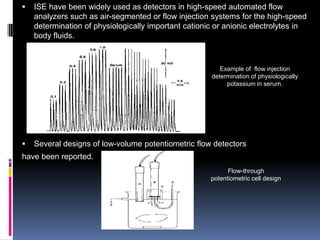
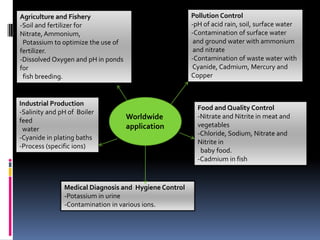
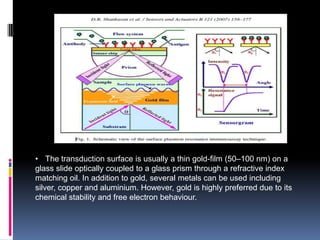
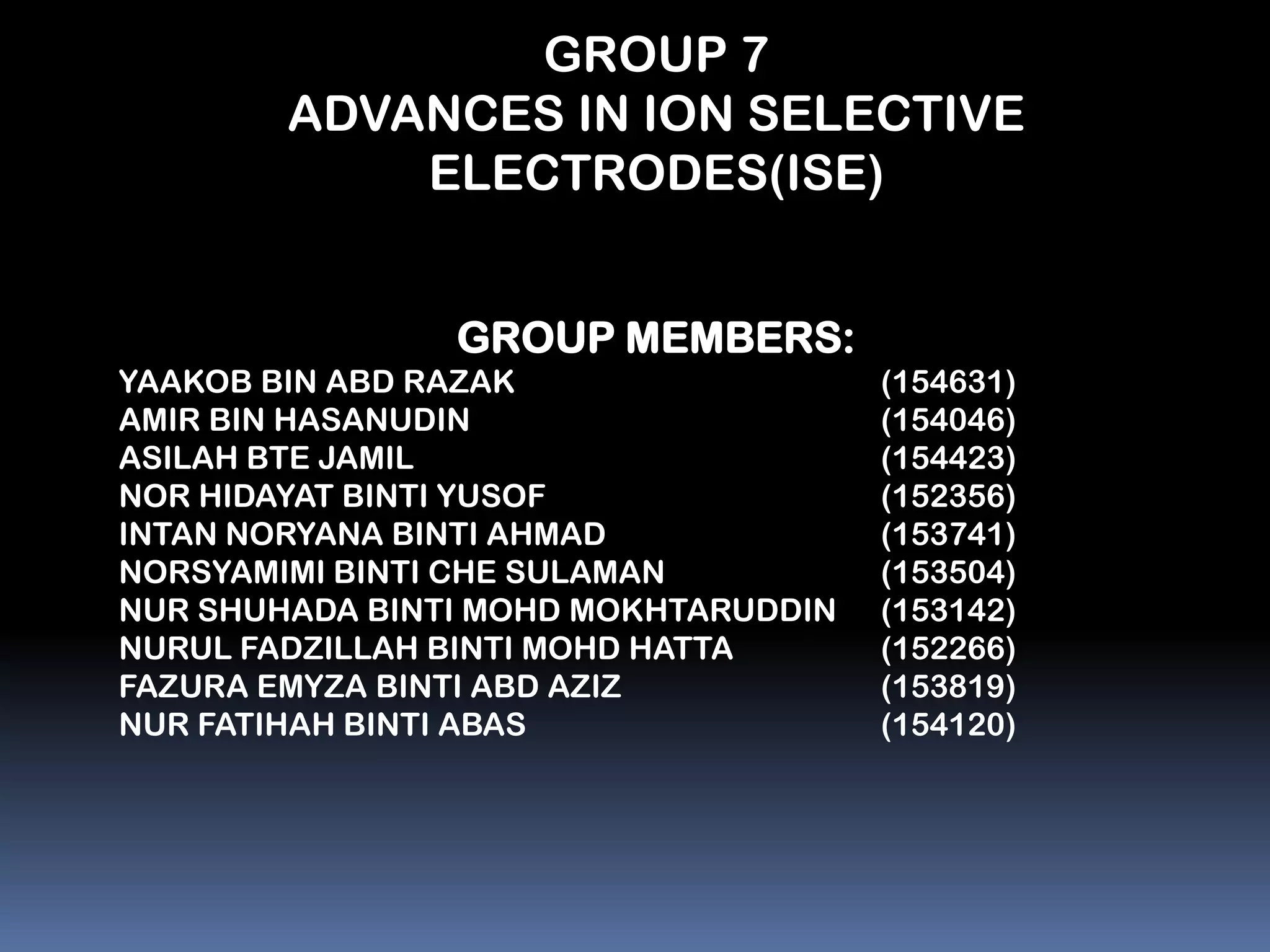
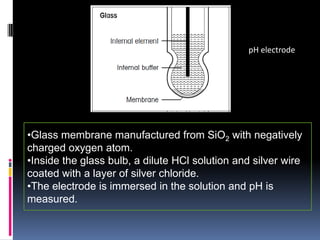
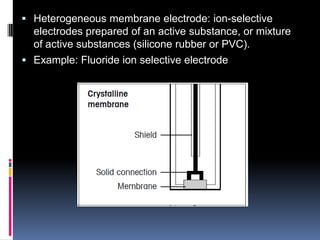
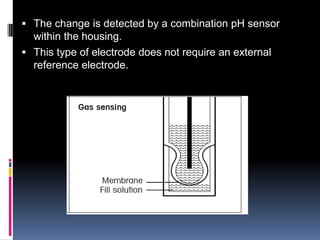
The document summarizes a group project on advances in ion selective electrodes. It discusses the different types of ion selective electrodes including glass membrane electrodes, solid state electrodes, liquid membrane electrodes, and gas sensing electrodes. It describes the key parameters that characterize ion selective electrodes such as sensitivity, selectivity, detection limit, and response time. The document also discusses various applications of ion selective electrodes for online, on-site, and in vivo potentiometric measurements. Recent advances in the applications of ion selective electrodes in areas such as agriculture, pollution control, food quality control, medical diagnostics, and industrial production are highlighted.














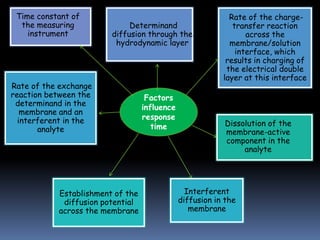
![Response time
From the time the ISE and a reference electrode are
dipped in the sample solution (or the time at which the
ion concentration in a solution in contact with ISE and a
reference electrode is changed ) to when the potential of
the cell becomes equal to its steady-state value within 1
[mV] or has reached 90% of the final value (in certain
cases also 63% or 95%).
The response time usually increases with decreasing
determinand concentration](https://image.slidesharecdn.com/group7-121209224310-phpapp01/85/Advances-in-Ion-Selective-Electrodes-ISE-20-320.jpg)