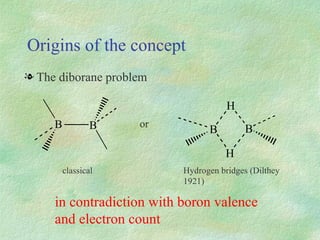
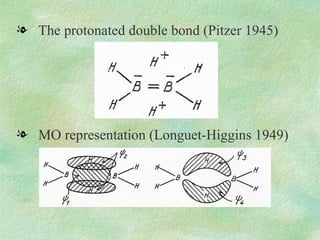
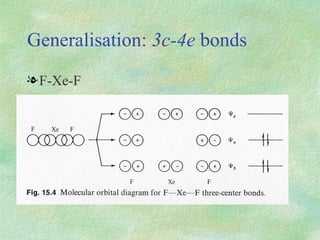
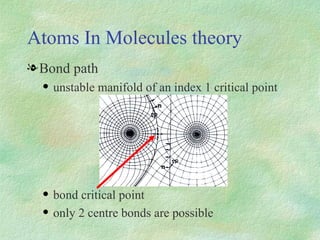
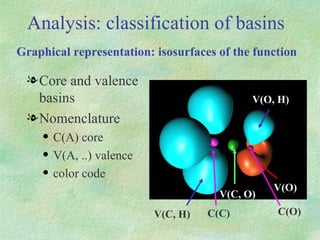
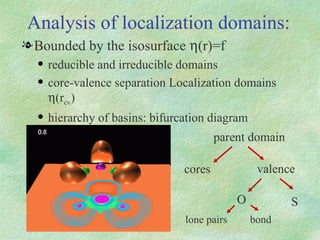
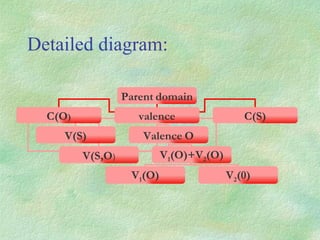
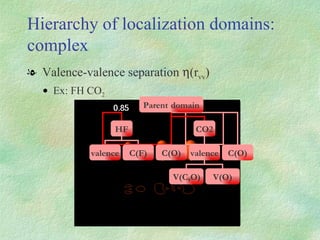
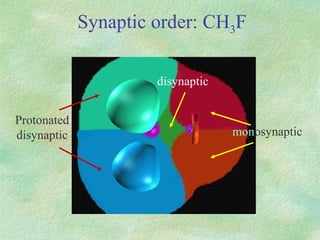
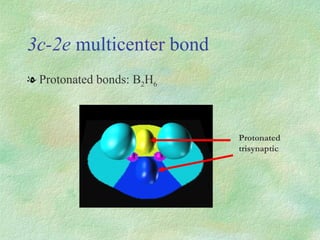
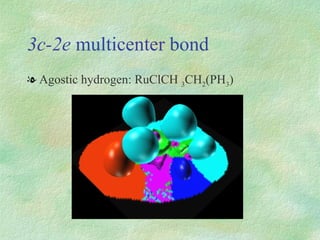
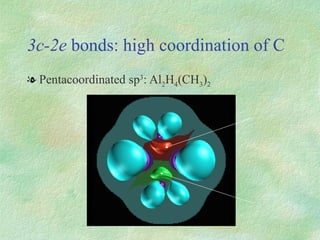
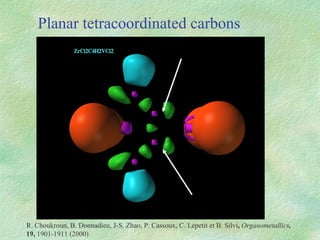
The document discusses the concept of multicenter bonding from the perspective of topological theories of bonding. It introduces the electron localization function (ELF) which can be used to analyze localization domains and classify bonds. ELF analysis provides definitions of multicenter bonds like 3c-2e bonds seen in B2H6 and 3c-4e bonds in hypervalent molecules like XeF2. The synaptic order of a localization basin, defined as the number of cores it shares a boundary with, is proposed as a descriptor of multicenter bonds that is complementary to the valence concept. While ELF provides a way to rigorously analyze bonding, the choice of localization function remains an open issue.