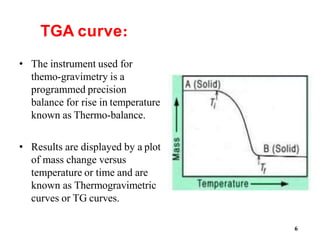
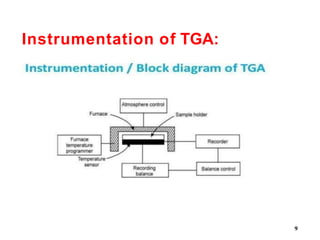
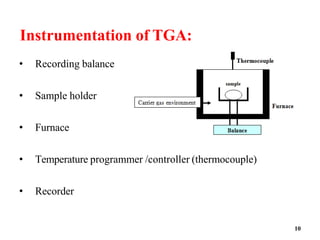
Thermal Gravimetric Analysis (TGA) is a technique that measures the change in mass of a sample as it is heated. It involves heating a sample in a controlled environment and measuring its mass loss over time or temperature. TGA can be used to determine purity, composition, thermal stability, and kinetics of reactions by producing a thermogravimetric curve that plots mass change against temperature or time.