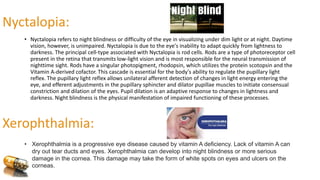
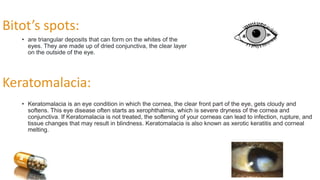
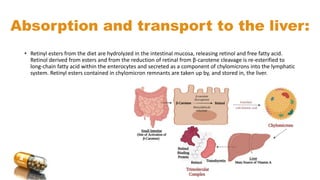
Vitamin A is a lipid-soluble vitamin essential for various bodily functions, particularly vision, immune support, and cell growth. It has multiple forms, including retinol, retinal, and retinoic acid, and is primarily sourced from animal products and certain vegetables rich in beta-carotene. Deficiency can lead to serious visual impairments such as night blindness and xerophthalmia, while excess intake can cause toxicity with severe health effects.





![Retinoic acid mechanism of action:
• Retinol is oxidized to retinoic acid. Retinoic acid binds with high affinity to specific receptor proteins (retinoic
acid receptors [RAR]) present in the nucleus of target tissues such as epithelial cells. The activated retinoic
acid–RAR complex binds to response elements on DNA and recruit’s activators or repressors to regulate
retinoid-specific RNA synthesis, resulting in control of the production of specific proteins that mediate
several physiologic functions. For example, retinoids control the expression of the gene for keratin in most
epithelial tissues of the body.](https://image.slidesharecdn.com/vitamina-240211114618-a8c3f9f9/85/vitamin-A-pptx-metabolism-and-function-7-320.jpg)