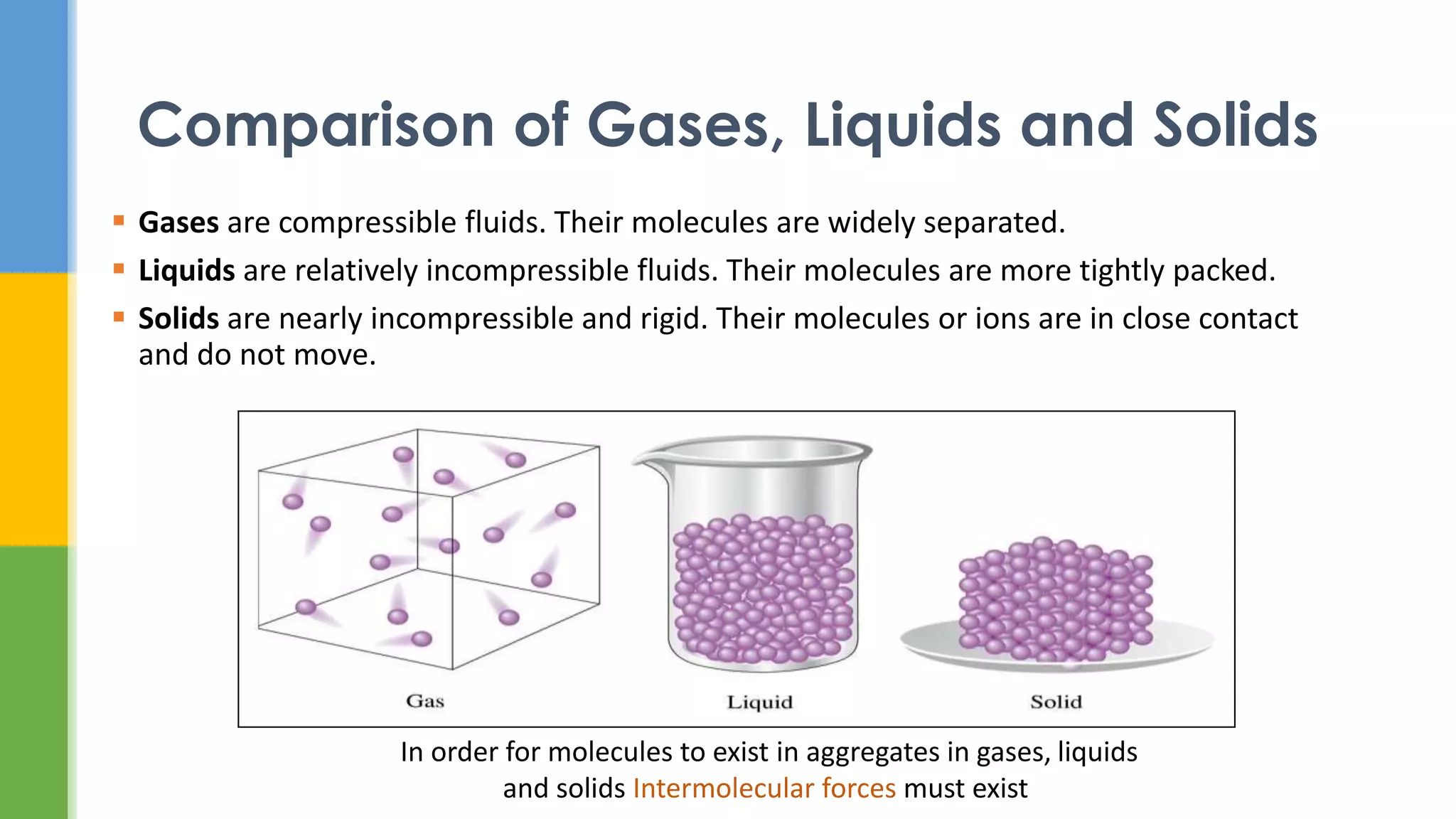
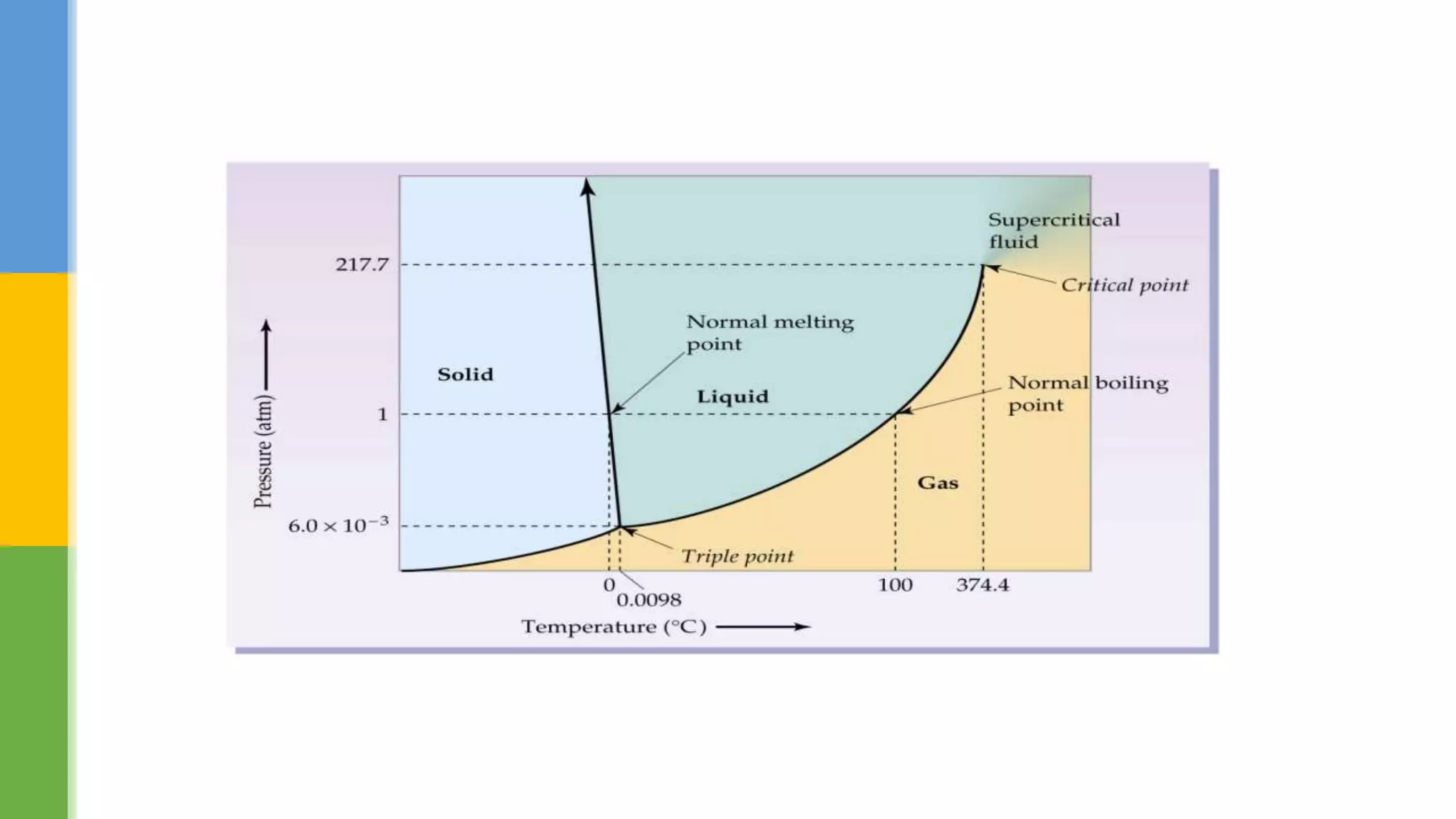
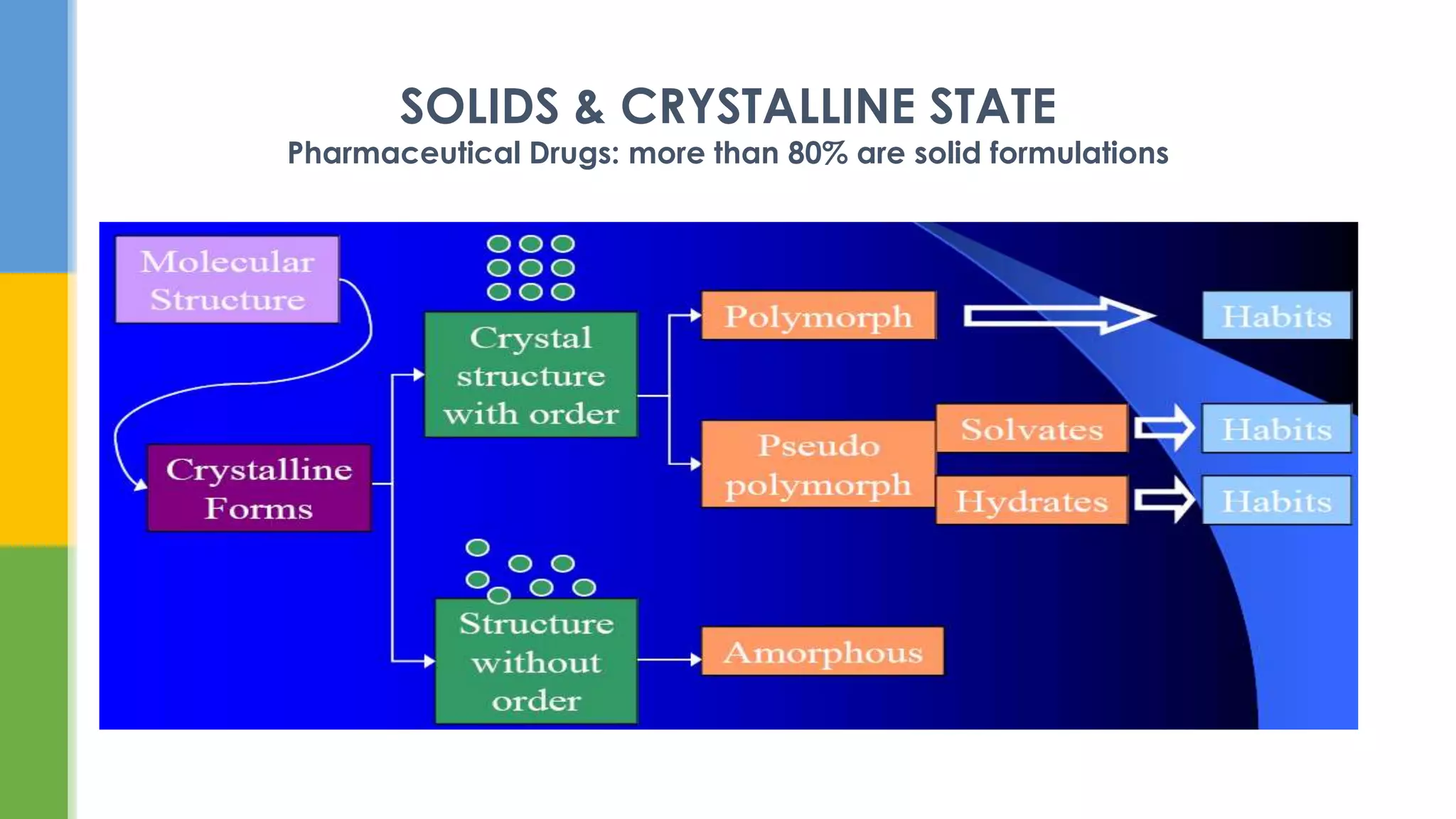
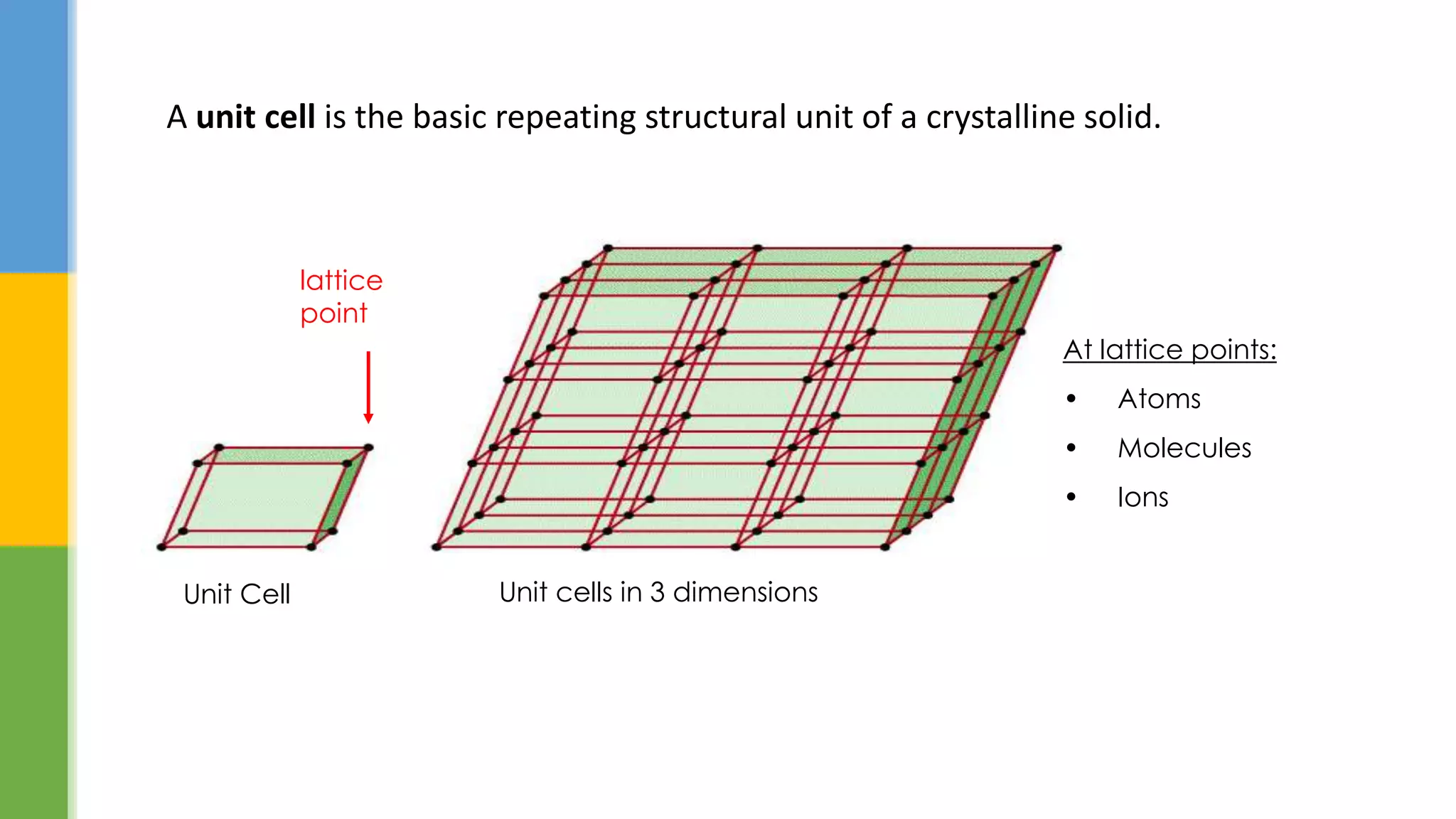
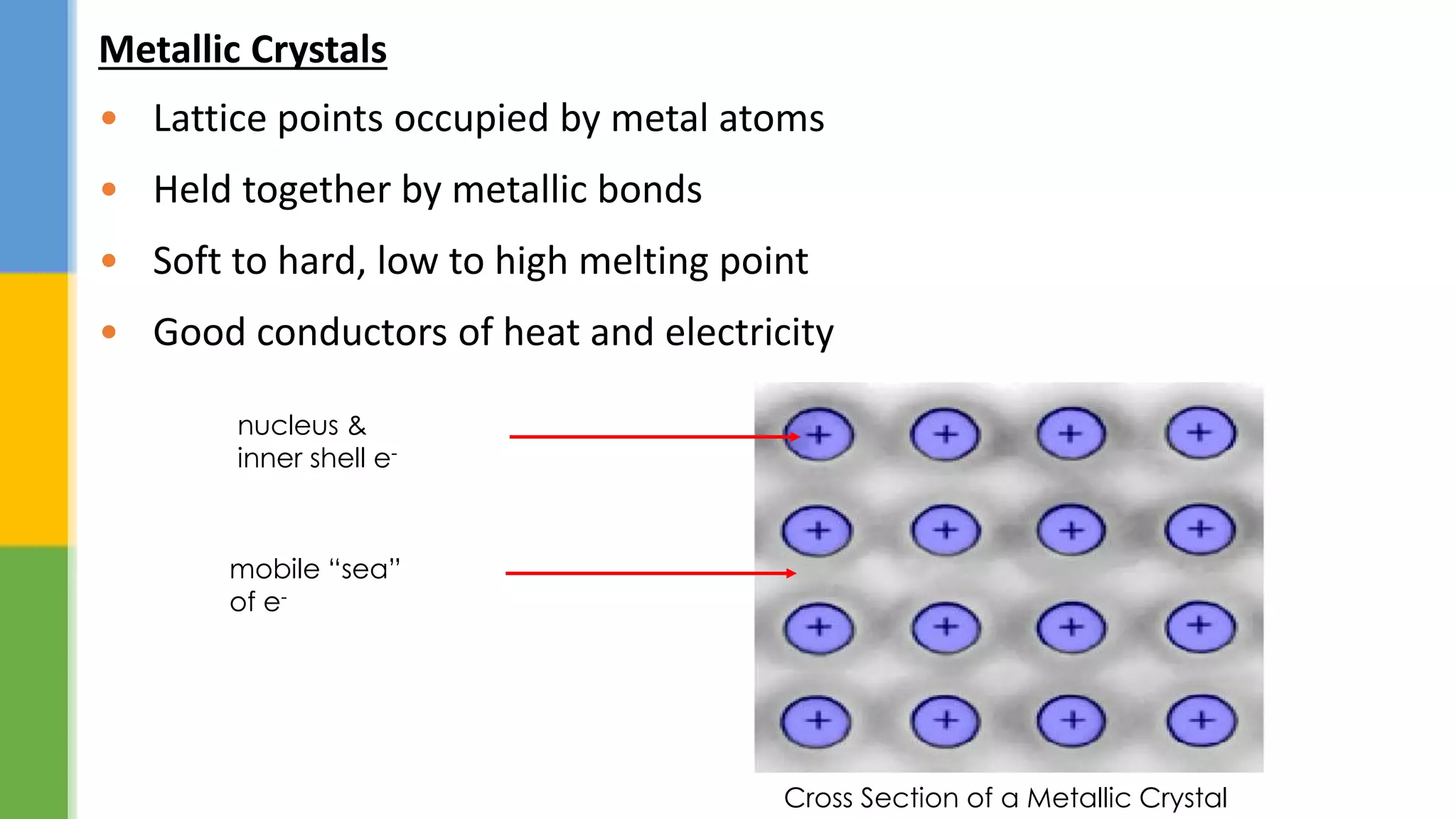
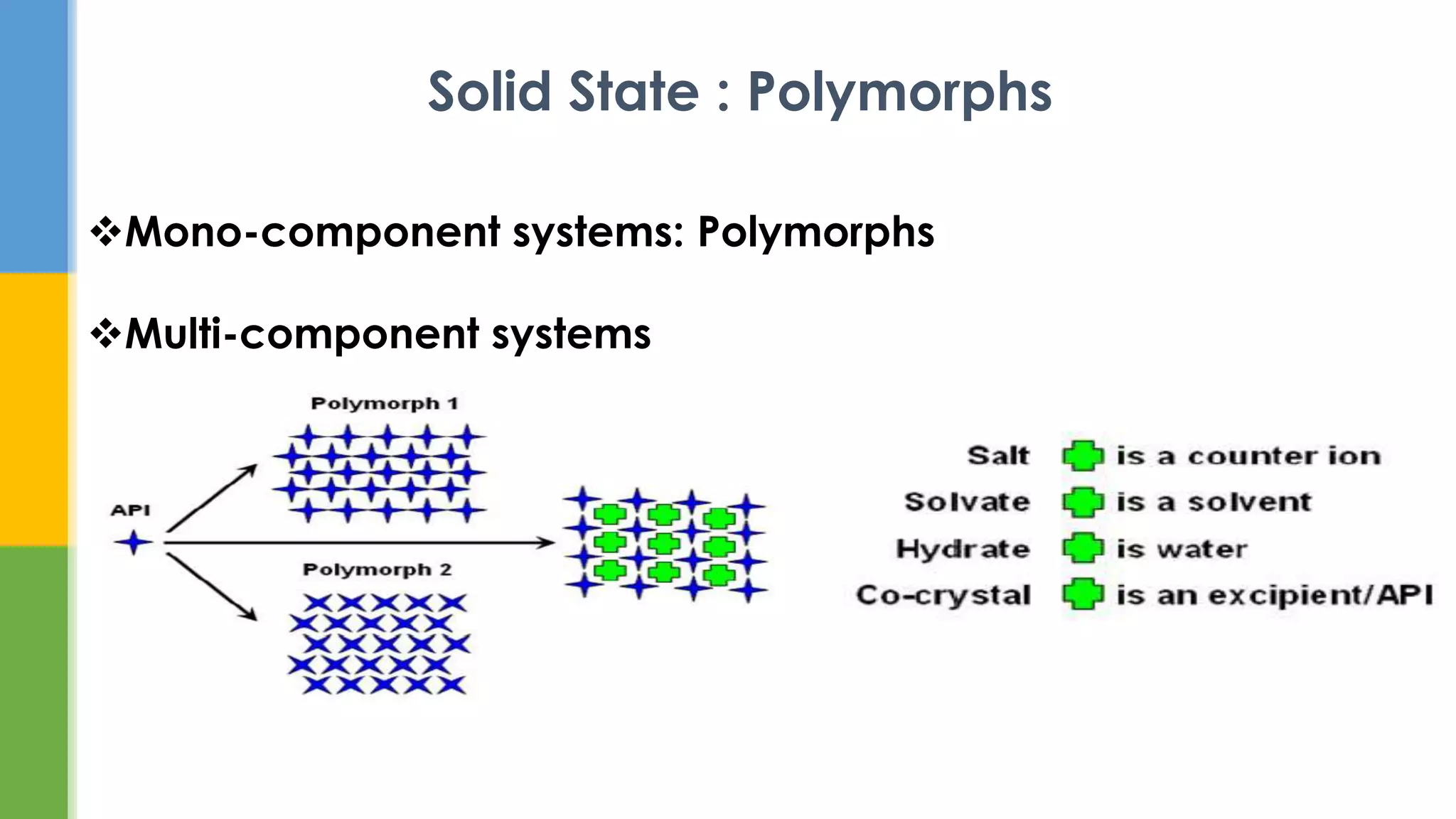
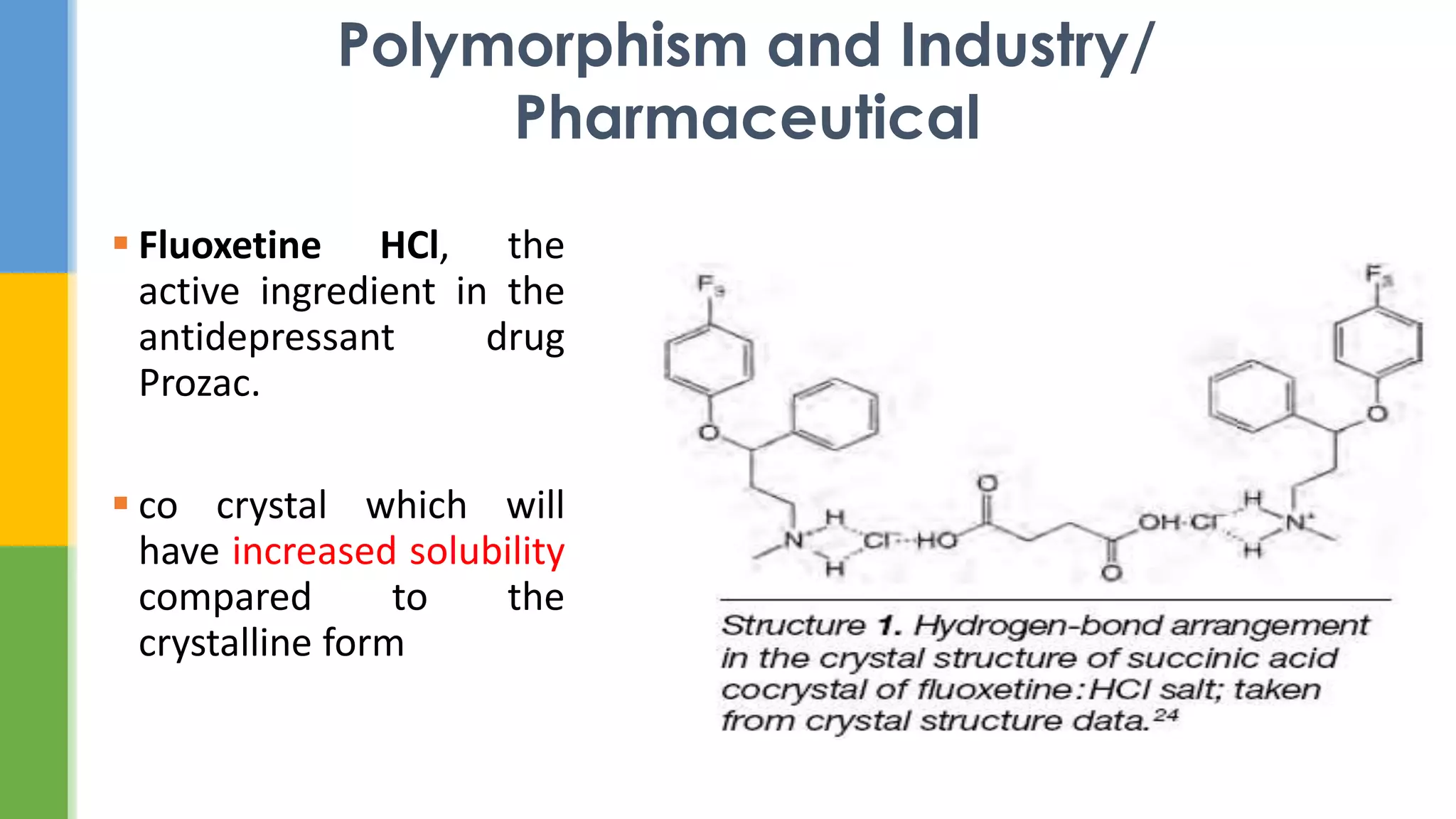
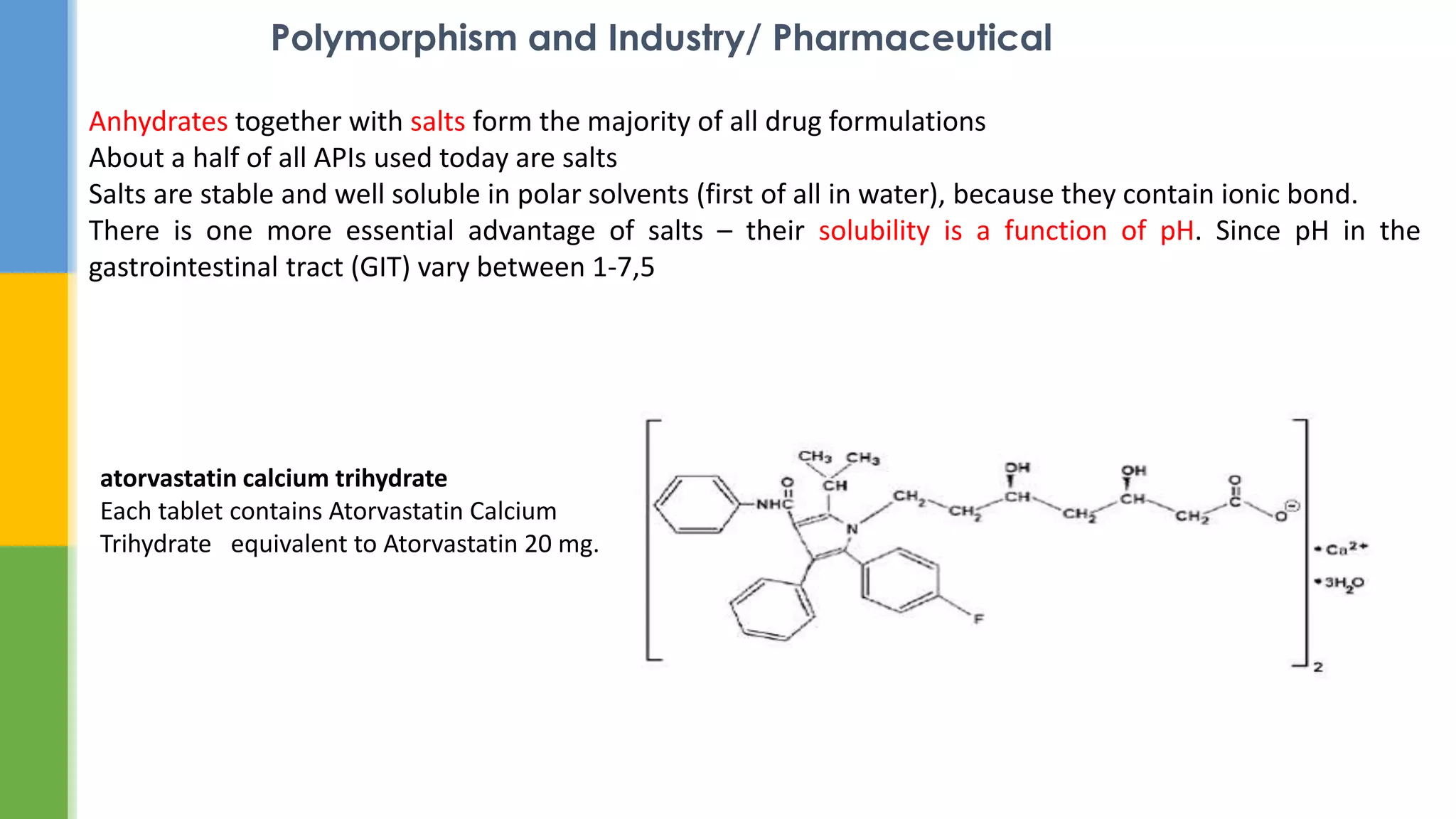
This document provides an overview of states of matter and polymorphism. It discusses the three main states of matter - gases, liquids, and solids - and how their molecular arrangements differ. Solids can exist in crystalline or amorphous forms, with crystalline solids possessing long-range molecular order. Polymorphism, where a substance can exist in multiple crystal structures, is described. The importance of polymorphism in pharmaceutical industry is highlighted, as different solid forms can impact properties like solubility, dissolution rate, and bioavailability. Specific drug examples like carbamazepine and ritonavir and their polymorphic forms are mentioned.