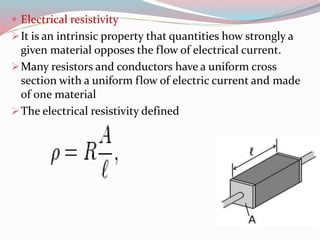
This document provides an overview of electrochemistry. It begins by defining electrochemistry as the study of chemical reactions at the interface of an electrode and electrolyte involving the interaction of electrical and chemical changes. The document then discusses the history and founders of electrochemistry, including Faraday's two laws of electrolysis. It explains key concepts such as oxidation-reduction reactions, balancing redox equations, and the Nernst equation. The document also covers applications including batteries, corrosion, electrolysis, and branches of electrochemistry like bioelectrochemistry and nanoelectrochemistry.