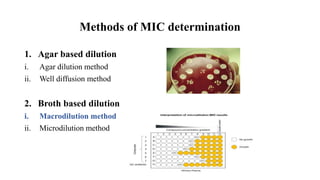
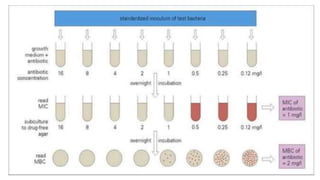
The document describes the macrodilution method for determining the minimal inhibitory concentration (MIC) of antibiotics. Key steps include:
1. Preparing stock solutions of antibiotics at high concentrations like 10 mg/mL and diluting them to make testing concentrations.
2. Creating a standardized bacterial inoculum of around 5x105 CFU/mL.
3. Diluting the antibiotic in a series of tubes containing broth and adding the bacterial inoculum.
4. Incubating the tubes overnight and finding the lowest antibiotic concentration tube that shows no visible growth, which is the MIC. The MBC can also be determined by culturing samples from clear tubes.