Vendor qualification flow chart
Manufacture of Medicinal Products and the Active Pharmaceutical Ingredients (APIs) used as starting materials in the production of these products is subject to strict good manufacturing practice regulations that are designed to ensure their quality, safety and efficacy. This ensures that patients worldwide and at any time can have confidence in the quality, safety and efficacy of medicines. The cGMP regulations for final medicinal products are clearly defined in each country and region. The content of the regulations may vary but the objectives are the same: - To deliver high quality, safe medicines manufactured and distributed following controlled procedures to treat diseases and - To prevent deaths, serious illnesses, adverse events or product recalls resulting from deficiencies in the manufacturing and distribution processes. While in the vast majority of cases, the pharmaceutical industry, under the oversight of the Regulatory Authorities and inspectorates consistently applies appropriate cGMP practices,
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Similar to Vendor qualification flow chart
Similar to Vendor qualification flow chart (20)
More from Bangaluru
More from Bangaluru (20)
Recently uploaded
Recently uploaded (20)
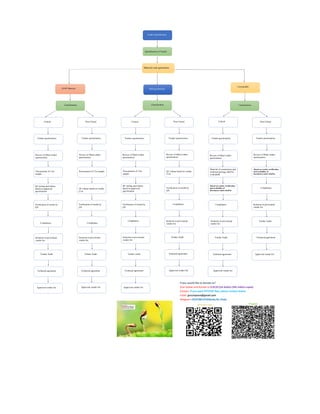
Vendor qualification flow chart
- 1. Vendor Qualification Identification of Vendor Material code generation RAW Material Packing Material Consumable Classification Non Critical Critical Vendor questionaries Review of filled vendor questionaries Procurement of 3 lot sample QC testing and release based on approved specification Verification of results by QA Compliance Inclusion in provisional vendor list Vendor Audit Technical agreement Approved vendor list Vendor questionaries Review of filled vendor questionaries Procurement of 3 lot sample QC release based on vendor CoA Verification of results by QA Compliance Inclusion in provisional vendor list Vendor Audit Technical agreement Approved vendor list Classification Critical Non Critical Vendor questionaries Vendor questionaries Review of filled vendor questionaries Review of filled vendor questionaries Procurement of 3 lot sample QC release based on vendor CoA QC testing and release based on approved specification Verification of results by QA Verification of results by QA Compliance Compliance Inclusion in provisional vendor list Inclusion in provisional vendor list Vendor Audit Technical agreement Approved vendor list Vendor Audit Technical agreement Approved vendor list Classification Critical Non Critical Vendor questionaries Vendor questionaries Review of filled vendor questionaries Review of filled vendor questionaries Material of construction and technical package shall be evaluatedd Based on vendor certification and availablity of document,vendor shall be Based on vendor certification and availablity of document,vendor shall be Compliance Compliance Inclusion in provisional vendor list Inclusion in provisional vendor list Vendor Audit Technical agreement Approved vendor list Vendor Audit Technical agreement Approved vendor list If you would like to donate us? Scan below and donate us 0.013$ (US dollar) (5Rs Indian rupee) Contact: If you want PPT/PDF files, please contact below. Email: gnccmysore@gmail.com Telegram:+919738137533(only for Chat)