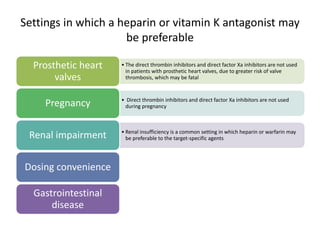
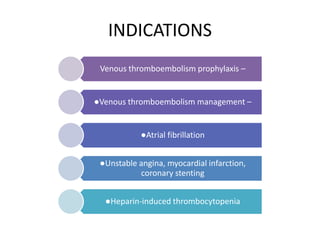
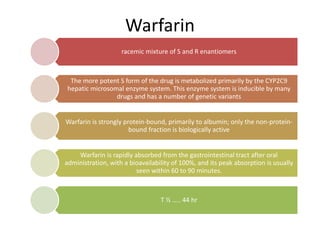
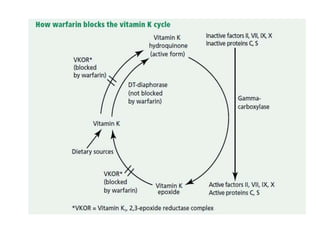
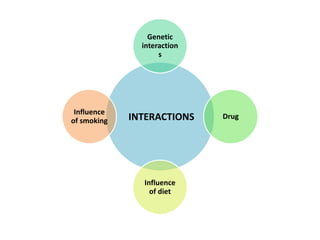
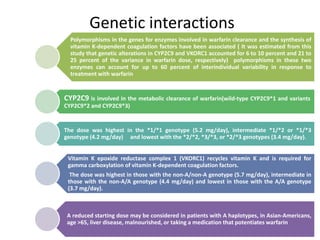
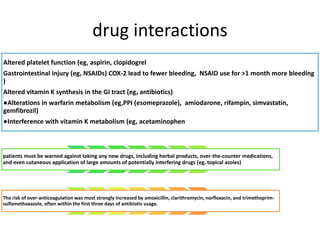
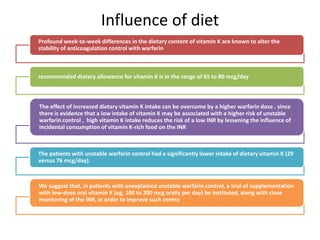
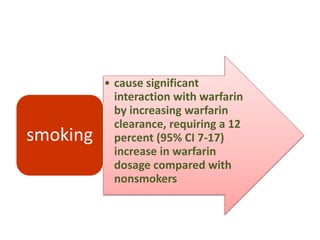
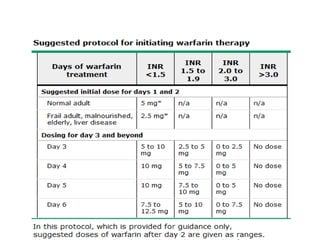
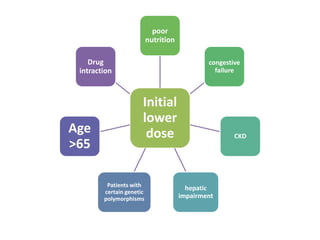
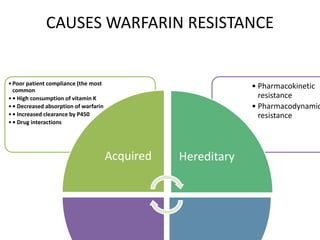
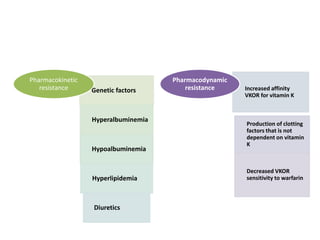
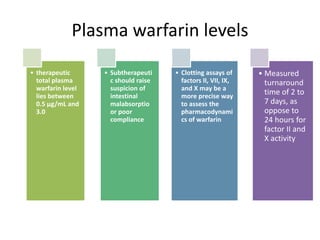
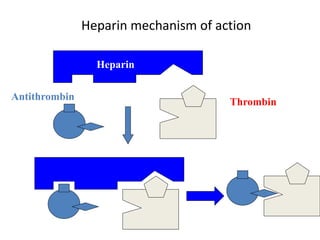
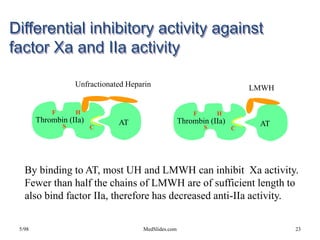
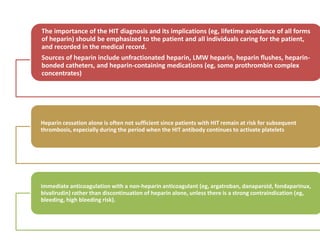
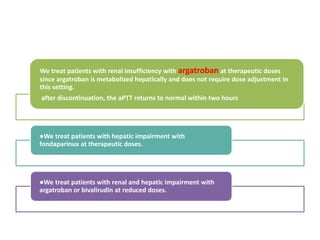
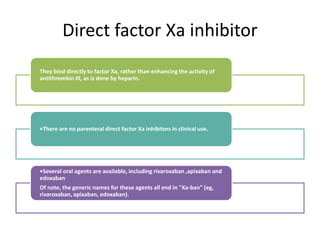
This document discusses treatment with anticoagulation drugs like warfarin and heparin. It provides details on their mechanisms of action, pharmacokinetics, drug interactions, and monitoring. Warfarin is a vitamin K antagonist that inhibits clotting factors. Its effects are delayed and it requires monitoring with an INR test. Heparin works by enhancing antithrombin, indirectly inhibiting thrombin and factor Xa. It has a risk of heparin-induced thrombocytopenia. Both drugs require careful dosing and monitoring due to their narrow therapeutic windows and variability between patients.


















![Advantages LMW heparins vs heparin
• greater bioavailability than unfractionated heparin when given by subcutaneous injection.
• ●The duration of the anticoagulant effect is greater because of reduced binding to macrophages and
endothelial cells, permitting administration only once or twice daily.
• ●The anticoagulant response (anti-Xa activity) to LMW heparin is highly correlated with body weight,
permitting administration of a fixed dose. However, the dose may have to be adjusted for patients who are
extremely obese or have renal
• ●Laboratory monitoring is not necessary in nonpregnant patients; in fact, there is little correlation
between anti-Xa activity and either bleeding or recurrent thrombosis.
• ●They are much less likely to induce immune-mediated thrombocytopenia (ie, heparin-induced
thrombocytopenia) than unfractionated heparin:
• ●They do not increase osteoclast number and activity as much as unfractionated heparin, and may
therefore produce less bone loss.
• ●LMW heparin can be safely administered in the outpatient setting
• ●In patients with acute VTE, fixed-dose subcutaneous LMW heparin is more effective than adjusted-dose
unfractionated heparin for reducing the incidence of symptomatic recurrent VTE, major hemorrhage, and
all-cause mortality [79].
• ●Home management using LMW heparin is cost effective, and is likely to be preferred by patients and](https://image.slidesharecdn.com/teachinganticoagulantsinccuward-161030105130/85/Treatment-with-anticoagulation-35-320.jpg)

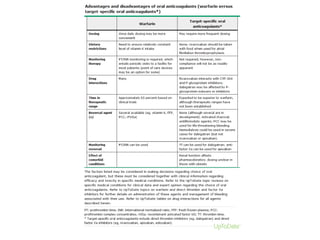
![Advantages over heparin and warfarin
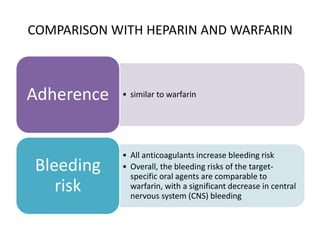
• Heparin and warfarin both have a relatively narrow therapeutic window and
more variable dose-response relationship that depends on a variety of factors;
these features lead to a requirement for frequent monitoring of clotting times
to optimize the therapeutic dose range and prevent bleeding [20,21]. Dose may
be affected by differing bioavailability, diet, and acute medical illnesses. In
contrast, the target-specific oral agents are generally used without a
requirement for monitoring of drug levels or coagulation (clotting) times. This
may be an advantage for patients for whom frequent monitoring is a greater
burden. It remains to be determined whether laboratory monitoring of any of
the target-specific oral agents can further improve their efficacy or safety
Laboratory
monitoring
• Warfarin pharmacokinetics is affected by the level of vitamin K intake and
production in the gastrointestinal tract, as well as induction of hepatic
cytochromes. Thus, warfarin effect can be altered by changes in diet,
administration of other medications, gastrointestinal disorders, and reduced
oral intake. Patients with difficulty controlling the prothrombin
time/international normalized ratio (PT/INR) may benefit from a target-specific
oral agent because these agents have less variability in drug effect for a given
dose than vitamin K antagonists. Affected patients may include those with
unavoidable drug-drug interactions (such as frequent need for antibiotics or a
large number of concomitant and variable medications) or unexplained poor
warfarin control. However, it is important to determine that the INR instability
with a vitamin K antagonist is not due to poor compliance, which may be easier
to monitor for vitamin K antagonists than for the target-specific agents.
Pharmacokinetics](https://image.slidesharecdn.com/teachinganticoagulantsinccuward-161030105130/85/Treatment-with-anticoagulation-40-320.jpg)