Hess's law
•Download as PPT, PDF•
1 like•3,321 views
Hess's law states that the total enthalpy change for a reaction is independent of the pathway between the initial and final states. To calculate the enthalpy change of a reaction, standard enthalpy change values can be used for the elementary steps that add up to the overall reaction through algebraic manipulation. For the reaction of 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g), using standard enthalpy change values for the elementary steps gives an overall exothermic enthalpy change of -906.3 kJ.
Report
Share
Report
Share
Recommended
Chemical Reactions: Thermochemistry

Lecture materials for the Introductory Chemistry course for Forensic Scientists, University of Lincoln, UK. See http://forensicchemistry.lincoln.ac.uk/ for more details.
Recommended
Chemical Reactions: Thermochemistry

Lecture materials for the Introductory Chemistry course for Forensic Scientists, University of Lincoln, UK. See http://forensicchemistry.lincoln.ac.uk/ for more details.
Enthalpy, Calorimetry, Hess's Law

Here's a simple presentation of Enthalpy, Calorimetry, and Hess's Law.
Chapter 3 Alkenes: Structures, Nomenclature, and an Introduction to Reacti...

Structures, Nomenclature, and an Introduction to Reactivity Thermodynamics and Kinetics
Unit 4 chemical kinetics (Class XII)

This unit includes: rate of a chemical reaction, graphs,, unit of rate, average rate& instantaneous rate,. factors influuncing rate of a reaction, Rate expression & rate constant, Order & molecularity of a reaction,, initiall rate method & integrated rate law equations, numerical problems,, Half life period, Pseudo first order reaction, Temperature of rate of reaction, Activation energy, collision frequency & effective collision, Collision theory, Arrhenius equation,, effect of catalyst on rate of reaction, numerical problems
More Related Content
What's hot
Enthalpy, Calorimetry, Hess's Law

Here's a simple presentation of Enthalpy, Calorimetry, and Hess's Law.
Chapter 3 Alkenes: Structures, Nomenclature, and an Introduction to Reacti...

Structures, Nomenclature, and an Introduction to Reactivity Thermodynamics and Kinetics
Unit 4 chemical kinetics (Class XII)

This unit includes: rate of a chemical reaction, graphs,, unit of rate, average rate& instantaneous rate,. factors influuncing rate of a reaction, Rate expression & rate constant, Order & molecularity of a reaction,, initiall rate method & integrated rate law equations, numerical problems,, Half life period, Pseudo first order reaction, Temperature of rate of reaction, Activation energy, collision frequency & effective collision, Collision theory, Arrhenius equation,, effect of catalyst on rate of reaction, numerical problems
What's hot (20)
IB Chemistry on Hess's Law, Enthalpy Formation and Combustion

IB Chemistry on Hess's Law, Enthalpy Formation and Combustion
Chapter 3 Alkenes: Structures, Nomenclature, and an Introduction to Reacti...

Chapter 3 Alkenes: Structures, Nomenclature, and an Introduction to Reacti...
Viewers also liked
Viewers also liked (6)
Similar to Hess's law
Module 7 - Energy Balance chemical process calculations

Chemical process calculations involve various computations and analyses to design, optimize, and understand chemical processes. Here are some descriptions highlighting different aspects of chemical process calculations:
Material Balances:
Material balances are the cornerstone of chemical process calculations, ensuring that the amounts of all components entering and leaving a system are properly accounted for.
These calculations involve tracking the flow rates and compositions of substances throughout a process, often using mass or mole balances.
Energy Balances:
Energy balances involve quantifying the energy inputs and outputs in a chemical process.
These calculations are crucial for understanding the heat transfer requirements, evaluating energy efficiency, and optimizing process conditions.
Reaction Kinetics:
Chemical reactions kinetics calculations focus on understanding the rates at which reactions occur and how they are influenced by various factors such as temperature, pressure, and catalysts.
These calculations help in determining the optimal reaction conditions and designing reactors for desired conversion rates.
Phase Equilibrium Calculations:
Phase equilibrium calculations deal with determining the distribution of components between different phases in a system, such as liquid-liquid or vapor-liquid equilibrium.
These calculations are essential for designing separation processes like distillation, extraction, and absorption.
Thermodynamic Calculations:
Thermodynamic calculations involve applying thermodynamic principles to predict the behavior of chemical systems.
These calculations include determining properties such as enthalpy, entropy, Gibbs free energy, and fugacity, which are crucial for process design and optimization.
Process Simulation:
Process simulation involves using computer software to model and simulate chemical processes.
These simulations allow engineers to predict process behavior under different operating conditions, optimize process parameters, and troubleshoot potential issues.
7
network error
Enthalpy diagram

Phase is a form of matter that is uniform with respect to chemical composition and the state of aggregation on both microscopic and macroscopic length scales.
Determination of Hess’s Law (Direct or Indirect)
Similar to Hess's law (20)
IB Chemistry on Hess's Law, Enthalpy Formation and Combustion

IB Chemistry on Hess's Law, Enthalpy Formation and Combustion
Module 7 - Energy Balance chemical process calculations

Module 7 - Energy Balance chemical process calculations
Hess law thermodynamic first law of thermodynamic .pdf

Hess law thermodynamic first law of thermodynamic .pdf
Tang 01b enthalpy, entropy, and gibb's free energy

Tang 01b enthalpy, entropy, and gibb's free energy
More from tanzmanj
More from tanzmanj (20)
Recently uploaded
Neuro-symbolic is not enough, we need neuro-*semantic*

Neuro-symbolic (NeSy) AI is on the rise. However, simply machine learning on just any symbolic structure is not sufficient to really harvest the gains of NeSy. These will only be gained when the symbolic structures have an actual semantics. I give an operational definition of semantics as “predictable inference”.
All of this illustrated with link prediction over knowledge graphs, but the argument is general.
Transcript: Selling digital books in 2024: Insights from industry leaders - T...

The publishing industry has been selling digital audiobooks and ebooks for over a decade and has found its groove. What’s changed? What has stayed the same? Where do we go from here? Join a group of leading sales peers from across the industry for a conversation about the lessons learned since the popularization of digital books, best practices, digital book supply chain management, and more.
Link to video recording: https://bnctechforum.ca/sessions/selling-digital-books-in-2024-insights-from-industry-leaders/
Presented by BookNet Canada on May 28, 2024, with support from the Department of Canadian Heritage.
De-mystifying Zero to One: Design Informed Techniques for Greenfield Innovati...

De-mystifying Zero to One: Design Informed Techniques for Greenfield Innovation With Your Product by VP of Product Design, Warner Music Group
The Art of the Pitch: WordPress Relationships and Sales

Clients don’t know what they don’t know. What web solutions are right for them? How does WordPress come into the picture? How do you make sure you understand scope and timeline? What do you do if sometime changes?
All these questions and more will be explored as we talk about matching clients’ needs with what your agency offers without pulling teeth or pulling your hair out. Practical tips, and strategies for successful relationship building that leads to closing the deal.
Quantum Computing: Current Landscape and the Future Role of APIs

The current state of quantum computing and the role of APIs to take it mainstream.
Connector Corner: Automate dynamic content and events by pushing a button

Here is something new! In our next Connector Corner webinar, we will demonstrate how you can use a single workflow to:
Create a campaign using Mailchimp with merge tags/fields
Send an interactive Slack channel message (using buttons)
Have the message received by managers and peers along with a test email for review
But there’s more:
In a second workflow supporting the same use case, you’ll see:
Your campaign sent to target colleagues for approval
If the “Approve” button is clicked, a Jira/Zendesk ticket is created for the marketing design team
But—if the “Reject” button is pushed, colleagues will be alerted via Slack message
Join us to learn more about this new, human-in-the-loop capability, brought to you by Integration Service connectors.
And...
Speakers:
Akshay Agnihotri, Product Manager
Charlie Greenberg, Host
Search and Society: Reimagining Information Access for Radical Futures

The field of Information retrieval (IR) is currently undergoing a transformative shift, at least partly due to the emerging applications of generative AI to information access. In this talk, we will deliberate on the sociotechnical implications of generative AI for information access. We will argue that there is both a critical necessity and an exciting opportunity for the IR community to re-center our research agendas on societal needs while dismantling the artificial separation between the work on fairness, accountability, transparency, and ethics in IR and the rest of IR research. Instead of adopting a reactionary strategy of trying to mitigate potential social harms from emerging technologies, the community should aim to proactively set the research agenda for the kinds of systems we should build inspired by diverse explicitly stated sociotechnical imaginaries. The sociotechnical imaginaries that underpin the design and development of information access technologies needs to be explicitly articulated, and we need to develop theories of change in context of these diverse perspectives. Our guiding future imaginaries must be informed by other academic fields, such as democratic theory and critical theory, and should be co-developed with social science scholars, legal scholars, civil rights and social justice activists, and artists, among others.
Smart TV Buyer Insights Survey 2024 by 91mobiles.pdf

91mobiles recently conducted a Smart TV Buyer Insights Survey in which we asked over 3,000 respondents about the TV they own, aspects they look at on a new TV, and their TV buying preferences.
Newntide latest company Introduction.pdf

NEWNTIDE, a leading brand in China's air energy industry, drives industry development with technological innovation, implementing national energy-saving and emission reduction policies. It pioneers an industry-focused multi-energy product line, adopting experiential marketing to meet diverse customer needs. The company has departments for R&D, marketing, operations, and sales, aiming to ultimately achieve "technological innovation, environmental friendliness, standardized management, and high-quality" as a high-tech enterprise integrating business and technical R&D, production, sales, and service.
NEWNTIDE boasts the most comprehensive support service network in the industry. Its earliest products cover 25 series, including split, integrated, wall-mounted, cabinet, and upright types, with over 100 diverse products. Commercial products include floor heating, air heaters, air conditioners for heating and cooling, oxidation and nitrogen air conditioners, and high-temperature heating. The products feature comprehensive intelligent technology management, cloud control technology, rapid heating technology, basic protection technology, remote control technology, DC inverter technology, and remote WIFI smart control, achieving a leading position in the industry with SMART interactive technology.
For over a decade, the company has adhered to a "people-oriented" business philosophy, strictly implementing industry 7S management, ISO9001/ISO14001 quality and environmental systems, and industry standards to ensure stable product quality and meet customers' dual requirements for product safety and environmental protection.
Leading the development of intelligence with technological innovation, NEWNTIDE has become a national demonstration base for the transformation of scientific and technological achievements, awarded the "China Energy Saving Technology Contribution Award" and "China Energy Science and Technology Progress Award". The company adopts a strategy of high standards, high quality, and high-tech for key products, holding core technologies and competitive advantages. It also organizes multiple strategic support projects known as the "18 Key Operational Projects" and "18 Key Operational Strategies," driving technology project approvals with multidimensional strategic product quality modules and comprehensive practical operations to enhance the quality of all products.
Since its establishment, NEWNTIDE has always committed to providing high-quality and high-end intelligent heat pump products, serving billions of global families with the goal of creating a sustainable and prosperous environment. The development of NEWNTIDE has been supported by various levels of government and widely recognized and cooperated with by internationally renowned institutions, taking on a social responsibility of providing tranquility and happiness while enjoying the environment.
Let safe heat pumps be a necessity for a beautiful human life.
Bits & Pixels using AI for Good.........

A whirlwind tour of tech & AI for socio-environmental impact.
JMeter webinar - integration with InfluxDB and Grafana

Watch this recorded webinar about real-time monitoring of application performance. See how to integrate Apache JMeter, the open-source leader in performance testing, with InfluxDB, the open-source time-series database, and Grafana, the open-source analytics and visualization application.
In this webinar, we will review the benefits of leveraging InfluxDB and Grafana when executing load tests and demonstrate how these tools are used to visualize performance metrics.
Length: 30 minutes
Session Overview
-------------------------------------------
During this webinar, we will cover the following topics while demonstrating the integrations of JMeter, InfluxDB and Grafana:
- What out-of-the-box solutions are available for real-time monitoring JMeter tests?
- What are the benefits of integrating InfluxDB and Grafana into the load testing stack?
- Which features are provided by Grafana?
- Demonstration of InfluxDB and Grafana using a practice web application
To view the webinar recording, go to:
https://www.rttsweb.com/jmeter-integration-webinar
GenAISummit 2024 May 28 Sri Ambati Keynote: AGI Belongs to The Community in O...

“AGI should be open source and in the public domain at the service of humanity and the planet.”
Assuring Contact Center Experiences for Your Customers With ThousandEyes

Presented by Suzanne Phillips and Alex Marcotte
When stars align: studies in data quality, knowledge graphs, and machine lear...

Keynote at DQMLKG workshop at the 21st European Semantic Web Conference 2024
State of ICS and IoT Cyber Threat Landscape Report 2024 preview

The IoT and OT threat landscape report has been prepared by the Threat Research Team at Sectrio using data from Sectrio, cyber threat intelligence farming facilities spread across over 85 cities around the world. In addition, Sectrio also runs AI-based advanced threat and payload engagement facilities that serve as sinks to attract and engage sophisticated threat actors, and newer malware including new variants and latent threats that are at an earlier stage of development.
The latest edition of the OT/ICS and IoT security Threat Landscape Report 2024 also covers:
State of global ICS asset and network exposure
Sectoral targets and attacks as well as the cost of ransom
Global APT activity, AI usage, actor and tactic profiles, and implications
Rise in volumes of AI-powered cyberattacks
Major cyber events in 2024
Malware and malicious payload trends
Cyberattack types and targets
Vulnerability exploit attempts on CVEs
Attacks on counties – USA
Expansion of bot farms – how, where, and why
In-depth analysis of the cyber threat landscape across North America, South America, Europe, APAC, and the Middle East
Why are attacks on smart factories rising?
Cyber risk predictions
Axis of attacks – Europe
Systemic attacks in the Middle East
Download the full report from here:
https://sectrio.com/resources/ot-threat-landscape-reports/sectrio-releases-ot-ics-and-iot-security-threat-landscape-report-2024/
Recently uploaded (20)
Neuro-symbolic is not enough, we need neuro-*semantic*

Neuro-symbolic is not enough, we need neuro-*semantic*
Transcript: Selling digital books in 2024: Insights from industry leaders - T...

Transcript: Selling digital books in 2024: Insights from industry leaders - T...
De-mystifying Zero to One: Design Informed Techniques for Greenfield Innovati...

De-mystifying Zero to One: Design Informed Techniques for Greenfield Innovati...
Free Complete Python - A step towards Data Science

Free Complete Python - A step towards Data Science
The Art of the Pitch: WordPress Relationships and Sales

The Art of the Pitch: WordPress Relationships and Sales
Quantum Computing: Current Landscape and the Future Role of APIs

Quantum Computing: Current Landscape and the Future Role of APIs
Connector Corner: Automate dynamic content and events by pushing a button

Connector Corner: Automate dynamic content and events by pushing a button
Search and Society: Reimagining Information Access for Radical Futures

Search and Society: Reimagining Information Access for Radical Futures
Smart TV Buyer Insights Survey 2024 by 91mobiles.pdf

Smart TV Buyer Insights Survey 2024 by 91mobiles.pdf
FIDO Alliance Osaka Seminar: Passkeys at Amazon.pdf

FIDO Alliance Osaka Seminar: Passkeys at Amazon.pdf
JMeter webinar - integration with InfluxDB and Grafana

JMeter webinar - integration with InfluxDB and Grafana
FIDO Alliance Osaka Seminar: FIDO Security Aspects.pdf

FIDO Alliance Osaka Seminar: FIDO Security Aspects.pdf
GenAISummit 2024 May 28 Sri Ambati Keynote: AGI Belongs to The Community in O...

GenAISummit 2024 May 28 Sri Ambati Keynote: AGI Belongs to The Community in O...
Assuring Contact Center Experiences for Your Customers With ThousandEyes

Assuring Contact Center Experiences for Your Customers With ThousandEyes
When stars align: studies in data quality, knowledge graphs, and machine lear...

When stars align: studies in data quality, knowledge graphs, and machine lear...
State of ICS and IoT Cyber Threat Landscape Report 2024 preview

State of ICS and IoT Cyber Threat Landscape Report 2024 preview
FIDO Alliance Osaka Seminar: Passkeys and the Road Ahead.pdf

FIDO Alliance Osaka Seminar: Passkeys and the Road Ahead.pdf
Hess's law
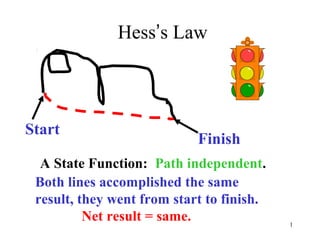
- 1. 1 Hess’s Law Start Finish A State Function: Path independent. Both lines accomplished the same result, they went from start to finish. Net result = same.
- 2. 2 Determine the heat of reaction for the reaction: 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) Using the following sets of reactions: N2(g) + O2(g) 2NO(g) ∆H = 180.6 kJ N2(g) + 3H2(g) 2NH3(g) ∆H = -91.8 kJ 2H2(g) + O2(g) 2H2O(g) ∆H = -483.7 kJ Hint: The three reactions must be algebraically manipulated to sum up to the desired reaction. and.. the ∆H values must be treated accordingly.
- 3. 3 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) Using the following sets of reactions: N2(g) + O2(g) 2NO(g) ∆H = 180.6 kJ N2(g) + 3H2(g) 2NH3(g) ∆H = -91.8 kJ 2H2(g) + O2(g) 2H2O(g) ∆H = -483.7 kJ Goal: NH3: O2 : NO: H2O: Reverse and x 2 4NH3 2N2 + 6H2 ∆H = +183.6 kJ Found in more than one place, SKIP IT (its hard). x2 2N2 + 2O2 4NO ∆H = 361.2 kJ x3 6H2 + 3O2 6H2O ∆H = -1451.1 kJ
- 4. 4 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g)Goal: NH3: O2 : NO: H2O: Reverse and x2 4NH3 2N2 + 6H2 ∆H = +183.6 kJ Found in more than one place, SKIP IT. x2 2N2 + 2O2 4NO ∆H = 361.2 kJ x3 6H2 + 3O2 6H2O ∆H = -1451.1 kJ Cancel terms and take sum. 4NH3 + 5O2 4NO + 6H2O ∆H = -906.3 kJ Is the reaction endothermic or exothermic?
- 5. 5 Determine the heat of reaction for the reaction: C2H4(g) + H2(g) C2H6(g) Use the following reactions: C2H4(g) + 3O2(g) 2CO2(g) + 2H2O(l) ∆H = -1401 kJ C2H6(g) + 7/2O2(g) 2CO2(g) + 3H2O(l) ∆H = -1550 kJ H2(g) + 1/2O2(g) H2O(l) ∆H = -286 kJ Consult your neighbor if necessary.
- 6. 6 Determine the heat of reaction for the reaction: Goal: C2H4(g) + H2(g) C2H6(g) ∆H = ? Use the following reactions: C2H4(g) + 3O2(g) 2CO2(g) + 2H2O(l) ∆H = -1401 kJ C2H6(g) + 7/2O2(g) 2CO2(g) + 3H2O(l) ∆H = -1550 kJ H2(g) + 1/2O2(g) H2O(l) ∆H = -286 kJ C2H4(g) :use 1 as is C2H4(g) + 3O2(g) 2CO2(g) + 2H2O(l) ∆H = -1401 kJ H2(g) :# 3 as is H2(g) + 1/2O2(g) H2O(l) ∆H = -286 kJ C2H6(g) : rev #2 2CO2(g) + 3H2O(l) C2H6(g) + 7/2O2(g) ∆H = +1550 kJ C2H4(g) + H2(g) C2H6(g) ∆H = -137 kJ
- 7. 7 Summary: enthalpy is a state function and is path independent.
- 8. 8
- 9. 9 Standard Enthalpies of formation:
- 10. 10 Thermodynamic Quantities of Selected Substances @ 298.15 K