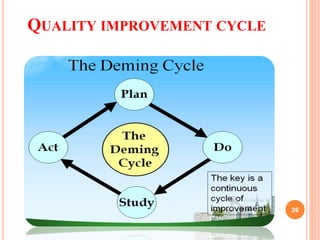
This document summarizes a seminar on statistical process control. It discusses key topics like process capability, estimating inherent capability from control charts, and Juran's 10 steps for quality improvement. Process capability refers to a process's ability to achieve results within specifications. It is quantified using indices like Cp and Cpk which measure potential and performance based on process data. Assumptions like statistical control are important for accurately applying these indices. Quality improvement involves building awareness, setting goals, training, implementing projects, and maintaining momentum through regular systems.