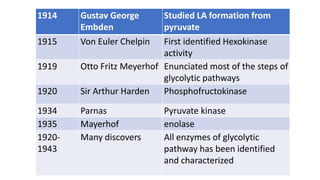
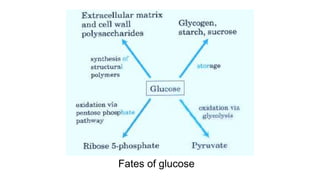
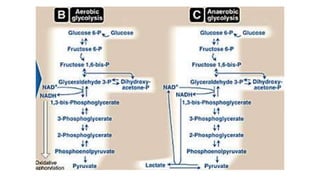
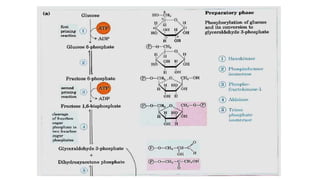
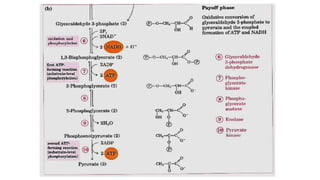
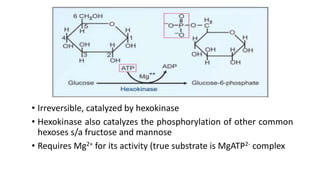
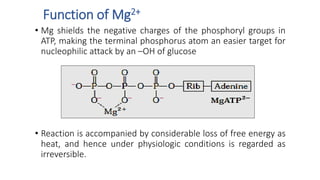
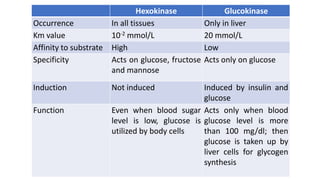
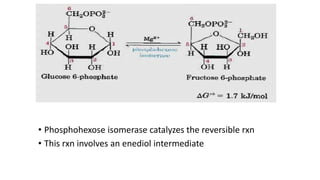
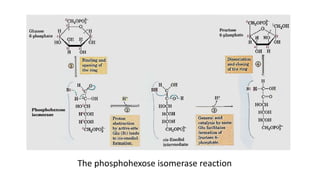
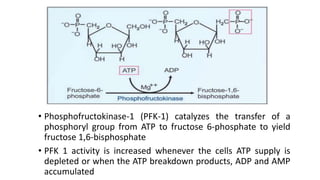
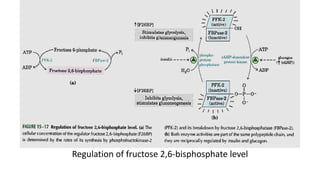
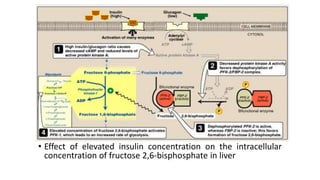
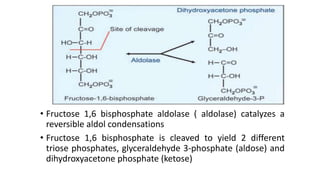
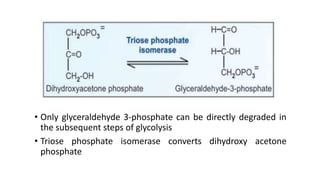
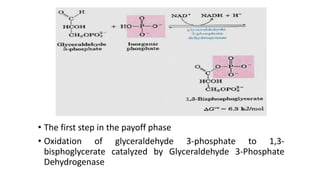
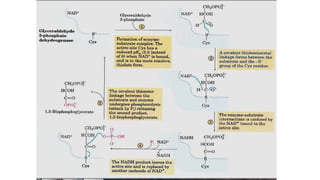
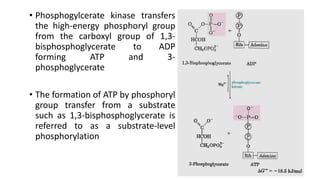
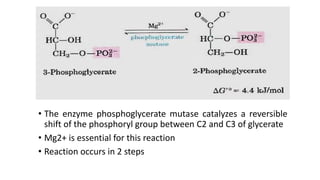
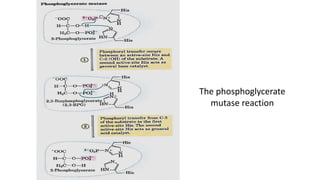
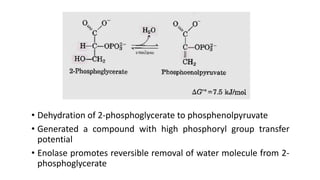
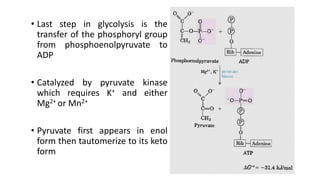
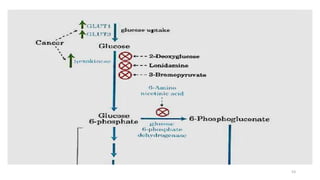
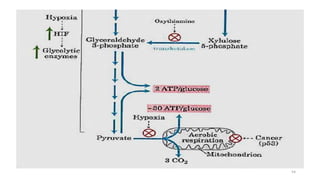
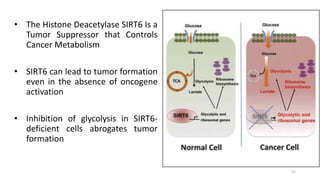
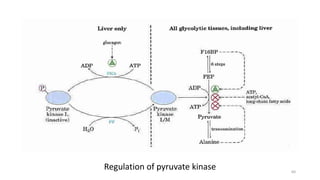
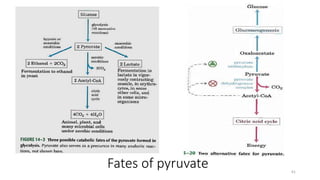
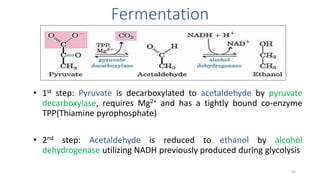
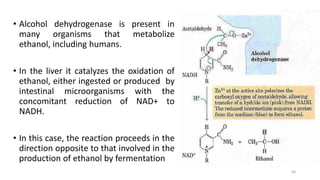
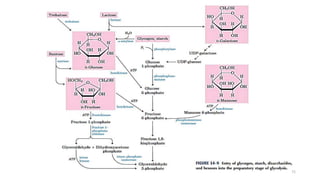
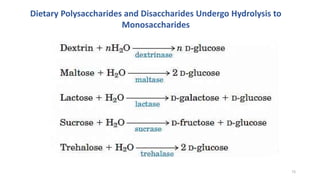
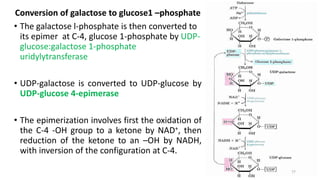
Glycolysis is the breakdown of glucose to pyruvate through a series of enzyme-catalyzed reactions. It occurs in the cytosol and consists of a preparatory phase requiring ATP and a payoff phase generating ATP. Key steps include phosphorylation by hexokinase, aldolase cleavage, substrate-level phosphorylation by phosphoglycerate kinase, and pyruvate formation by pyruvate kinase. Glycolytic enzymes are regulated by feedback inhibition and metabolites like fructose 2,6-bisphosphate and AMP/ATP ratios to control flux through the pathway.