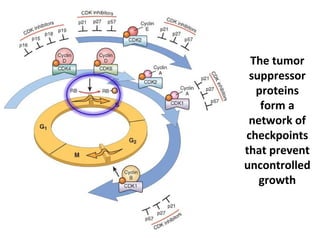
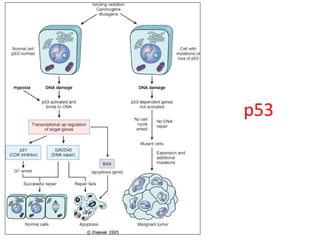
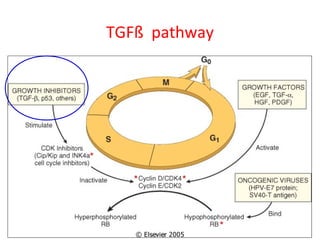
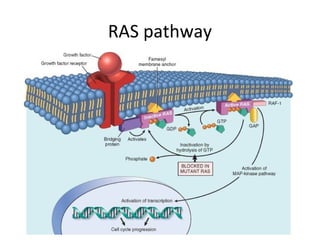
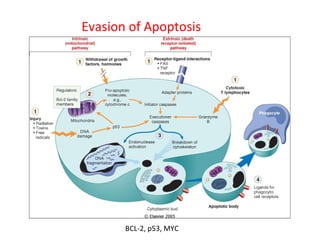
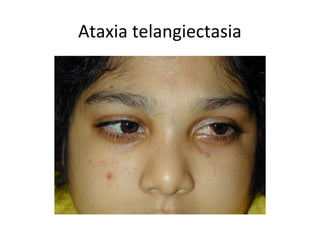
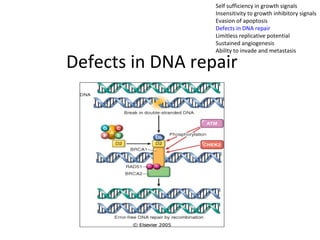
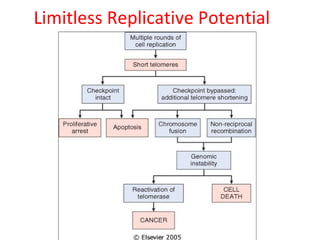
1) Tumor suppressor genes normally apply brakes to cell proliferation through proteins that form checkpoints to prevent uncontrolled growth. Loss of function of these genes allows tumor development.
2) The proteins encoded by tumor suppressor genes regulate cell cycle control, apoptosis, and cell survival/growth through mechanisms like transcription factors, cell cycle inhibitors, and DNA damage response.
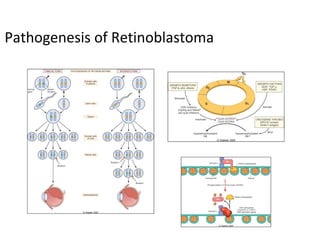
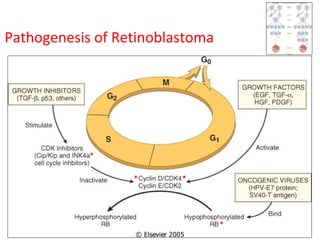
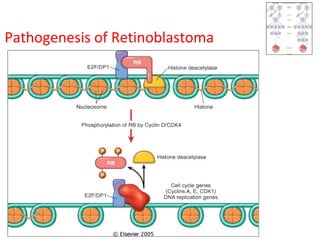
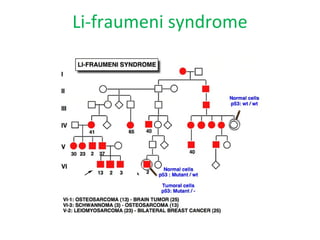
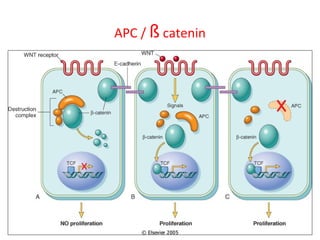
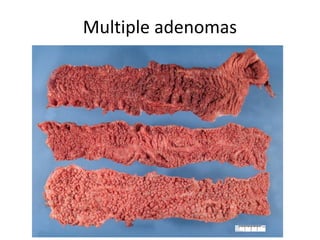
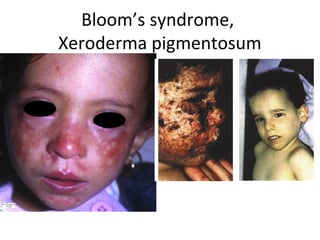
3) Famous tumor suppressor genes include RB, p53, APC, and WT1. Mutation of both copies is required for loss of function, leading to cancers like retinoblastoma, Li-Fraumeni syndrome, colon cancer, and Wilms tumor.