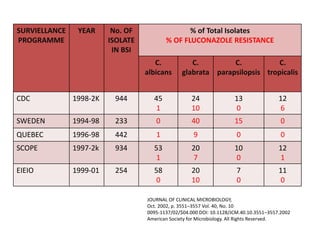
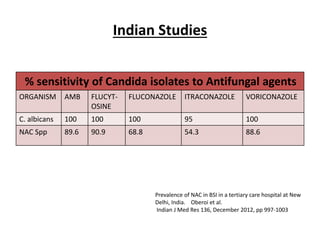
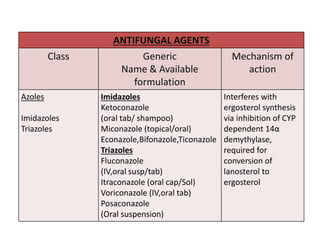
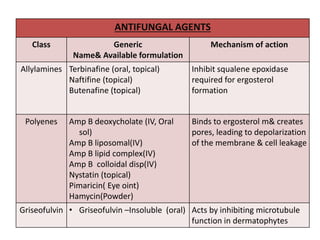
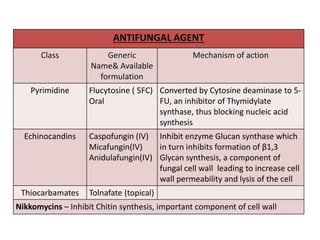
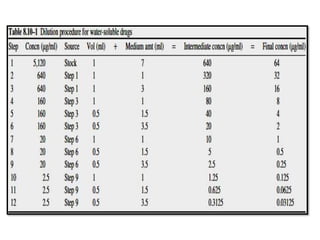
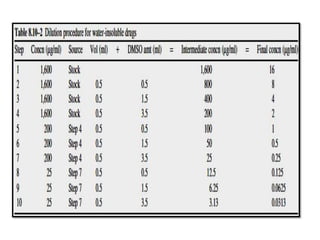
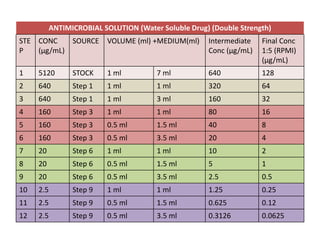
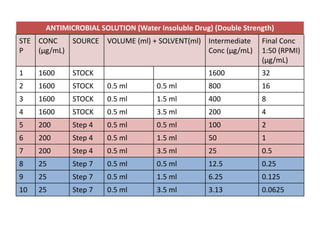
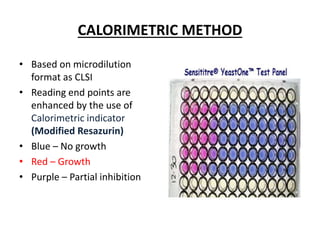
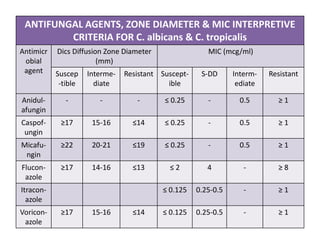
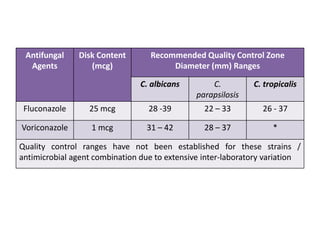
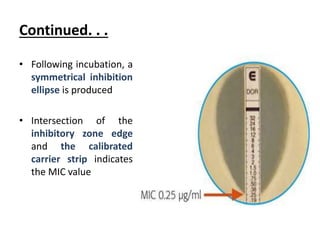
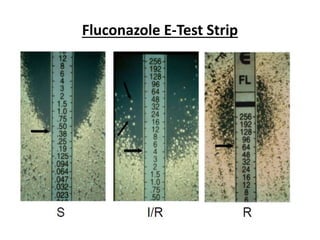
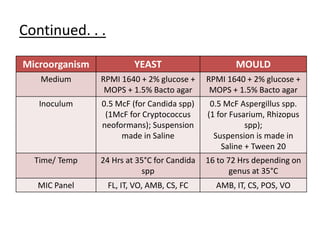
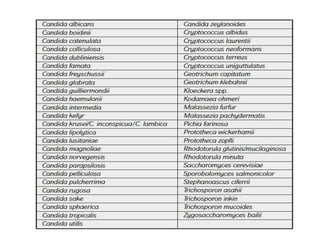
This document discusses antifungal susceptibility testing. It provides background on the history of antifungal susceptibility testing and why it is needed. It describes different methods for testing including broth dilution and disk diffusion. It discusses various antifungal agents and their mechanisms of action. The document outlines the procedures for broth microdilution and macrodilution testing according to CLSI guidelines, including preparation of inoculum, drug solutions, reading results, and testing of filamentous fungi.