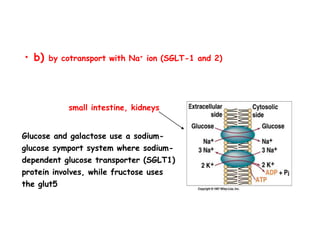
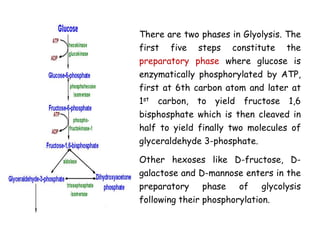
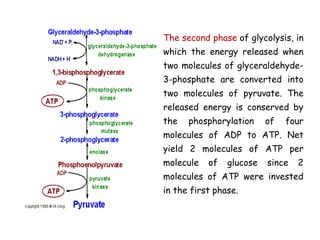
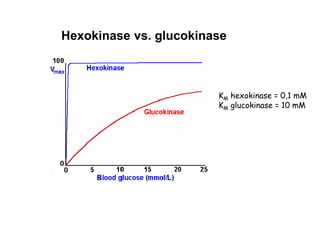
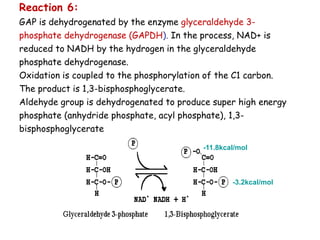
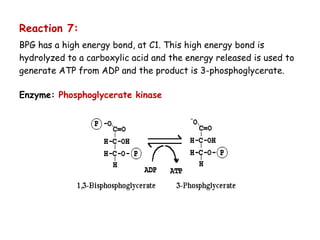
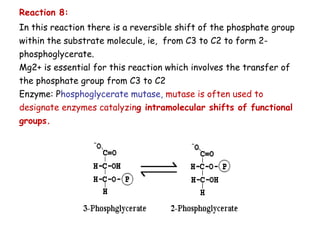
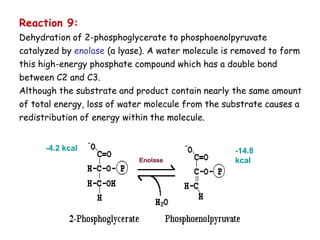
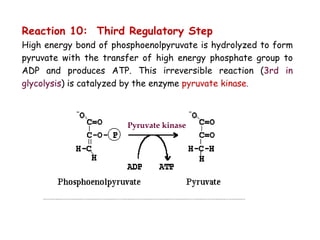
The document details the metabolic pathways of glucose, including its entry into cells via glucose transporters and the mechanisms of glycolysis, which breaks down glucose to produce energy. It outlines the regulatory steps in glycolysis, the enzymes involved, and the conversion of glucose derivatives like fructose and galactose into glycolytic intermediates. Additionally, it discusses conditions like galactosemia caused by genetic defects in galactose metabolism.