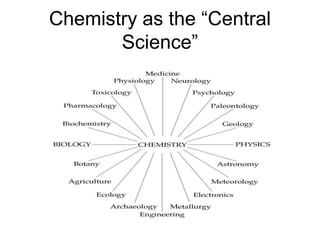
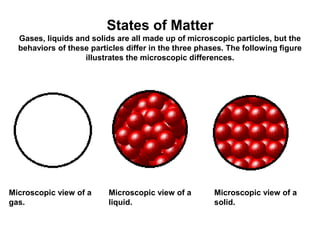
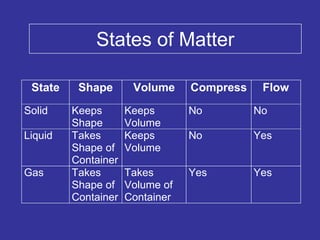
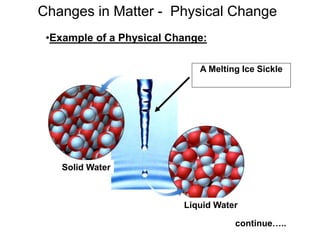
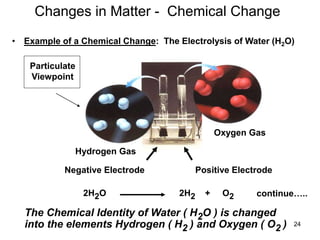
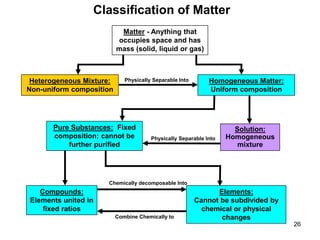
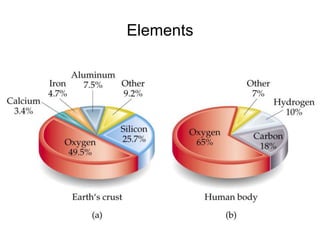
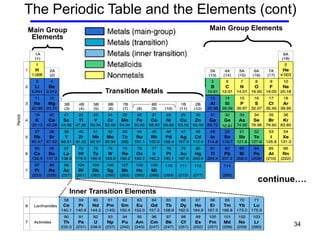
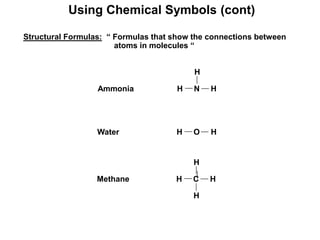
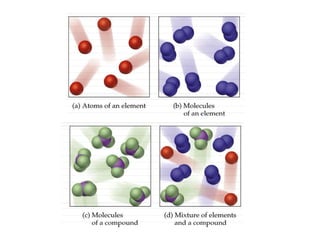
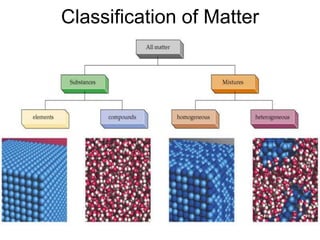
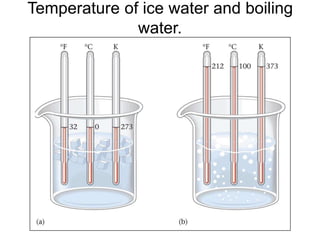
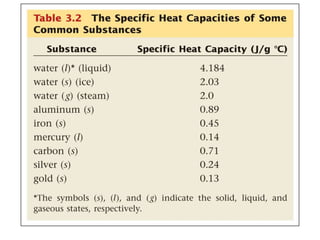
The document discusses the importance and relevance of studying chemistry. It notes that chemistry is important to understand as an informed consumer, to make better decisions, and to develop problem-solving and analytical thinking skills. It also explains that chemistry is the study of matter and its transformations, and that everything in the world can be described through a chemical lens. Chemistry is described as the "central science" because it is interconnected with many other fields like art, economics, politics, and natural resources.