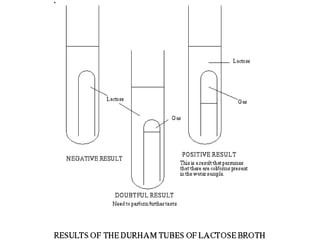
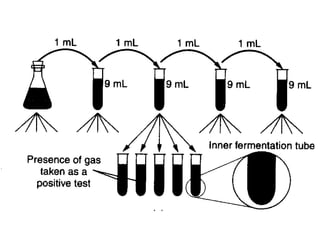
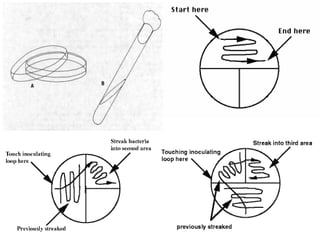
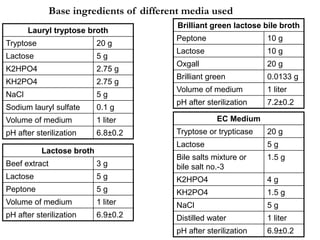
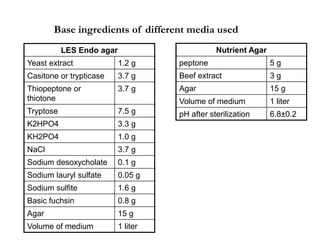
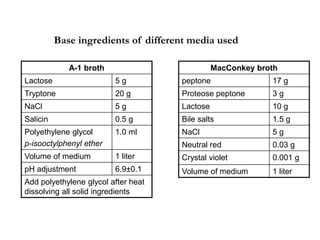
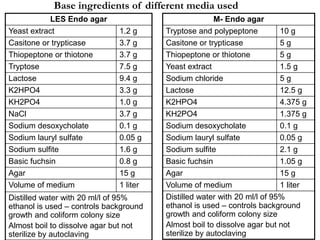
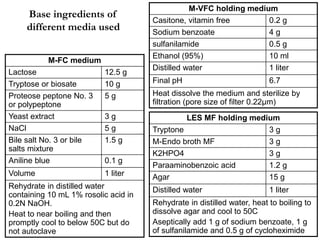
This document describes the MPN (Most Probable Number) test for testing biological water quality and detecting the presence of fecal contamination and pathogens. It involves testing for the indicator organism E. coli using a multiple tube fermentation technique with three phases - presumptive, confirmed, and completed tests. Samples are collected and stored properly then inoculated into lactose broth tubes at serial dilutions and incubated to detect coliform growth. Positive tubes are then tested with BGLB/MacConkey broth and EC/A1 broth to confirm total and fecal coliforms, respectively. Definitive identification involves plating and gram staining from positive confirmed tubes.