakthroughs in solid-state battery technology and their implications for automotive applications
7. Challenges and Opportunities:
- Scalability
AD5585E436EE38423C9C5B5AD75FD8FD.pptx

This document provides an introduction to electrolysis. It defines electrolysis as the process where ionic substances are broken down into simpler substances using electricity. The document then outlines several hands-on activities for students to learn about and describe the process of electrolysis, including defining key terms, writing word and balanced chemical equations, and explaining the movement of ions during electrolysis.
Basic Fundamentals of Fuel Cells

This document summarizes a seminar presentation about fuel cells. It begins with an introduction that defines fuel cells and batteries. It then describes the basic components and chemical reactions of different types of fuel cells, including hydrogen-oxygen, molten carbonate, PEM, and hydrocarbon-oxygen fuel cells. Applications of fuel cells currently include buses and cars that run on hydrogen fuel cells. With further technological advancements, fuel cells could potentially be used more widely for clean energy in industries and to power electronic devices. However, fuel cells also have limitations like high costs and needing specific operating conditions.
Unit 5 elektrokimia

1. The document discusses electrolysis and differentiates between conductors, electrolytes, and non-electrolytes based on their ability to conduct electricity and undergo chemical changes.
2. It describes the components of an electrolytic cell including the electrodes (anode and cathode) and explains that electrolysis is the process of decomposing an electrolyte into its constituent elements using an electric current.
3. The summary explains that during electrolysis, electric energy is converted to chemical energy as ions in the electrolyte migrate to the electrodes. At the anode, anions are discharged and at the cathode, cations are discharged.
ORR

The presentation discusses the working principle and applications of fuel cells, the oxygen reduction reaction (ORR) in fuel cells, and various electrocatalysts used for ORR. It outlines the working principle of fuel cells, where hydrogen is oxidized at the anode to produce protons and electrons. At the cathode, oxygen reacts with protons and electrons to form water. Various electrocatalysts discussed that catalyze the ORR include platinum, carbon materials like graphite and nanotubes, transition metals and their compounds. The presentation provides details on the mechanisms of ORR on different catalysts and how doping or alloying can influence their activity.
Chemistry perfect-score-module-form-4-set-3

1) Electrolysis involves passing an electric current through an electrolyte to cause non-spontaneous redox reactions to occur at the electrodes.
2) Products of electrolysis depend on the nature of the electrolyte and electrodes. Selective discharge of ions occurs based on their position in the electrochemical series.
3) Voltaic cells involve spontaneous redox reactions that generate electricity, with electrons flowing from the negative to the positive terminal.
Batteries ppt

This document discusses novel materials for batteries. It begins by introducing solid state batteries and the requirements for electrode materials, including low working potential, high specific capacity, good interface with electrolytes, and high electrode kinetics. It then discusses various materials that could be used as electrodes, including lithium carbon electrodes using graphite and graphite intercalation compounds. Different types of graphite like natural, synthetic, and HOPG are described. The document also discusses intercalation of lithium ions into carbon and potential carbon-sodium electrodes. Finally, it discusses various material classes like rutile, perovskite, and spinel materials that could be used as cathodes in rechargeable lithium ion batteries. Specific
Updated ppt notes Unit-2_compressed_ec31d11e401e3dd93bf67ff9cda06486 (1).pdf

The document provides information about instrumental methods of analysis taught as part of the Engineering Chemistry course at Sinhgad Institute of Technology, Lonavala. It discusses topics like conductometry, pHmetry and different types of electrodes - reference electrodes like calomel electrode and indicator electrodes like glass electrode. The key details provided include the course contents, objectives, outcomes, introduction to various analytical techniques and applications of instrumental methods in industries, agriculture, environment etc. Important concepts around electrode potential, measurement of potential using reference electrodes and construction of calomel electrode with its cell representation are also summarized.
AP_Chapter_18_Electrochemistry.ppt

The document provides information about electrochemistry. It discusses oxidation-reduction reactions and how they involve the transfer of electrons between species. It explains how to assign oxidation numbers to keep track of electrons gained and lost. Balancing oxidation-reduction reactions using the half-reaction method is also covered. Finally, the document discusses voltaic cells, electrolytic cells, and applications of electrochemistry such as electroplating.
engineering material atomic structure of atom Lecture2.

1) The document describes the structure of materials at different length scales from atomic to macroscopic levels. It discusses how atomic structure influences properties and technological applications of materials.
2) Key concepts covered include the structure of the atom, electronic configuration, periodic table, different types of atomic bonding, and how bonding influences properties like conductivity and strength.
3) Examples calculate the number of atoms in materials and compare properties like electronegativity between elements. Diagrams illustrate different types of bonding and how structure determines properties.
Semiconductor

This slide give you idea about the atomic structure, classification of solids based on valance electron, free electron, energy band description, why the silicon is used as semiconductor substance compare to germanium, semiconductor and its types.
Similar to Combined sci c5 syllabus (20)
Updated ppt notes Unit-2_compressed_ec31d11e401e3dd93bf67ff9cda06486 (1).pdf

Updated ppt notes Unit-2_compressed_ec31d11e401e3dd93bf67ff9cda06486 (1).pdf
engineering material atomic structure of atom Lecture2.

engineering material atomic structure of atom Lecture2.
More from cartlidge
1 Cell Biology KnowIT.pdf

Here are the answers to the questions:
1. In a prokaryotic cell, the genetic material is a single loop of DNA not enclosed in a nucleus.
2. In a eukaryotic cell, the genetic material (DNA) is enclosed within a nucleus.
3. See table below
Prefix Multiple Standard form
centi (cm) 1 cm = 0.01 m x 10-2
milli (mm) 1 mm = 0.001 m x 10-3
micro (μm) 1 μm = 0.000 001 m x 10-6
nano (nm) 1 nm = 0.000 000 001 m x 10-9
4
1 Cell Biology.pptx

The document provides information on cell biology topics including cell structure, transport processes, and cell division. It contains:
1) Descriptions of plant and animal cell structures and comparisons between eukaryotic and prokaryotic cells.
2) Explanations of three transport processes - diffusion, osmosis, and active transport - and examples of each in cells and organisms.
3) An overview of cell division through mitosis and its role in growth and repair of cells.
6 Inh Variation and Evolution.pptx

Here are the key steps in cloning plants:
1) Plants can reproduce asexually through processes like producing runners or stolons, which will grow into clones that are genetically identical to the parent plant.
2) For example, a spider plant grows a rooting side branch called a stolon that will become a clone of the parent plant. A gardener may also take cuttings from a desirable plant to grow into clones.
3) The clones are genetically identical to the original parent plant and are called clones. The only variation between clones will come from environmental factors, not genetic differences. Asexual reproduction produces clones while sexual reproduction produces offspring with genetic variation.
5 Homeostasis resp KnowIT nervous-system.pptx

Here are the answers to the questions about the structure and function of the human nervous system:
1. What is a stimulus? Any change in the surroundings.
2. What is a receptor? Cells that detect a change.
3. Name the two parts of the central nervous system. Brain and spinal cord.
4. What is an effector? A muscle or gland.
5. What does the CNS coordinate? The response of effectors.
6. Put these in the correct order: receptor, stimulus, response, coordinator, effector. Stimulus → receptor → coordinator → effector → response.
7. What is the role of the sensory neurone? To carry impulses from
2 Organisation KnowIT.pdf

The document discusses the human digestive system as an organ system where organs like the mouth, esophagus, stomach, and intestines work together to digest and absorb food using enzymes. It explains that digestion breaks down large insoluble molecules into smaller soluble ones that can be absorbed, and this process relies on enzymes released by organs like the salivary glands, pancreas, and stomach. Key enzymes and their roles in breaking down carbohydrates, proteins, and lipids are identified.
3 Infection and Resp KnowIT.pdf

The document discusses different types of infectious diseases caused by pathogens such as viruses, bacteria, fungi and protists. It provides examples of viral diseases like measles and HIV, bacterial diseases including salmonella and gonorrhoea, and the fungal disease rose black spot and the protist disease malaria. The document explains how these diseases are transmitted and potential treatments or methods of control.
4 Bioenergetics KnowIT.pdf

Photosynthesis is the process by which plants produce glucose from carbon dioxide and water using energy from sunlight. The rate of photosynthesis is affected by several factors including carbon dioxide concentration, temperature, and light intensity, with each factor limiting the rate once a certain threshold is reached. Experiments can measure the rate of photosynthesis by counting oxygen bubbles produced over time as light intensity or other factors are varied.
6 inh variation evol knowit.pptx

Here are the key advantages summarized:
- Sexual reproduction leads to variation in offspring through meiosis and mixing of genes from two parents. This variation provides a survival advantage if the environment changes.
- Asexual reproduction requires only one parent so it is more efficient in terms of time, energy and resources as no mate needs to be found. However, offspring are identical clones with no genetic variation.
- Both types of reproduction have advantages depending on circumstances. Sexual reproduction promotes variation for changing environments while asexual is more efficient for stable conditions.
7 ecology knowit.pptx

The document discusses adaptations, interdependence and competition within ecological communities. It describes the different levels of ecological organization, including organisms, populations, communities, and ecosystems. Communities consist of many interacting populations that depend on each other through relationships like pollination, seed dispersal, and predator-prey interactions. Both biotic factors (living things) and abiotic factors (non-living things) influence communities. Organisms within communities compete for resources and adapt in ways that allow them to survive and reproduce under the prevailing environmental conditions. Competition and adaptations both help maintain a balanced community structure.
6.6 Hormones Homeo and Repro (Chris Paine)

Thyroxin is a hormone produced by the thyroid gland that controls metabolism and affects growth and physiological processes. It regulates the metabolic rate and helps control body temperature. Most hormones affect multiple target tissues and have more than one effect. The document discusses thyroid hormones and their roles, as well as hormones involved in the menstrual cycle and their feedback mechanisms.
6.5 neurons and synapses (chris paine)

The document discusses neurons and synapses in the human brain. It begins by showing an image of a small segment of the human brain, with lines representing neurons and dots representing synapses. It notes that synapses are crucial for neural communication and thought. While the number of brain cells does not increase much after birth, the connections between neurons through synapses continue developing. The document then provides explanations of key concepts regarding neurons, synapses, and neural signaling. It explains how neurons transmit electrical signals, how synapses allow signals to be transmitted between neurons, and how signals propagate along axons through action potentials.
6.4 Gas Exchange (Chris Paine)

The document discusses gas exchange in the lungs. It explains that two processes maintain concentration gradients of oxygen and carbon dioxide between the alveoli and blood: 1) circulation brings deoxygenated blood to the alveoli, and 2) ventilation increases and decreases lung volume through muscle contractions, ensuring a supply of oxygenated air reaches the alveoli. The diaphragm and intercostal muscles contract during inhalation to inflate the lungs, allowing for gas exchange by diffusion across the alveoli.
6.3 defence (chris paine)

The human body has structures and processes that resist the continuous threat of invasion by pathogens. These include leukocytes that ingest and destroy pathogens, as well as physical barriers like the skin and mucous membranes. The body also has mechanisms for blood clotting to prevent the entry of pathogens through cuts in the skin. Lymphocytes produce antibodies that provide specific immunity against particular pathogens. While antibiotics can treat bacterial infections by blocking bacterial processes, they are ineffective against viruses which lack metabolism and cannot be treated in the same way.
6.2 Blood System (Chris Paine)

The blood system continuously transports substances to cells and collects waste through three main types of blood vessels. Arteries carry blood away from the heart to tissues and have muscle and elastic fibers to maintain blood pressure. Capillaries have thin walls that penetrate tissues and allow for exchange. Veins collect blood from tissues and return it to the heart, using valves to prevent backflow. William Harvey discovered the circulation of blood in the early 1600s, overturning earlier theories, and the heart acts as a pump initiated by the sinoatrial node to circulate blood continuously through this system.
6.1 Digestion and Absorption (Chris Paine)

The small intestine has a structure that allows for both digestion and absorption of food. It has a folded inner wall and muscular outer layers that help move food along. The inner wall contains villi and microvilli that greatly increase the surface area for digestion and absorption. Enzymes in the small intestine digest macromolecules in food into smaller monomers. Absorption then occurs as these smaller molecules like glucose, amino acids, and fatty acids pass through the epithelial cells lining the villi into the bloodstream using various membrane transport processes. The liver plays an important role in processing absorbed glucose from the small intestine.
C ecology & conservation syllabus statements

The document discusses various topics relating to ecology and conservation, including species and communities, ecosystems, impacts of humans, conservation of biodiversity, population ecology, and nitrogen and phosphorus cycles. It provides essential ideas, understandings, applications, and skills for each topic. For example, for species and communities the essential idea is that community structure emerges from ecosystem properties, and understandings include how limiting factors affect species distribution and the unique role each species plays.
Academic honesty in ib

The document discusses academic honesty and misconduct in the International Baccalaureate (IB) program. It defines key terms like plagiarism, collusion, and exam misconduct. It notes that over half of investigated cases involve plagiarism while a quarter involve collusion. The document warns students about the consequences of getting caught cheating, like being banned from future exams or failing their diploma. It emphasizes that students are responsible for knowing what constitutes academic dishonesty and how to properly cite sources to avoid plagiarism.
Cas an introduction

This document provides information about CAS (Creativity, Activity, Service) which is an important component of the International Baccalaureate Diploma Programme. It explains that CAS involves learning through experience, personal development, and making a difference to others. Students must be involved in a variety of CAS experiences over 18 months including activities in creativity, activity, and service, as well as a CAS project and ongoing reflection. The document outlines examples of acceptable CAS experiences and provides guidance on documenting CAS in a portfolio using the learning outcomes framework.
11.2 muscle contraction

There are three main types of muscle tissue - skeletal, smooth, and cardiac. Muscle fibers are multinucleated cells formed from the fusion of individual embryonic muscle cells. Within each fiber are many parallel myofibrils composed of repeating sarcomere units. A sarcomere contains overlapping actin and myosin filaments. When an action potential reaches the neuromuscular junction, acetylcholine is released causing calcium ions to enter the fiber and allow the myosin heads to bind to actin, pulling the filaments together and contracting the muscle fiber. ATP provides energy to break the binding and reset the muscle for the next contraction.
11.2 muscle contraction

There are three main types of muscle tissue - skeletal, smooth, and cardiac. Muscle fibers are multinucleate cells formed from the fusion of individual embryonic muscle cells. Within each fiber are many parallel myofibrils composed of repeating sarcomere units. A sarcomere contains overlapping actin and myosin filaments. When an action potential reaches the neuromuscular junction, acetylcholine is released causing calcium ions to enter the fiber and allow the myosin heads to bind to actin, pulling the filaments together and contracting the muscle fiber. ATP provides energy to break the binding and reset the filaments for the next contraction.
More from cartlidge (20)
Recently uploaded
How to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective UpskillingExcellence Foundation for South Sudan
Strategies for Effective Upskilling is a presentation by Chinwendu Peace in a Your Skill Boost Masterclass organisation by the Excellence Foundation for South Sudan on 08th and 09th June 2024 from 1 PM to 3 PM on each day.RHEOLOGY Physical pharmaceutics-II notes for B.pharm 4th sem students

Physical pharmaceutics notes for B.pharm students
Temple of Asclepius in Thrace. Excavation results

The temple and the sanctuary around were dedicated to Asklepios Zmidrenus. This name has been known since 1875 when an inscription dedicated to him was discovered in Rome. The inscription is dated in 227 AD and was left by soldiers originating from the city of Philippopolis (modern Plovdiv).
Walmart Business+ and Spark Good for Nonprofits.pdf

"Learn about all the ways Walmart supports nonprofit organizations.
You will hear from Liz Willett, the Head of Nonprofits, and hear about what Walmart is doing to help nonprofits, including Walmart Business and Spark Good. Walmart Business+ is a new offer for nonprofits that offers discounts and also streamlines nonprofits order and expense tracking, saving time and money.
The webinar may also give some examples on how nonprofits can best leverage Walmart Business+.
The event will cover the following::
Walmart Business + (https://business.walmart.com/plus) is a new shopping experience for nonprofits, schools, and local business customers that connects an exclusive online shopping experience to stores. Benefits include free delivery and shipping, a 'Spend Analytics” feature, special discounts, deals and tax-exempt shopping.
Special TechSoup offer for a free 180 days membership, and up to $150 in discounts on eligible orders.
Spark Good (walmart.com/sparkgood) is a charitable platform that enables nonprofits to receive donations directly from customers and associates.
Answers about how you can do more with Walmart!"
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...Nguyen Thanh Tu Collection
https://app.box.com/s/y977uz6bpd3af4qsebv7r9b7s21935vdWound healing PPT

This document provides an overview of wound healing, its functions, stages, mechanisms, factors affecting it, and complications.
A wound is a break in the integrity of the skin or tissues, which may be associated with disruption of the structure and function.
Healing is the body’s response to injury in an attempt to restore normal structure and functions.
Healing can occur in two ways: Regeneration and Repair
There are 4 phases of wound healing: hemostasis, inflammation, proliferation, and remodeling. This document also describes the mechanism of wound healing. Factors that affect healing include infection, uncontrolled diabetes, poor nutrition, age, anemia, the presence of foreign bodies, etc.
Complications of wound healing like infection, hyperpigmentation of scar, contractures, and keloid formation.
Mule event processing models | MuleSoft Mysore Meetup #47

Mule event processing models | MuleSoft Mysore Meetup #47
Event Link:- https://meetups.mulesoft.com/events/details/mulesoft-mysore-presents-mule-event-processing-models/
Agenda
● What is event processing in MuleSoft?
● Types of event processing models in Mule 4
● Distinction between the reactive, parallel, blocking & non-blocking processing
For Upcoming Meetups Join Mysore Meetup Group - https://meetups.mulesoft.com/mysore/YouTube:- youtube.com/@mulesoftmysore
Mysore WhatsApp group:- https://chat.whatsapp.com/EhqtHtCC75vCAX7gaO842N
Speaker:-
Shivani Yasaswi - https://www.linkedin.com/in/shivaniyasaswi/
Organizers:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Giridhar Meka - https://www.linkedin.com/in/giridharmeka
Priya Shaw - https://www.linkedin.com/in/priya-shaw
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

Denis is a dynamic and results-driven Chief Information Officer (CIO) with a distinguished career spanning information systems analysis and technical project management. With a proven track record of spearheading the design and delivery of cutting-edge Information Management solutions, he has consistently elevated business operations, streamlined reporting functions, and maximized process efficiency.
Certified as an ISO/IEC 27001: Information Security Management Systems (ISMS) Lead Implementer, Data Protection Officer, and Cyber Risks Analyst, Denis brings a heightened focus on data security, privacy, and cyber resilience to every endeavor.
His expertise extends across a diverse spectrum of reporting, database, and web development applications, underpinned by an exceptional grasp of data storage and virtualization technologies. His proficiency in application testing, database administration, and data cleansing ensures seamless execution of complex projects.
What sets Denis apart is his comprehensive understanding of Business and Systems Analysis technologies, honed through involvement in all phases of the Software Development Lifecycle (SDLC). From meticulous requirements gathering to precise analysis, innovative design, rigorous development, thorough testing, and successful implementation, he has consistently delivered exceptional results.
Throughout his career, he has taken on multifaceted roles, from leading technical project management teams to owning solutions that drive operational excellence. His conscientious and proactive approach is unwavering, whether he is working independently or collaboratively within a team. His ability to connect with colleagues on a personal level underscores his commitment to fostering a harmonious and productive workplace environment.
Date: May 29, 2024
Tags: Information Security, ISO/IEC 27001, ISO/IEC 42001, Artificial Intelligence, GDPR
-------------------------------------------------------------------------------
Find out more about ISO training and certification services
Training: ISO/IEC 27001 Information Security Management System - EN | PECB
ISO/IEC 42001 Artificial Intelligence Management System - EN | PECB
General Data Protection Regulation (GDPR) - Training Courses - EN | PECB
Webinars: https://pecb.com/webinars
Article: https://pecb.com/article
-------------------------------------------------------------------------------
For more information about PECB:
Website: https://pecb.com/
LinkedIn: https://www.linkedin.com/company/pecb/
Facebook: https://www.facebook.com/PECBInternational/
Slideshare: http://www.slideshare.net/PECBCERTIFICATION
What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...

What is Digital Literacy? A guest blog from Andy McLaughlin, University of Aberdeen
ANATOMY AND BIOMECHANICS OF HIP JOINT.pdf

it describes the bony anatomy including the femoral head , acetabulum, labrum . also discusses the capsule , ligaments . muscle that act on the hip joint and the range of motion are outlined. factors affecting hip joint stability and weight transmission through the joint are summarized.
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...Nguyen Thanh Tu Collection
https://app.box.com/s/tacvl9ekroe9hqupdnjruiypvm9rdaneSolutons Maths Escape Room Spatial .pptx

Solutions of Puzzles of Mathematics Escape Room Game in Spatial.io
Recently uploaded (20)
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective Upskilling
RHEOLOGY Physical pharmaceutics-II notes for B.pharm 4th sem students

RHEOLOGY Physical pharmaceutics-II notes for B.pharm 4th sem students
IGCSE Biology Chapter 14- Reproduction in Plants.pdf

IGCSE Biology Chapter 14- Reproduction in Plants.pdf
Walmart Business+ and Spark Good for Nonprofits.pdf

Walmart Business+ and Spark Good for Nonprofits.pdf
BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...

BÀI TẬP BỔ TRỢ TIẾNG ANH 8 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2023-2024 (CÓ FI...
B. Ed Syllabus for babasaheb ambedkar education university.pdf

B. Ed Syllabus for babasaheb ambedkar education university.pdf
Mule event processing models | MuleSoft Mysore Meetup #47

Mule event processing models | MuleSoft Mysore Meetup #47
ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...

ISO/IEC 27001, ISO/IEC 42001, and GDPR: Best Practices for Implementation and...
What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...

What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...
Combined sci c5 syllabus
- 1. 12/04/16 C5 Electricity & ChemistryC5 Electricity & Chemistry Syllabus statementsSyllabus statements Statements from Cambridge IGCSE Combined Science syllabus 0653 (for exams in 2016 – 2018)
- 2. C5 Electricity & ChemistryC5 Electricity & Chemistry 1. State that electrolysis is the chemical effect of electricity on ionic compounds, causing them to break up into simpler substances, usually elements. 2. Use the terms electrode, electrolyte, anode and cathode. 3. Describe electrolysis in terms of the ions present and the reactions at the electrodes.
- 3. C5 Electricity & ChemistryC5 Electricity & Chemistry 4. Describe the electrode products, using inert electrodes, in the electrolysis of: •molten lead(II) bromide; •aqueous copper chloride. 5. Predict the products of the electrolysis of a specified binary compound in the molten state.