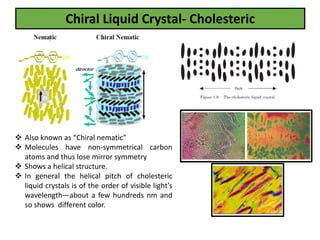
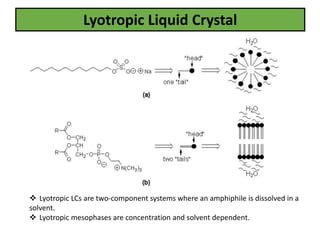
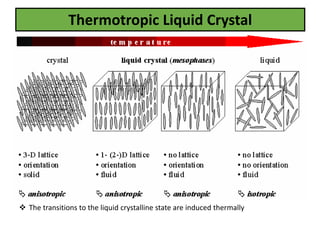
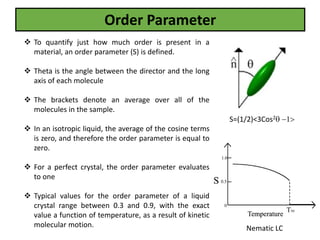
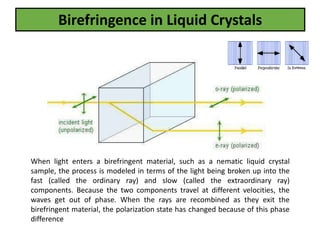
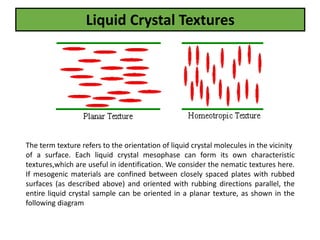
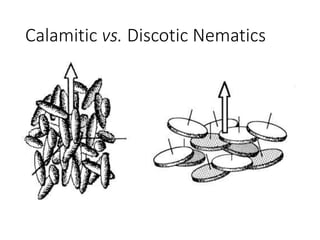
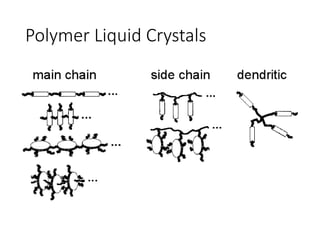
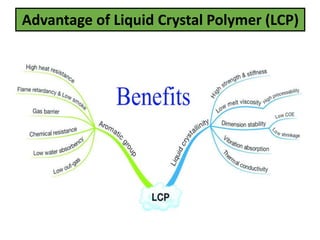
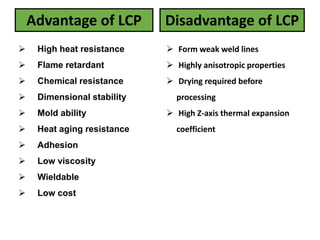
The document discusses liquid crystals and liquid crystal polymers. It notes that liquid crystals have properties between solids and liquids, with some positional and orientational order. They can exist in nematic, smectic, and cholesteric phases. Liquid crystal phases are important in biological systems like cell membranes and the brain. Liquid crystal polymers are highly resistant to heat and chemicals. They have applications in displays, body armor like Kevlar, and as heat sensors.






![Liquid Crystals
Thermotropic Lyotropic
High molecular
(molar) mass
[ polymers]
Low molecular
(molar) mass
Main-chain
polymers
Side-chain
polymers
Rod-like or
lath-like
molecules
Calamitic
Disc-like
molecules
Discotic
Single or multicomponent
systems
Homo- or co-polymers
Figure 9.1 The liquid crystal family tree.](https://image.slidesharecdn.com/lcp-150412153516-conversion-gate01/85/Liquid-Crystal-and-Liquid-Crystal-Polymer-7-320.jpg)