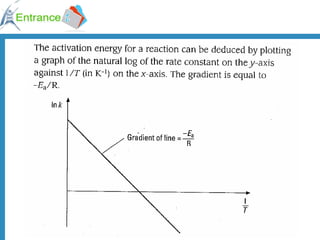
This document discusses chemical kinetics and factors that affect reaction rates:
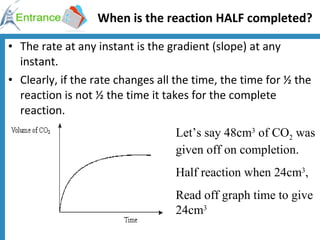
1) Reaction rates can be defined as the decrease in reactants or increase in products per unit time. Rates depend on concentrations and temperature.
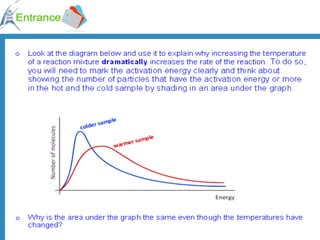
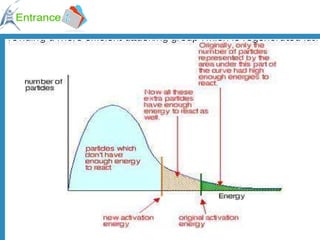
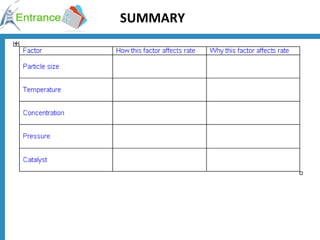
2) Factors that increase reaction rates include higher concentrations, increased surface area, and higher temperatures. Higher temperatures provide more collisions with energy above the activation energy.
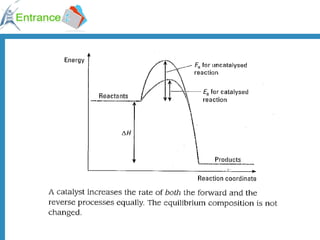
3) A catalyst provides an alternative reaction pathway with lower activation energy, increasing the number of effective collisions and thereby increasing the reaction rate.





















![Equation Example The reaction NO 2 ( g ) + CO ( g ) -> NO ( g ) + CO 2 ( g ) occurs in two steps: NO 2 + NO 2 -> NO + NO 3 (slow step) NO 3 + CO -> NO 2 + CO 2 (fast step) The slow step is the rate determining step and the rate equation should be Rate = k[NO 2 ] 2](https://image.slidesharecdn.com/chemicalkineticsok1294986988-110401065228-phpapp02/85/Chemical-kinetics-ok1294986988-30-320.jpg)

![Rate = k [ A ] Rate = k [(CH 3 ) 3 CCl](https://image.slidesharecdn.com/chemicalkineticsok1294986988-110401065228-phpapp02/85/Chemical-kinetics-ok1294986988-33-320.jpg)