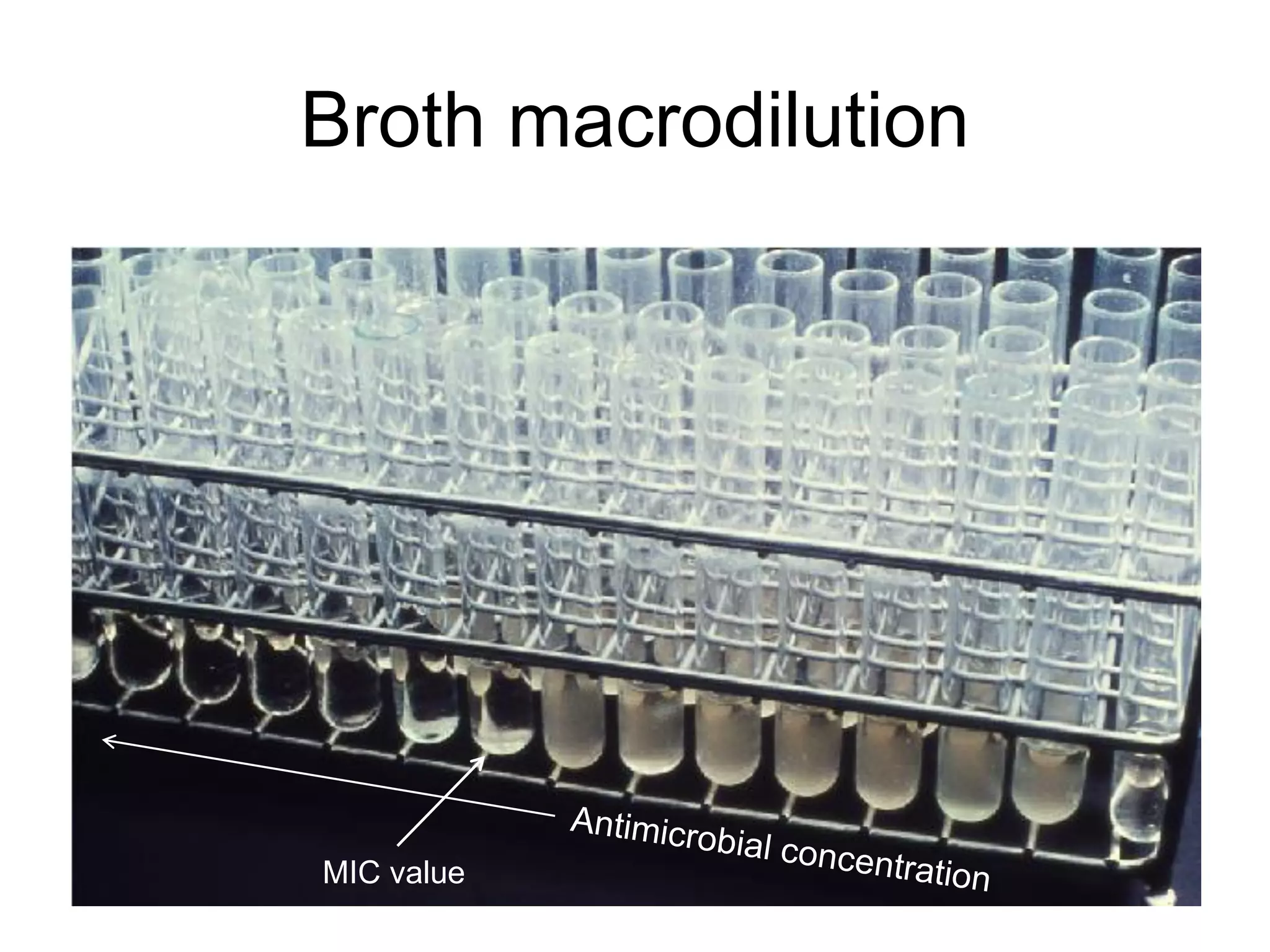
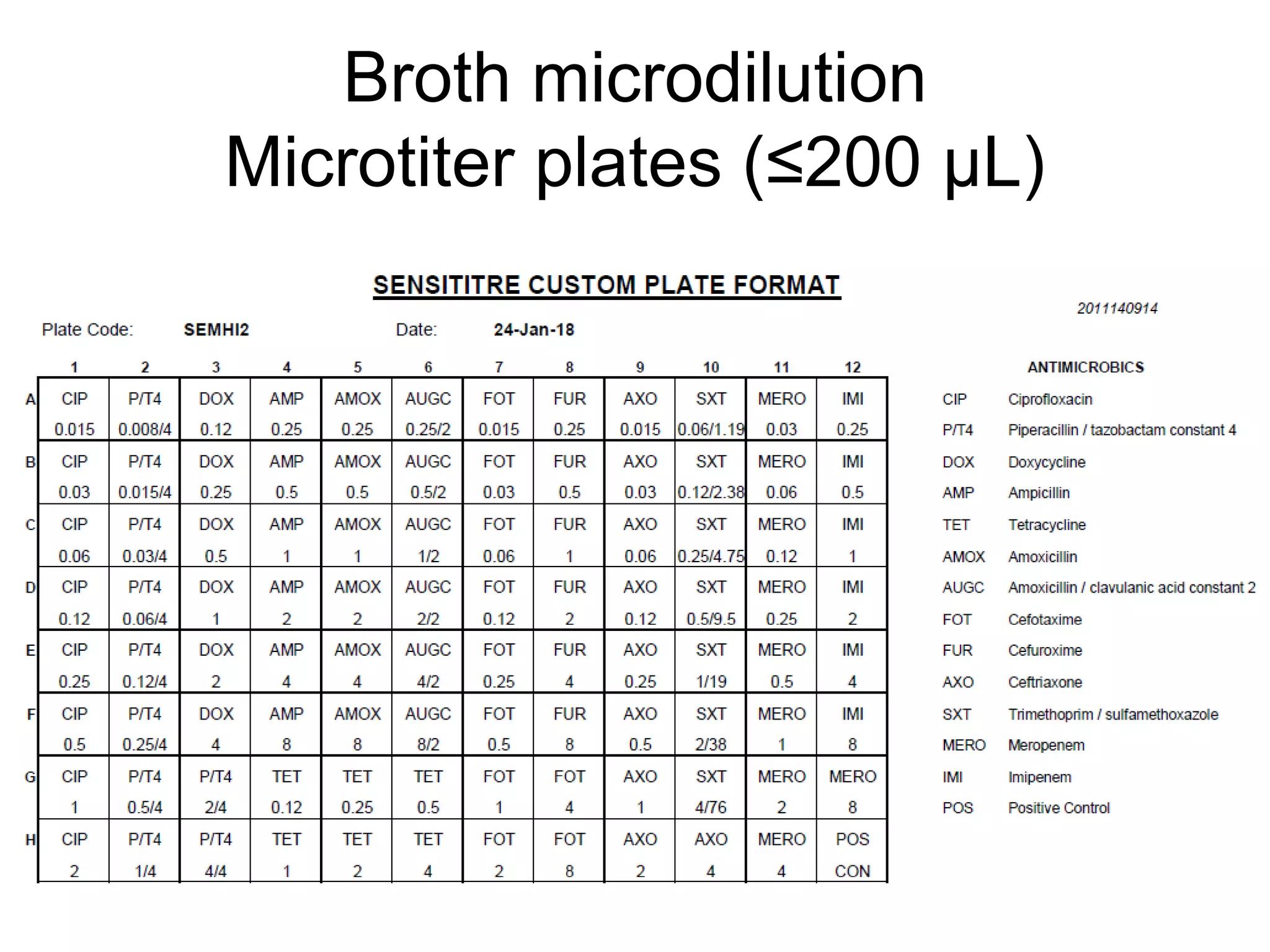
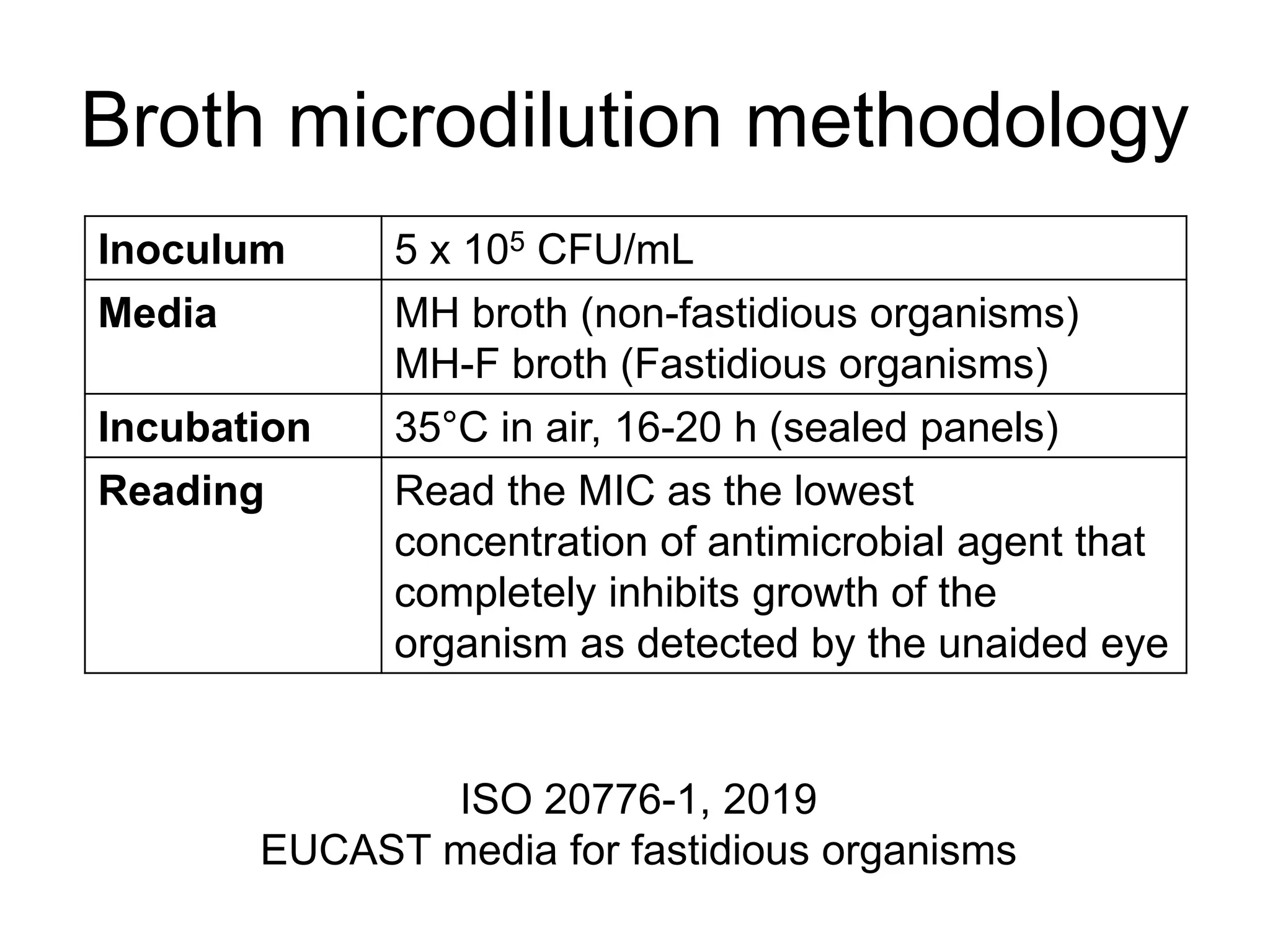
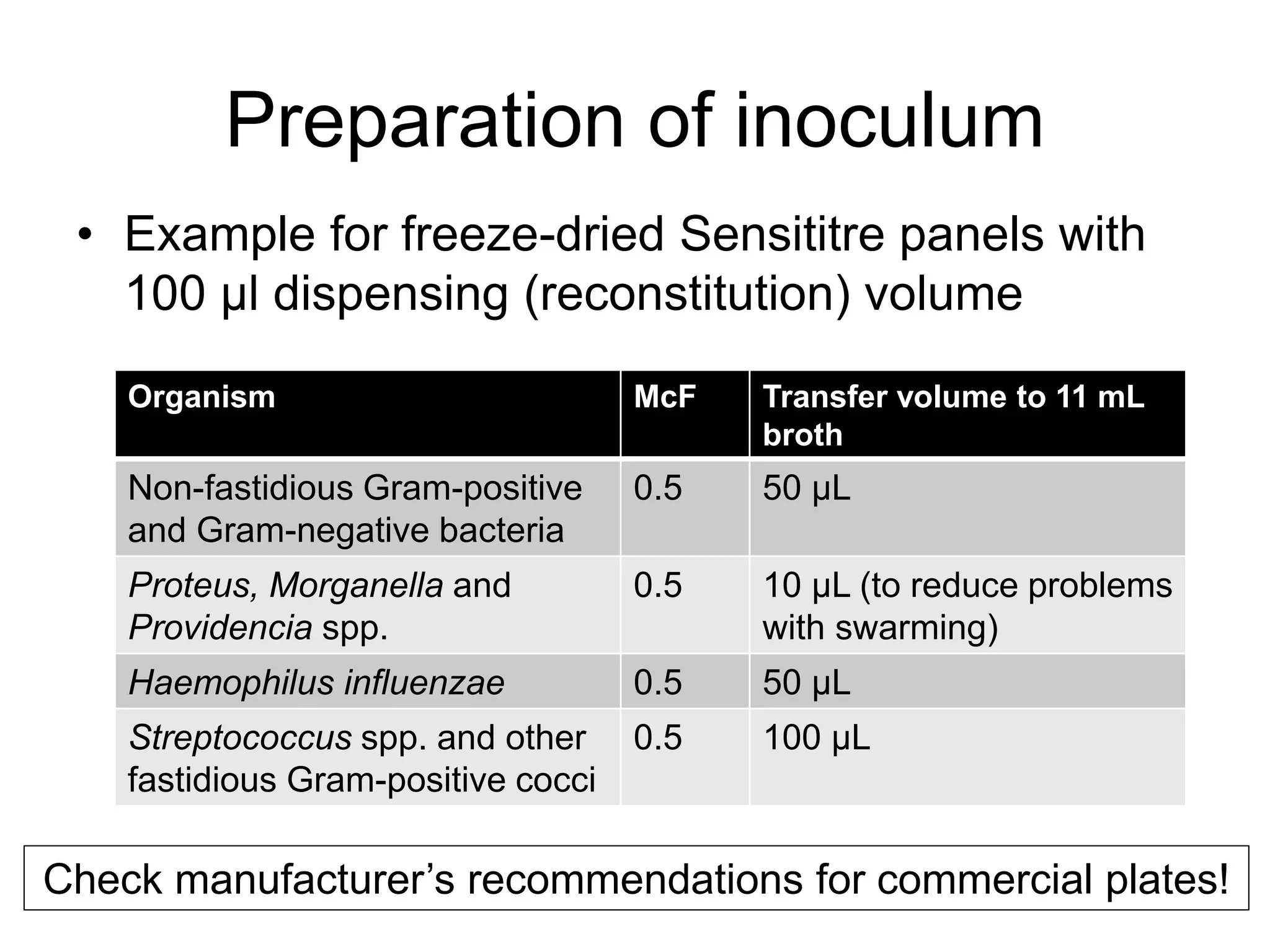
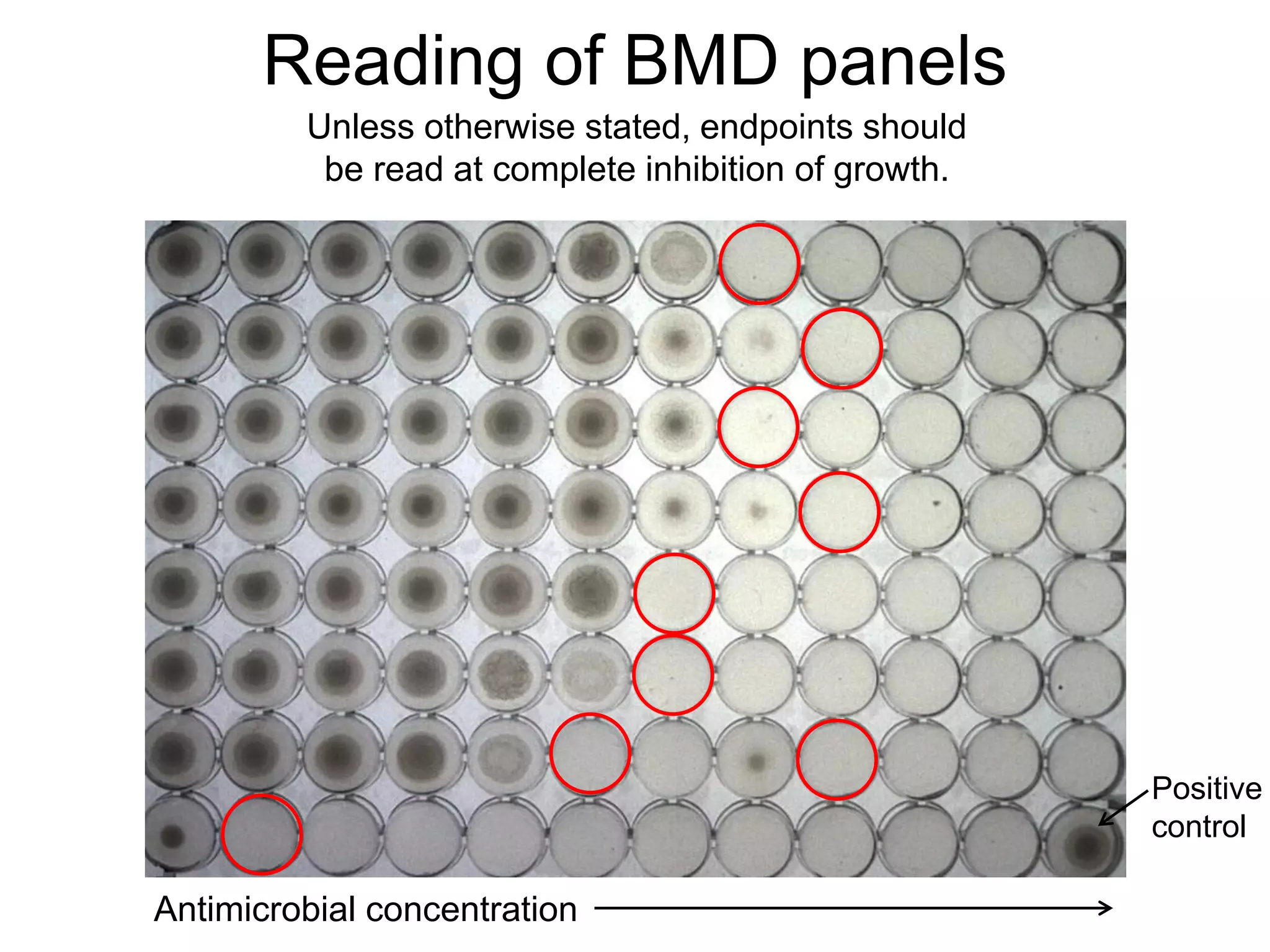
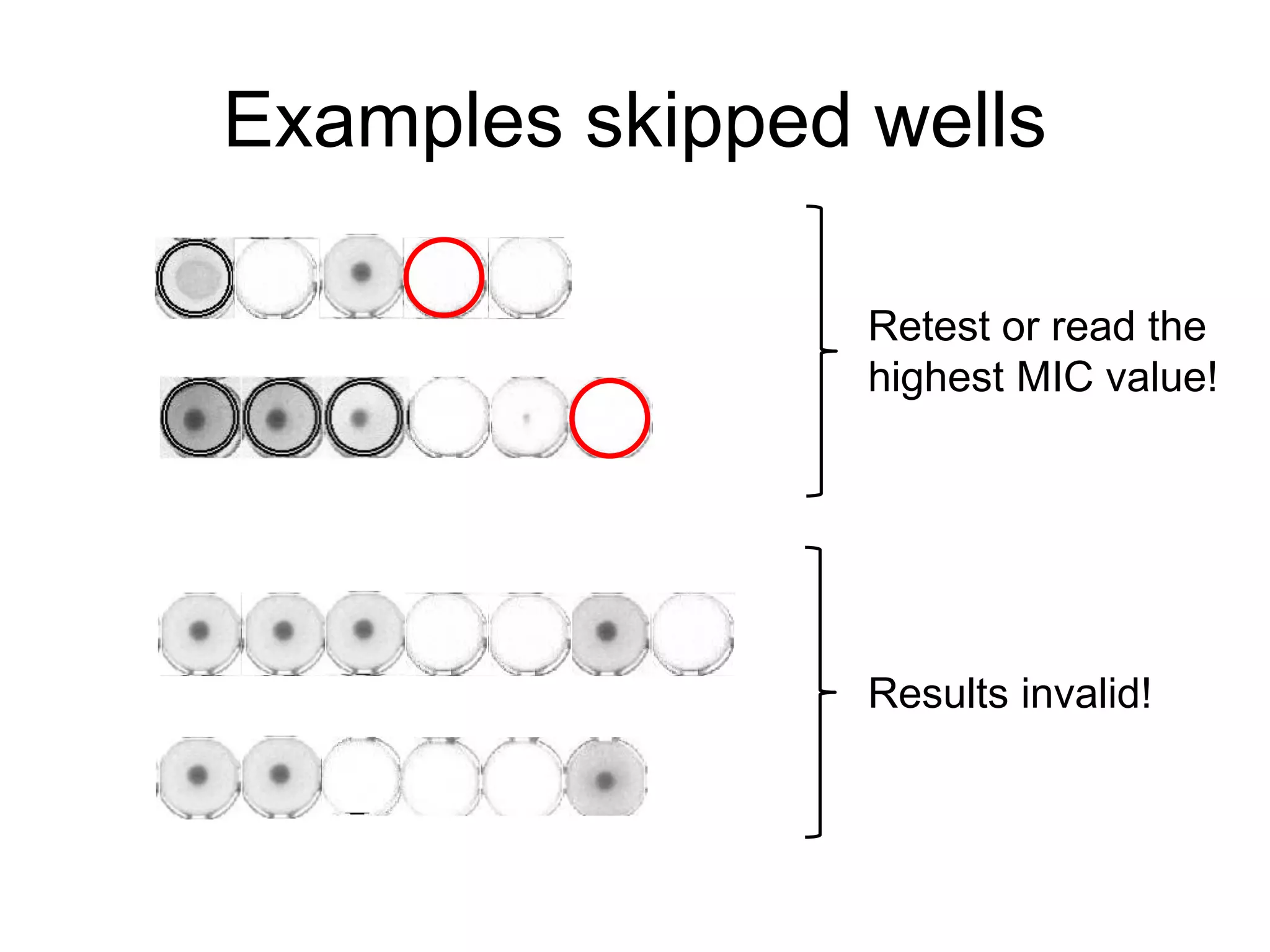
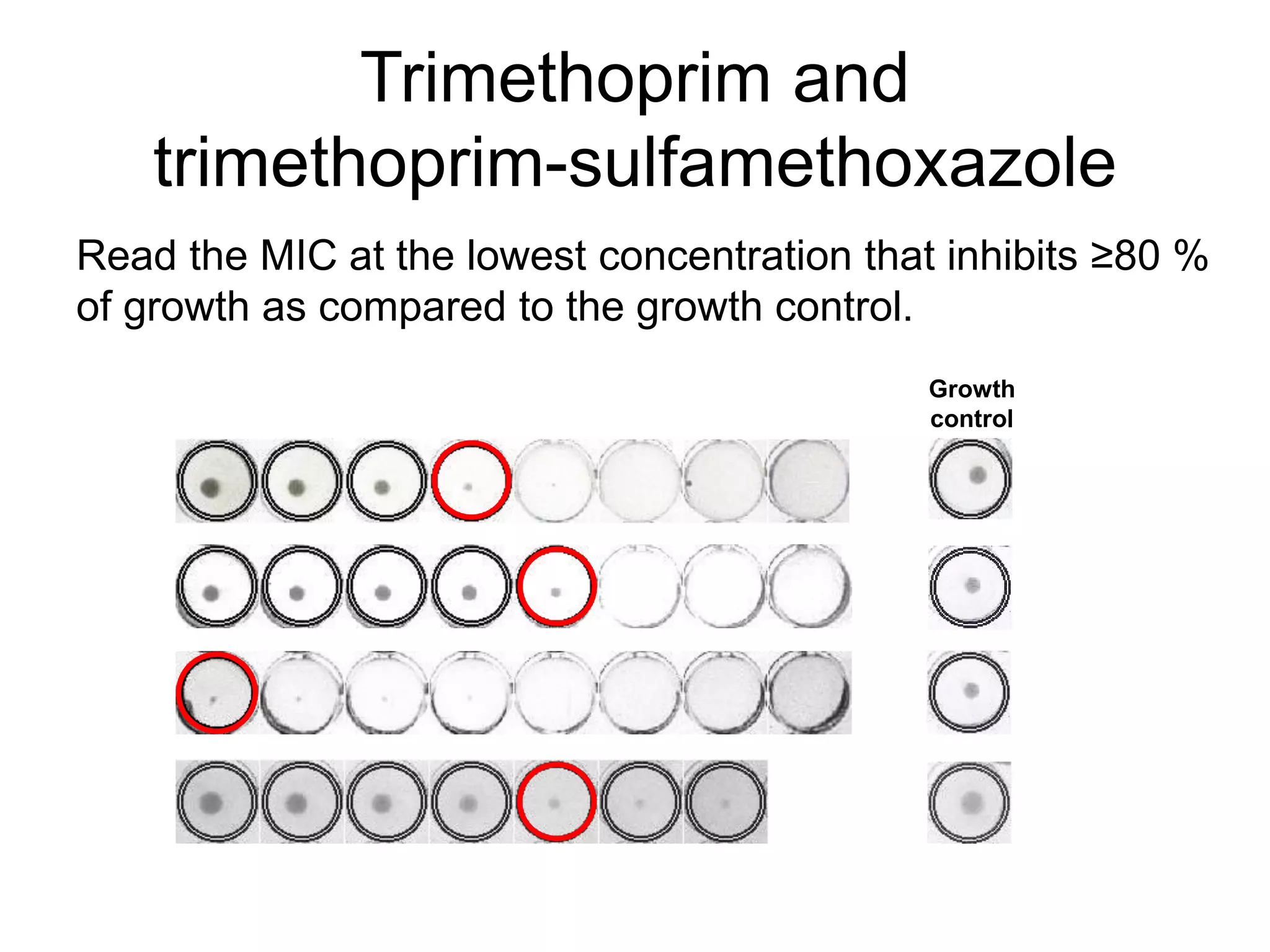
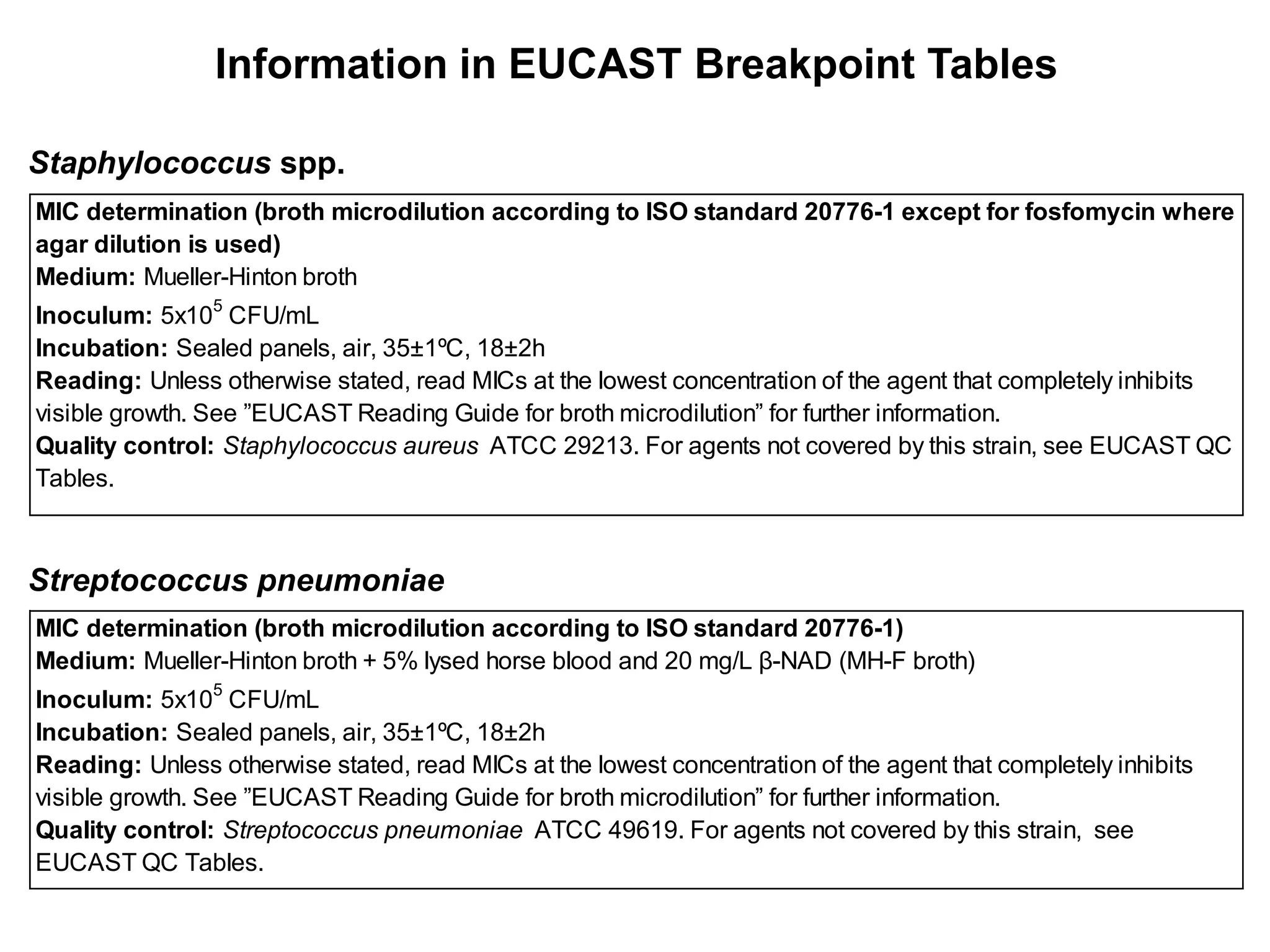
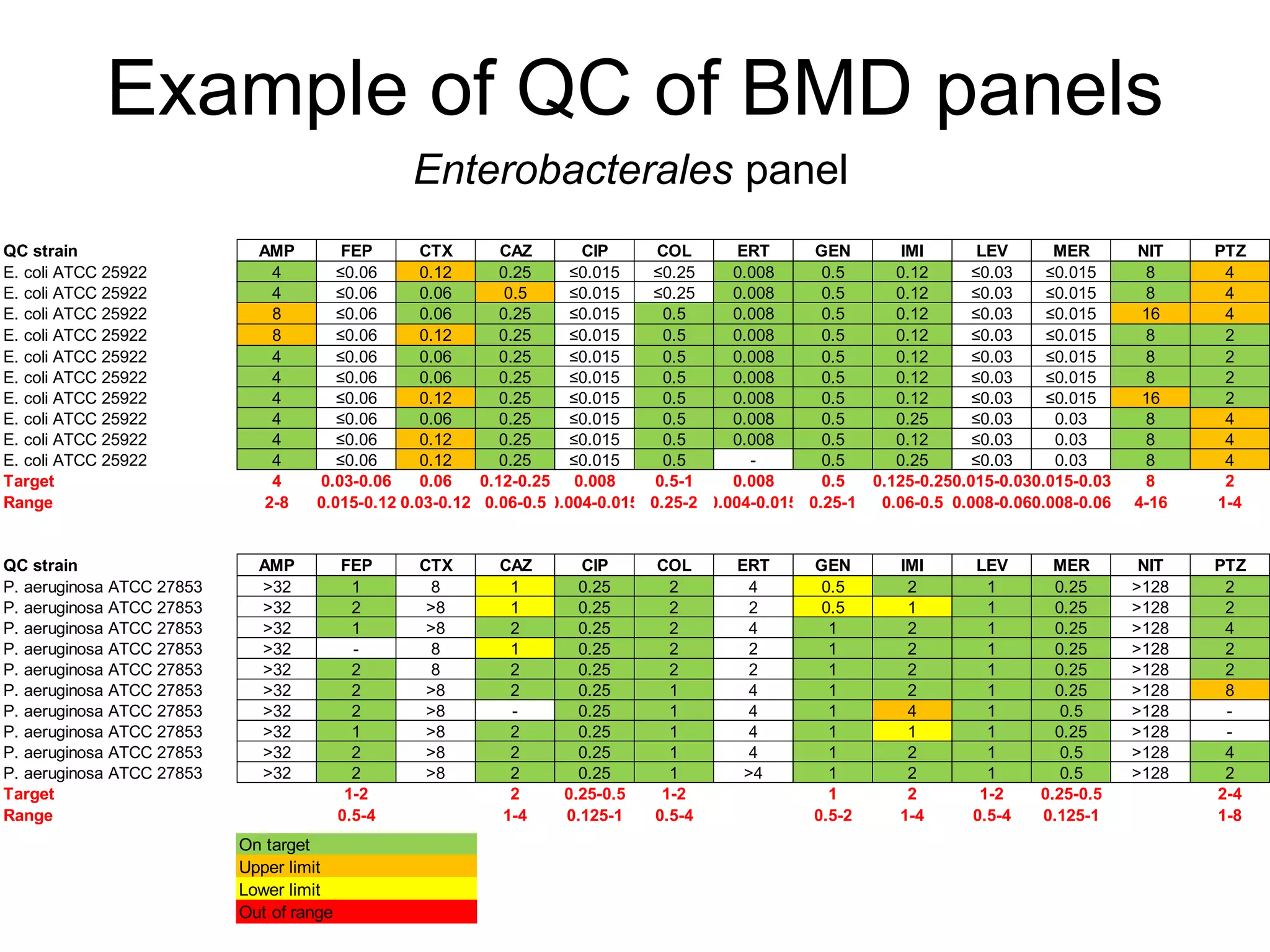
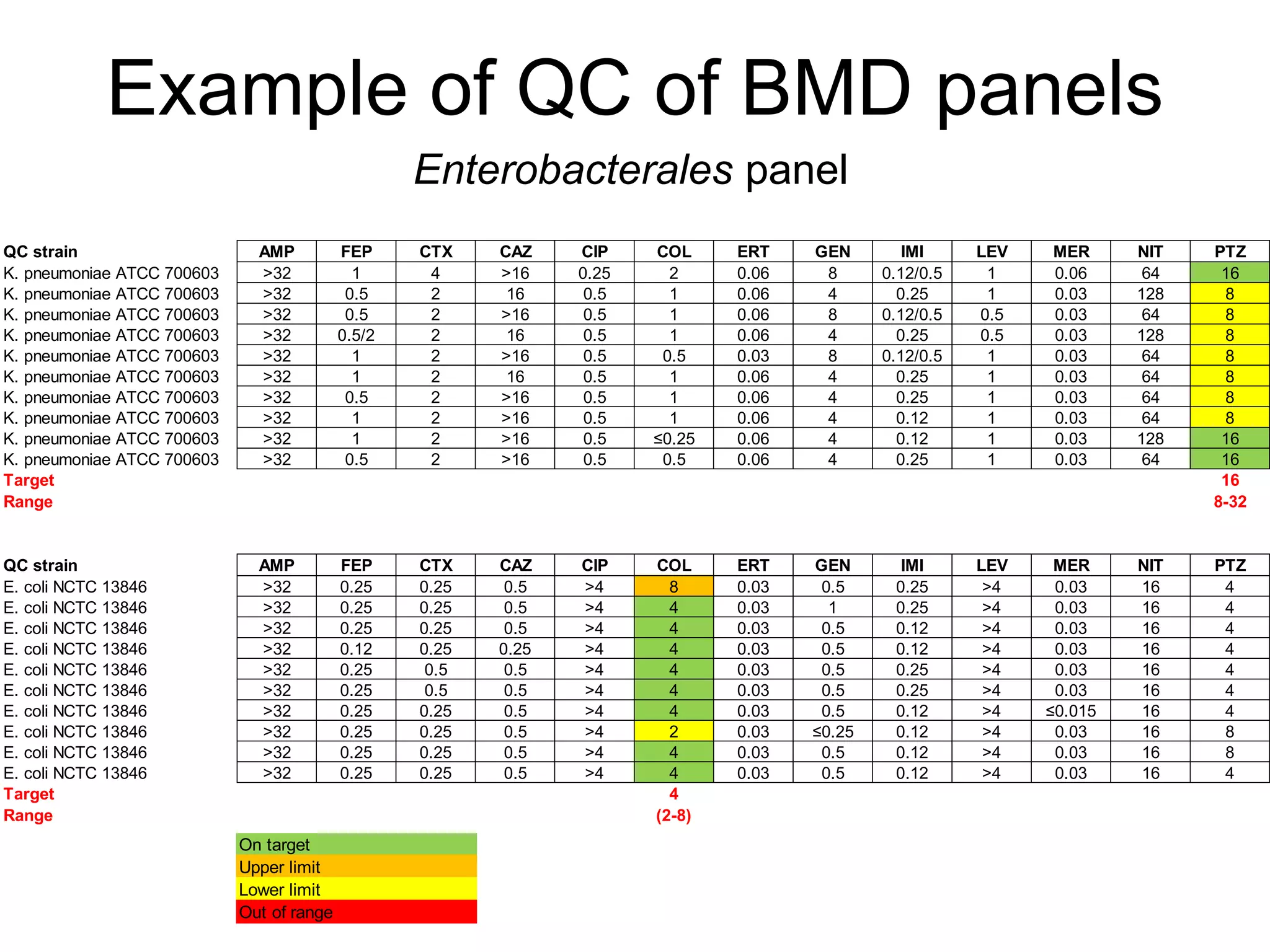
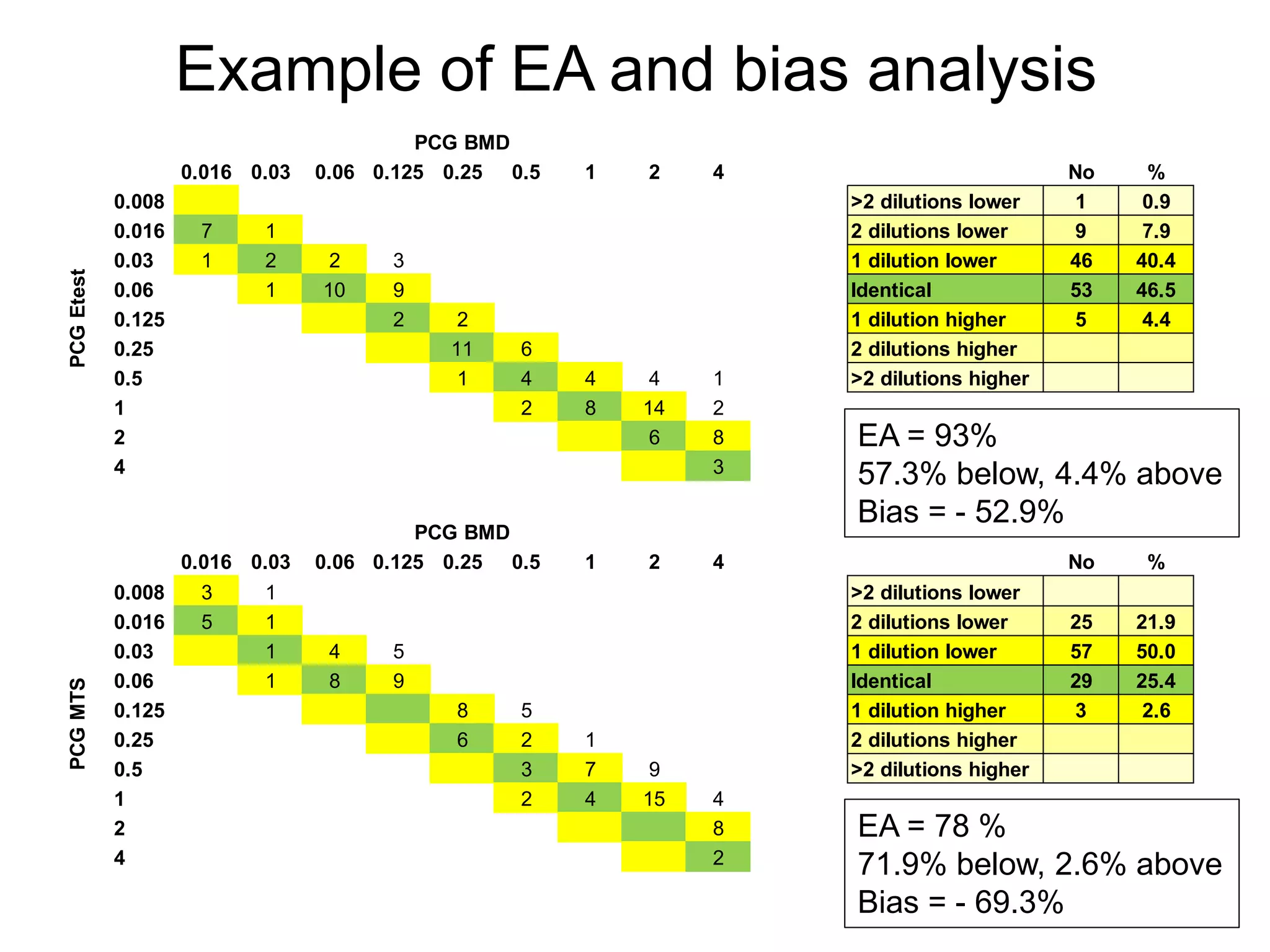
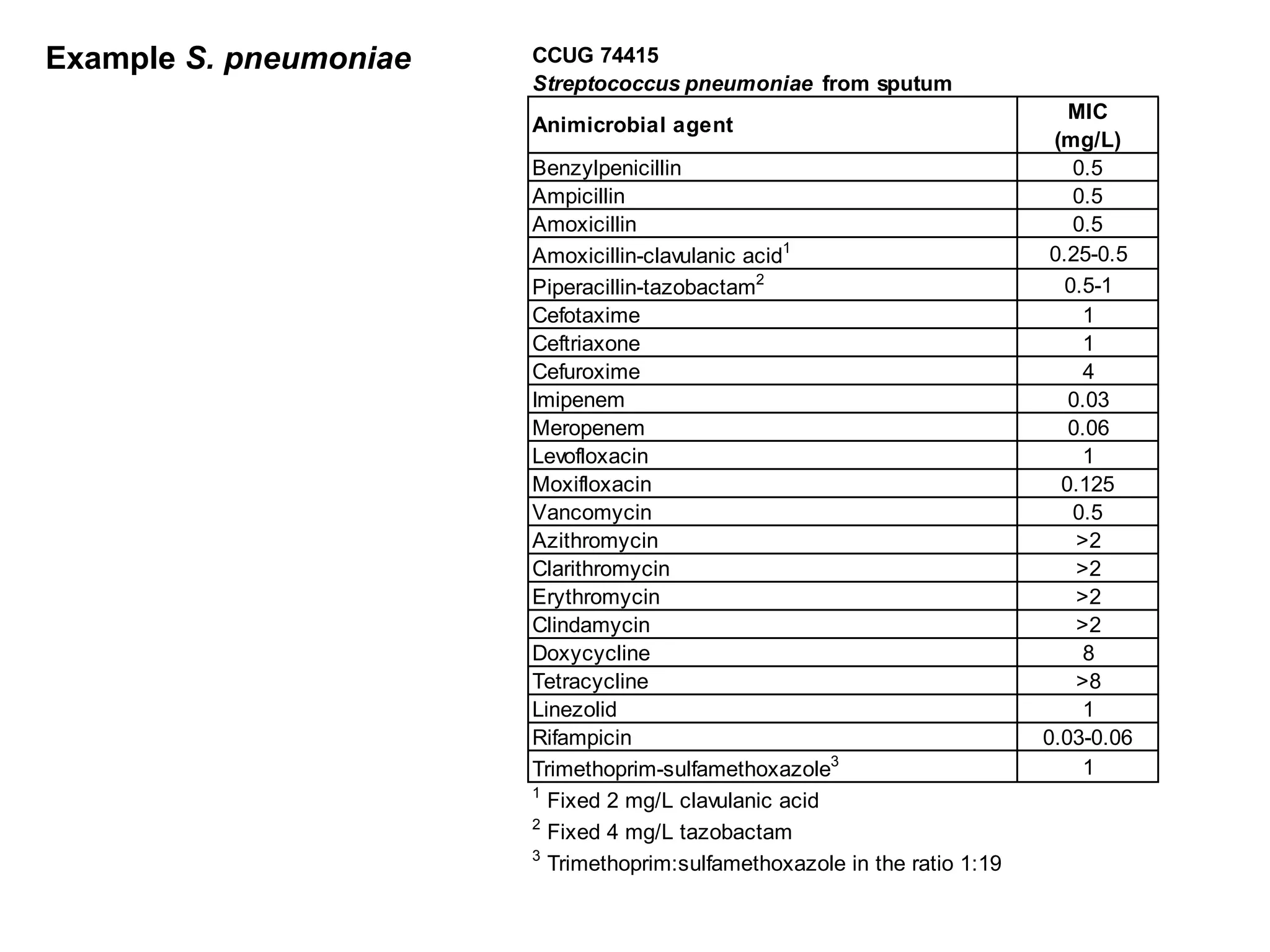
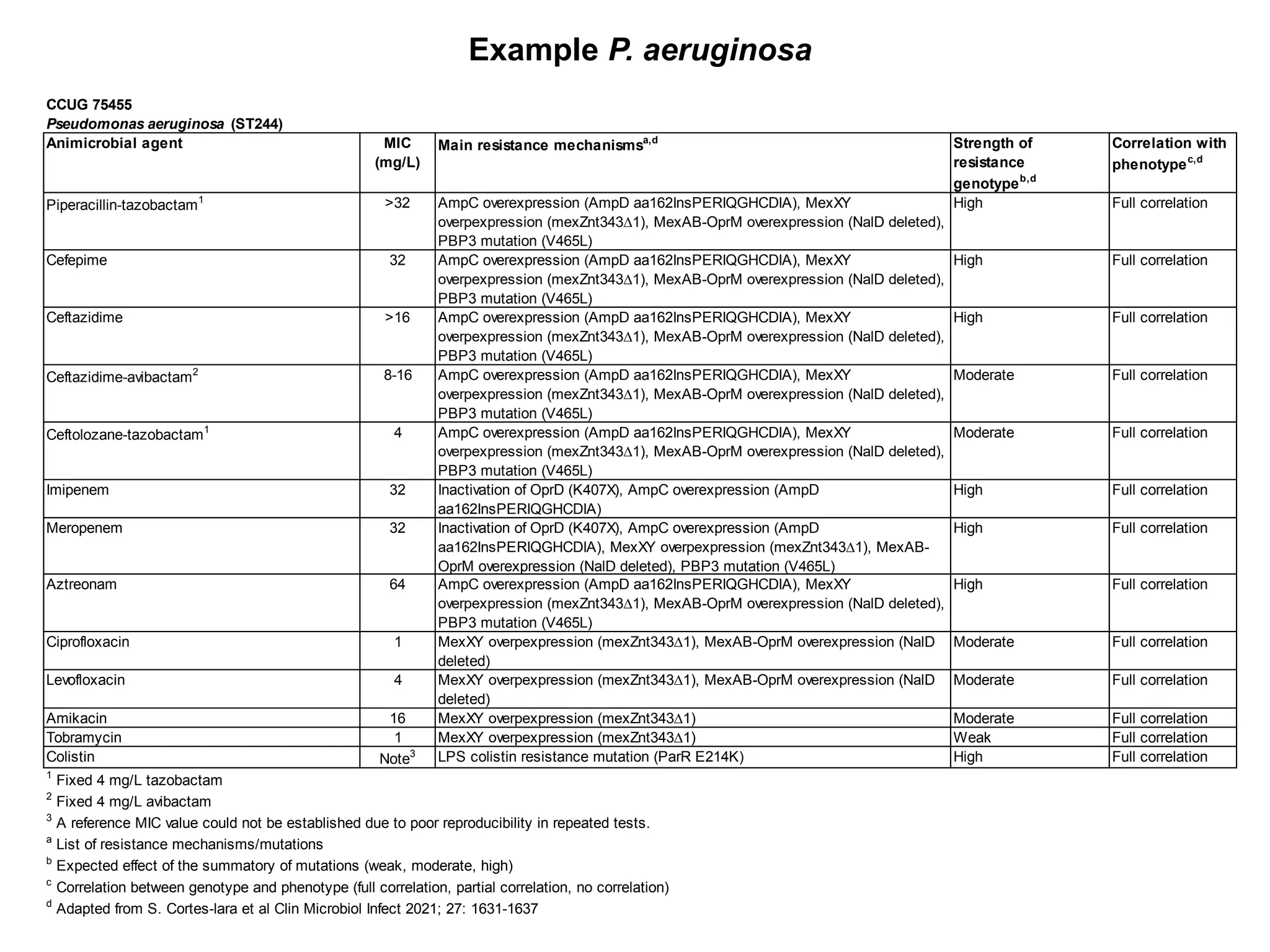
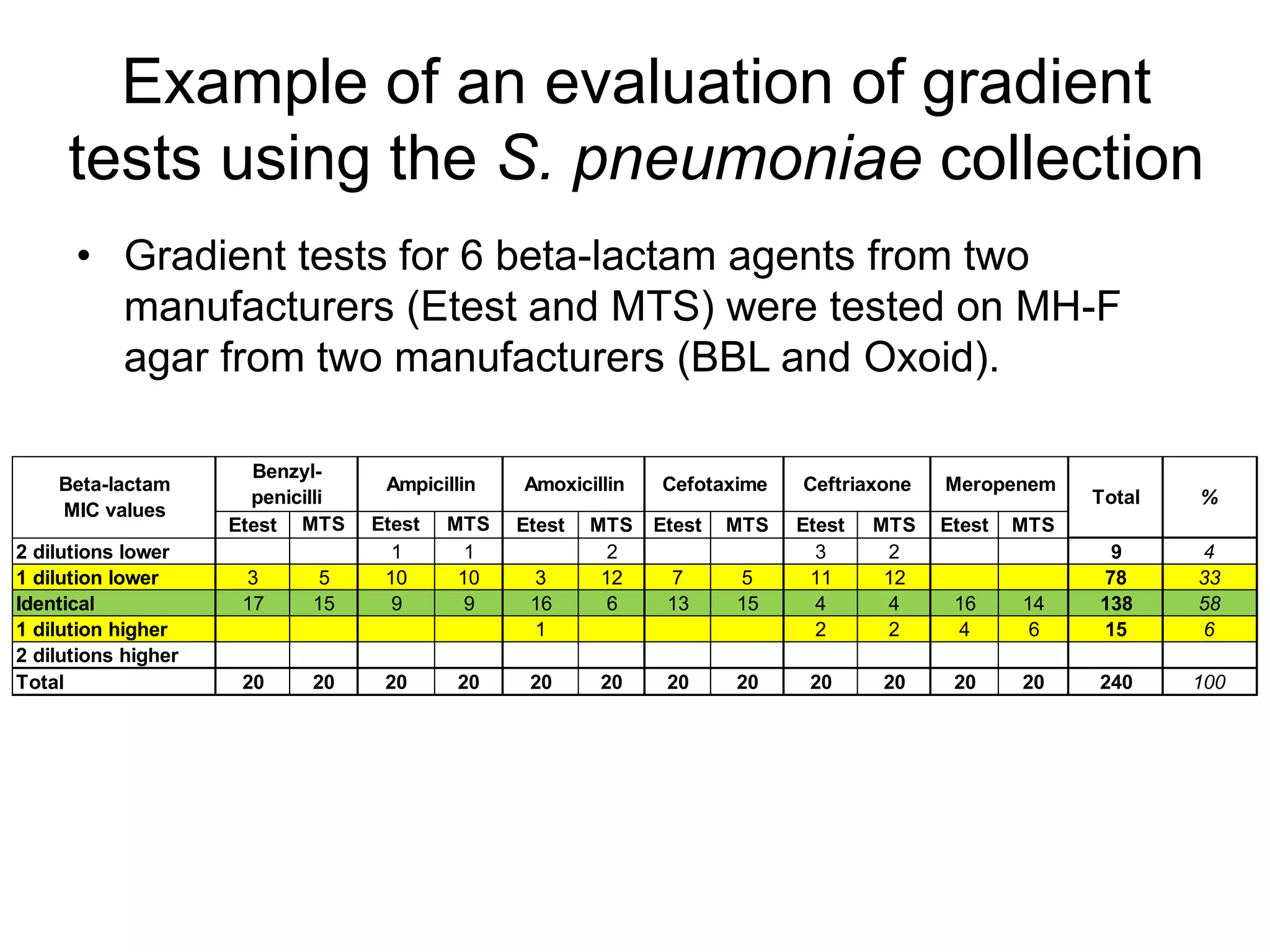
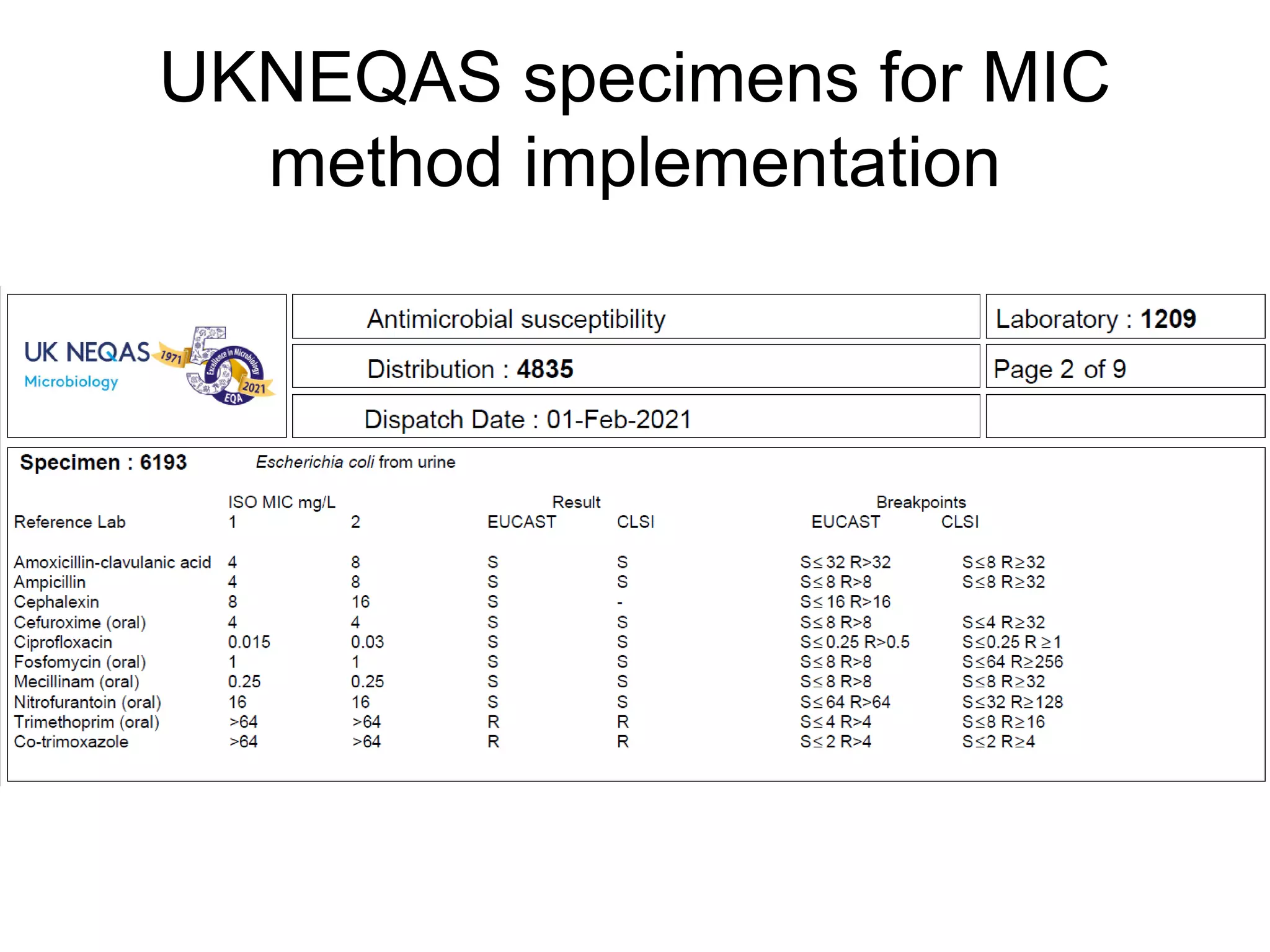
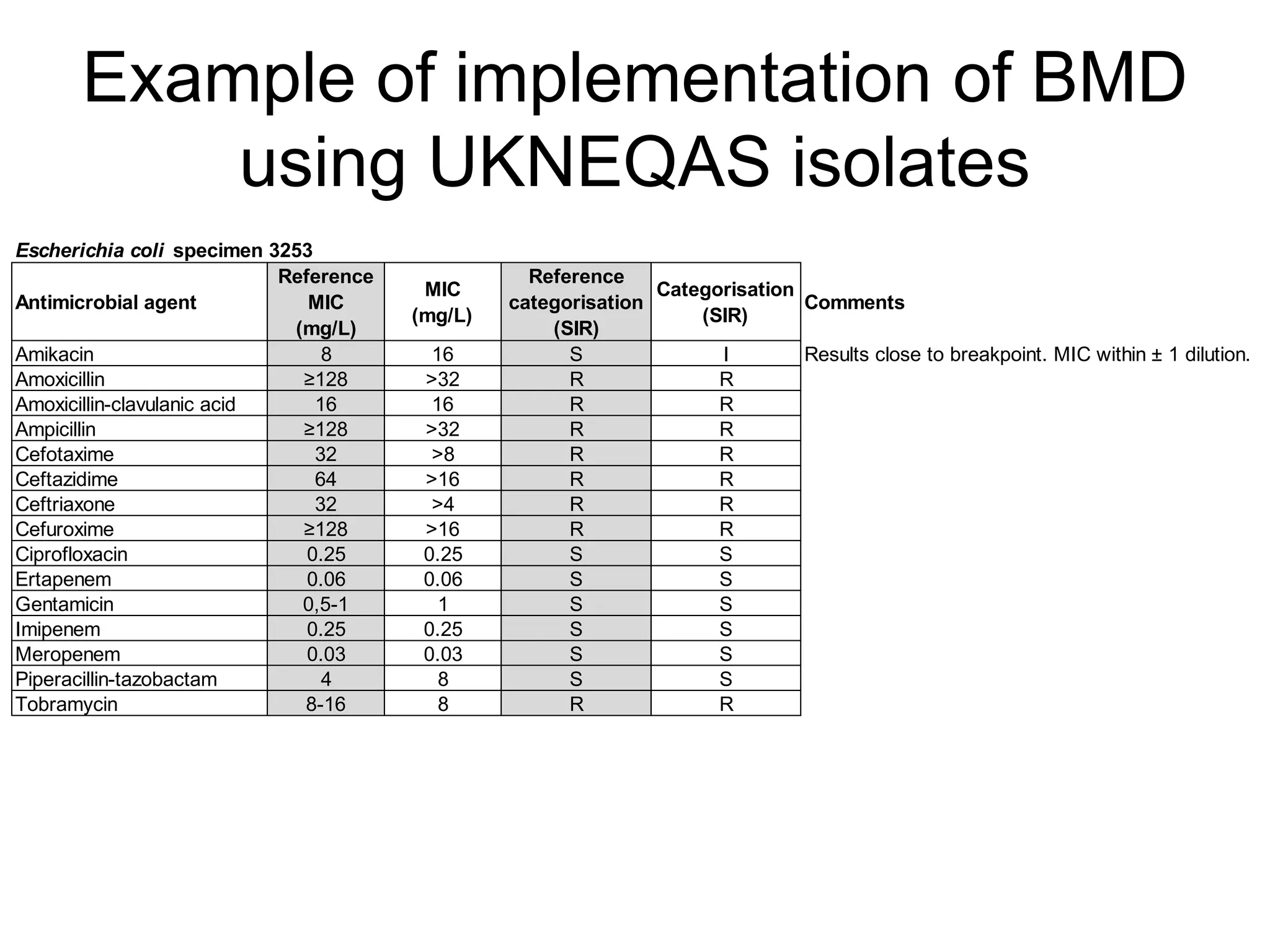
The document outlines the broth microdilution reference methodology for antimicrobial susceptibility testing (AST), emphasizing the importance of strict adherence to parameter standardization for reliable results. It details the procedure for determining the minimum inhibitory concentration (MIC) using specific media, inoculum, incubation conditions, and result interpretations based on EUCAST guidelines. Additionally, it discusses quality control requirements and specific reading instructions for various antimicrobial agents to ensure accurate testing of both non-fastidious and fastidious organisms.