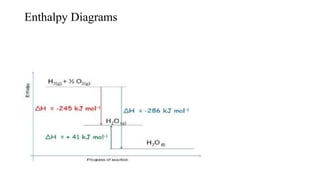
The document discusses enthalpy diagrams, which illustrate the relative internal energy of a system before and after a reaction, and how they relate to phases of matter. It explains Hess's Law, showing that the enthalpy change for an overall process can be determined by summing the enthalpy changes of individual steps, regardless of the path taken. The document also provides examples of calculating standard enthalpies of reaction and how to apply Hess's Law in chemical equations.












![Standard Enthalpies of Reaction
Use Hess s law to calculate the standard enthalpy of reaction for the decomposition of
sodium bicarbonate, a minor reaction that occurs when baking soda is used in baking.
2𝑁𝑎𝐻𝐶𝑂3 𝑠 → 𝑁𝑎2 𝐶𝑂3 + 𝐻2 𝑂 𝑙 + 𝐶𝑂2 𝑔 ∆H° = ?
From Hess s law, we see that the following four equations yield the above given equation
when added together.
𝑎): 2𝑁𝑎𝐻𝐶𝑂3 𝑠 → 2𝑁𝑎 𝑠 + 𝐻2 𝑔 + 2𝐶 𝑔𝑟𝑎𝑝ℎ𝑖𝑡𝑒 + 3𝑂2 (g)
∆H° =-2 x ∆Hf° = [NaHCO3 (s)]
𝑏): 2𝑁𝑎 𝑠 + 𝐶 𝑔𝑟𝑎𝑝ℎ𝑖𝑡𝑒 +
3
2
𝑂2 𝑔 → 𝑁𝑎2 𝐶𝑂3 𝑠
∆H° = ∆Hf°[Na2CO3) (s)](https://image.slidesharecdn.com/enthalpydiagram-200409020944/85/Enthalpy-diagram-14-320.jpg)
![𝐶) ∶ 𝐻2 𝑔 +
1
2
𝑂2 𝑔 → 𝐻2 𝑂 𝑙 ∆H° = ∆Hf°[H2O] (l)
d): C(graphite) + 𝑂2 𝑔 → 𝐶𝑂2 𝑔 ∆H° = ∆Hf°[CO2] (g)
for reaction (a) is the negative of twice ∆Hf°[Na2CO3) (s).
Equations (b), (c) and (d) represent the formation of one mole each of Na2CO3 (s), CO2
(g) and H2O (l) and Thus, we can express the value of for the decomposition reaction as
∆H° = ∆Hf°[Na2CO3] (s) + ∆Hf°[H2O] (l) +∆Hf°[CO2] (g) - 2 * ∆Hf°[NaHCO3] (s)
The enthalpy change for the overall reaction is the sum of the standard
enthalpy changes of the individual steps](https://image.slidesharecdn.com/enthalpydiagram-200409020944/85/Enthalpy-diagram-15-320.jpg)
![∆𝐻 𝑜 = ∆𝐻 𝑑𝑒𝑐𝑜𝑚𝑝𝑜𝑠𝑖𝑡𝑖𝑜𝑛
𝑜
+ ∆𝐻𝑟𝑒𝑐𝑜𝑚𝑏𝑖𝑛𝑎𝑡𝑖𝑜𝑛
𝑜
∆ 𝐻 𝑑𝑒𝑐𝑜𝑚𝑝𝑜𝑠𝑖𝑡𝑖𝑜𝑛
𝑜
= ∗ 2 𝑥 ∆𝐻𝑓
𝑜
[𝑁𝑎𝐻𝐶𝑂3 𝑠 ]
∆ 𝐻𝑟𝑒𝑐𝑜𝑚𝑏𝑖𝑛𝑎𝑡𝑖𝑜𝑛
𝑜
= ∆𝐻𝑓
𝑜
𝑁𝑎2 𝐶𝑂3 𝑠 + ∆𝐻𝑓
𝑜
𝐻2 𝑂 𝑙 + ∆𝐻𝑓
𝑜
[ 𝐶𝑂2 𝑔 ]
∆𝐻 𝑜
= ∆𝐻𝑓
𝑜
𝑁𝑎2 𝐶𝑂3 𝑠 + ∆𝐻𝑓
𝑜
𝐻2 𝑂 𝑙 + ∆𝐻𝑓
𝑜
𝐶𝑂2 𝑔 − 2 𝑥 ∆𝐻𝑓
𝑜
[𝑁𝑎𝐻𝐶𝑂3 𝑠 ]
∆𝐻 𝑜
= 𝑣 𝑃 𝐻𝑓
𝑜
(𝑃𝑟𝑜𝑑𝑢𝑐𝑡𝑠) − 𝑣𝑔 ∆ 𝐻 𝑜
(𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡𝑠) 7.21](https://image.slidesharecdn.com/enthalpydiagram-200409020944/85/Enthalpy-diagram-16-320.jpg)

![Calculating an Unknown Value
Use the data here and in Table 7.2 to calculate of benzene, C6H6 (l)
2𝐶2 𝐻6 𝑙 + 15 𝑂2 𝑔 → 12𝐶𝑂2 𝑔 + 6𝐻2 𝑂 𝑙 ∆𝐻0 = −6535 𝑘𝐽
Solution
2𝐶2 𝐻6 𝑙 + 15 𝑂2 𝑔 → 12𝐶𝑂2 𝑔 + 6𝐻2 𝑂 𝑙 ∆𝐻0
= −6535 𝑘𝐽
? 0 -393.5 -2858
∆𝐻 𝑜
= 𝑣 𝑃 𝐻𝑓
𝑜
(𝑃𝑟𝑜𝑑𝑢𝑐𝑡𝑠) − 𝑣𝑟 ∆ 𝐻 𝑓
𝑜
(𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡𝑠) 7.21
∆𝐻 𝑜
= 12 𝑚𝑜𝑙 𝐶𝑂2 𝑥 −393.5
𝑘𝐽
𝑚𝑜𝑙
+ 6 𝑚𝑜𝑙 𝐻2 𝑂 𝑥 −285.5
𝑘𝐽
𝑚𝑜𝑙
− 2 𝑚𝑜𝑙 𝐶6 𝐻6 𝑥 ∆𝐻𝑓
0
𝐶6 𝐻6 𝑙 } = −6535 𝑘𝐽
∆𝐻𝑓
0
𝐶6 𝐻6 𝑙 = ? [Solve]](https://image.slidesharecdn.com/enthalpydiagram-200409020944/85/Enthalpy-diagram-18-320.jpg)

![∆ 𝐻 𝑜 = 1 𝑚𝑜𝑙 𝐻2 𝑂 𝑥 ∆𝐻𝑓
𝑜
𝐻2 𝑂 𝑙
− 1 𝑚𝑜𝑙 𝐻+
𝑥 ∆𝐻𝑓
𝑜
𝑎𝑞 ] + 1 𝑚𝑜𝑙 𝑂𝐻−
𝑥 ∆𝐻𝑓
𝑜
(𝑎𝑞)] = −55.8 𝑘𝐽
∆𝐻𝑓
𝑜
𝑂𝐻− 𝑎𝑞 =
55.8 𝑘𝐽 + (1 𝑚𝑜𝑙 𝐻2 𝑂 ∆ 𝐻𝑓
𝑜
𝐻2 𝑂 𝑙 − ( 1 𝑚𝑜𝑙 𝐻+
𝑥 ∆𝐻𝑓
𝑜
𝐻+
𝑎𝑞 )
1 𝑚𝑜𝑙 𝑂𝐻−
∆𝐻𝑓
𝑜
𝑂𝐻− 𝑎𝑞 =
55.8 𝑘𝐽 − 285.8𝑘𝐽 − 0 𝑘𝐽
1 𝑚𝑜𝑙 𝑂𝐻−
= −230.0
𝑘𝐽
𝑚𝑜𝑙
𝑂𝐻−](https://image.slidesharecdn.com/enthalpydiagram-200409020944/85/Enthalpy-diagram-20-320.jpg)
![Problem Statement
Ammonia is used in the industrial preparation of nitric acid according to the equation:
4 NH3(g) + 5O2(g) → 4 NO(g) + 6 H2O(g)
What is the standard enthalpy change ∆H for this reaction?
∆𝐻 𝑜 = 𝑣 𝑃 𝐻𝑓
𝑜
(𝑃𝑟𝑜𝑑𝑢𝑐𝑡𝑠) − 𝑣𝑟 ∆ 𝐻𝑓
𝑜
(𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡𝑠) 7.21
∆Ho = [4 × (90.3) + 6 × (-241.8)] – [4 × (-45.9) + 5 × (0)]
= -906 kJ
Solution](https://image.slidesharecdn.com/enthalpydiagram-200409020944/85/Enthalpy-diagram-21-320.jpg)