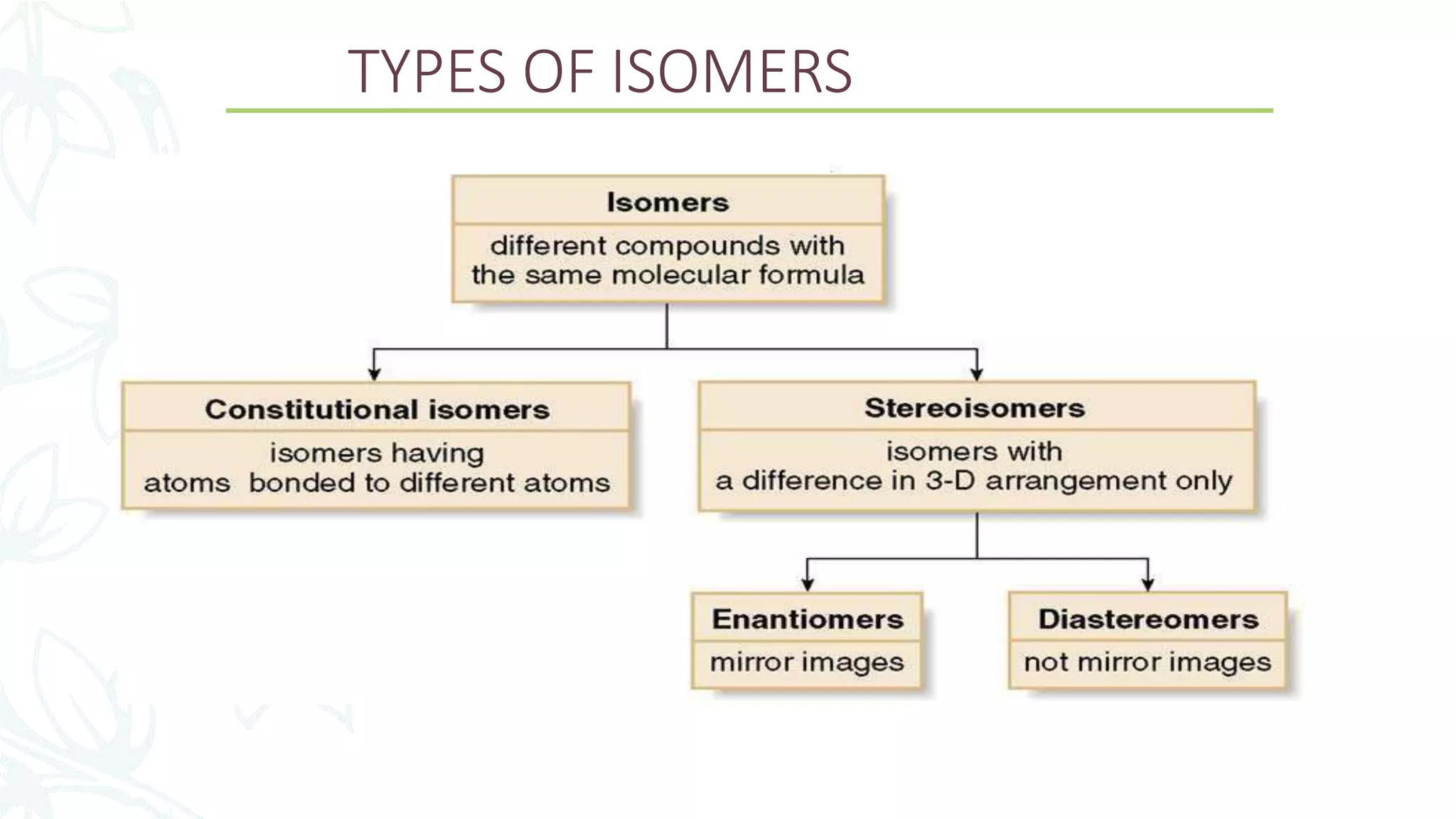
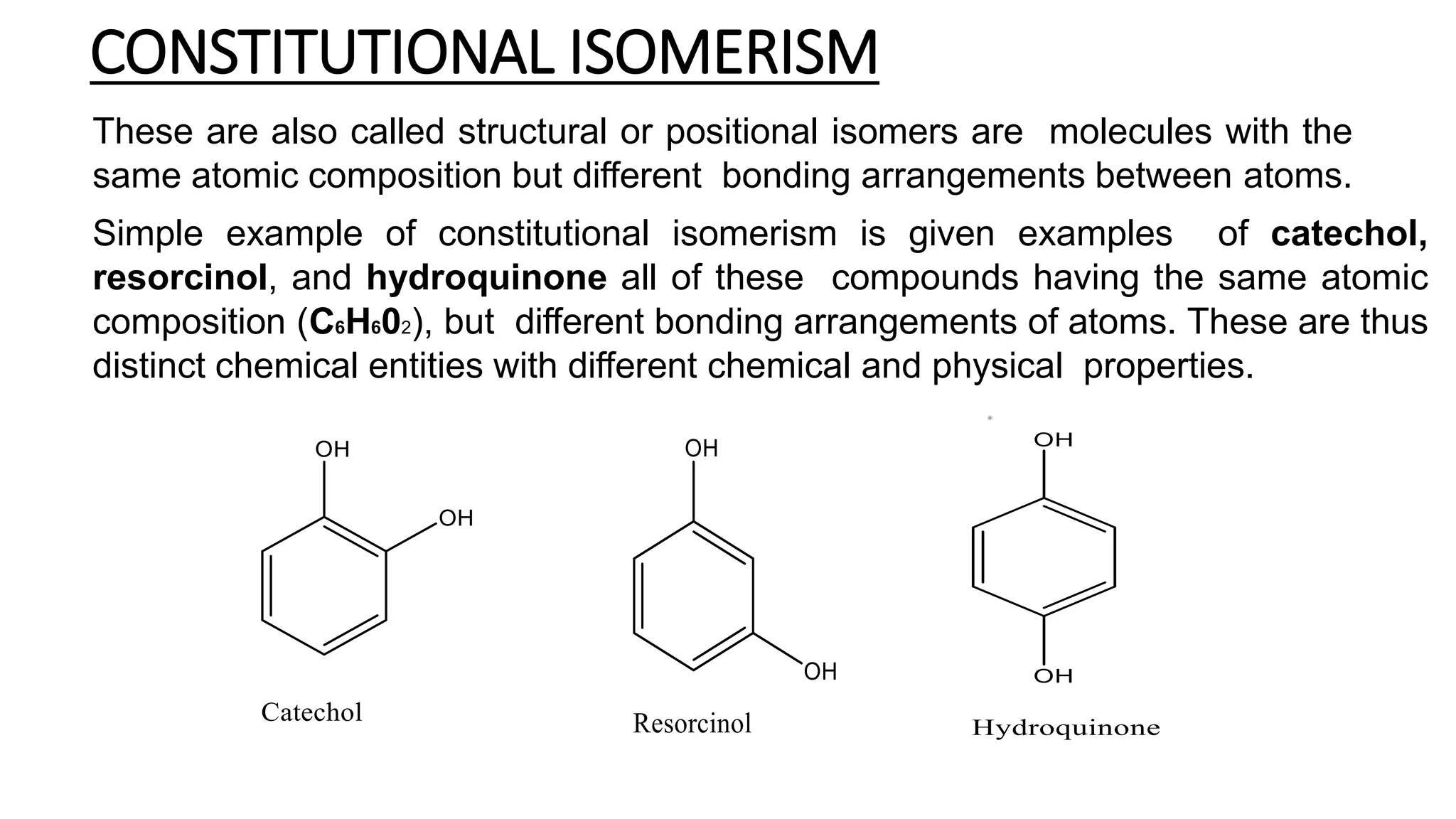
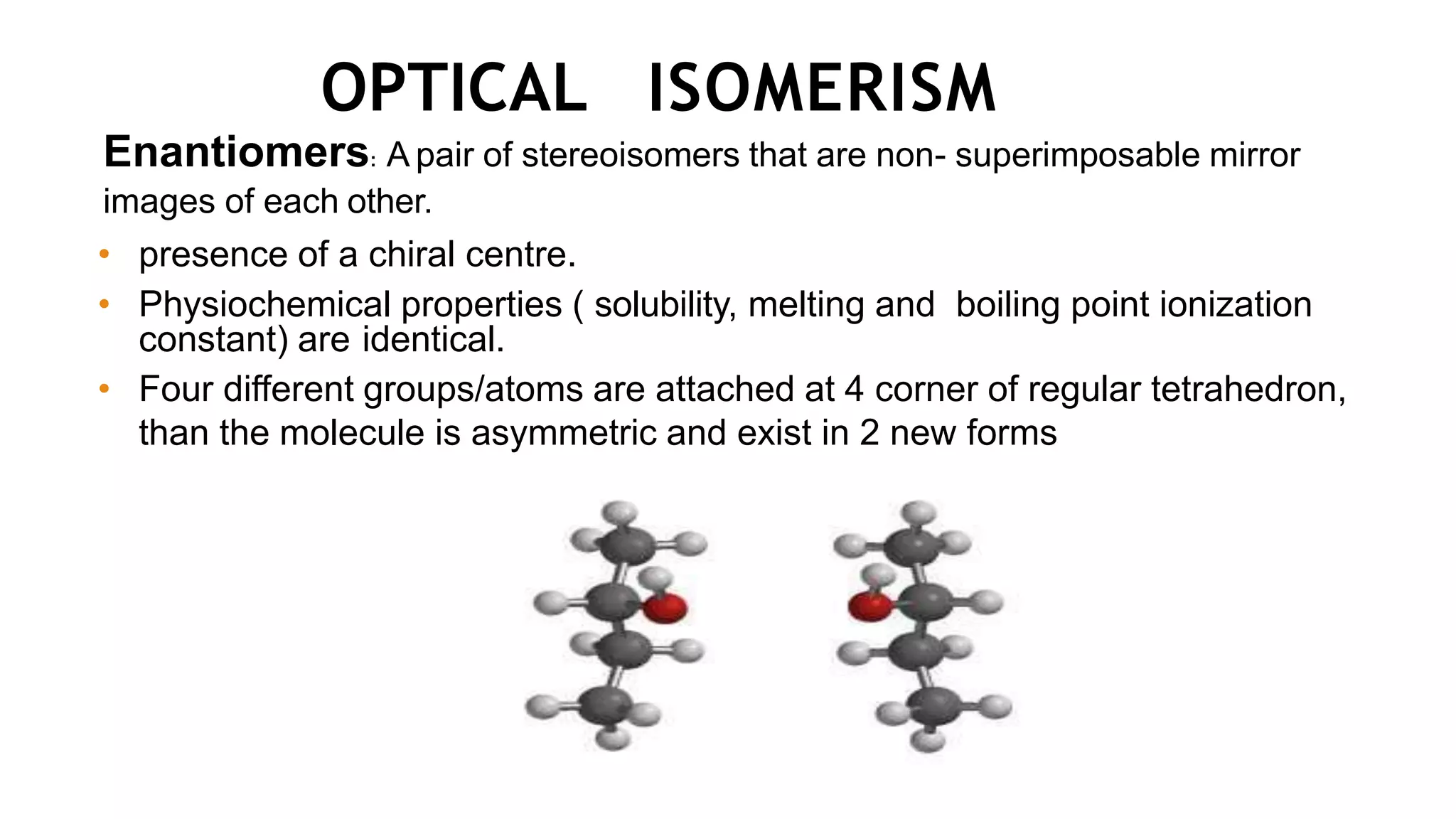
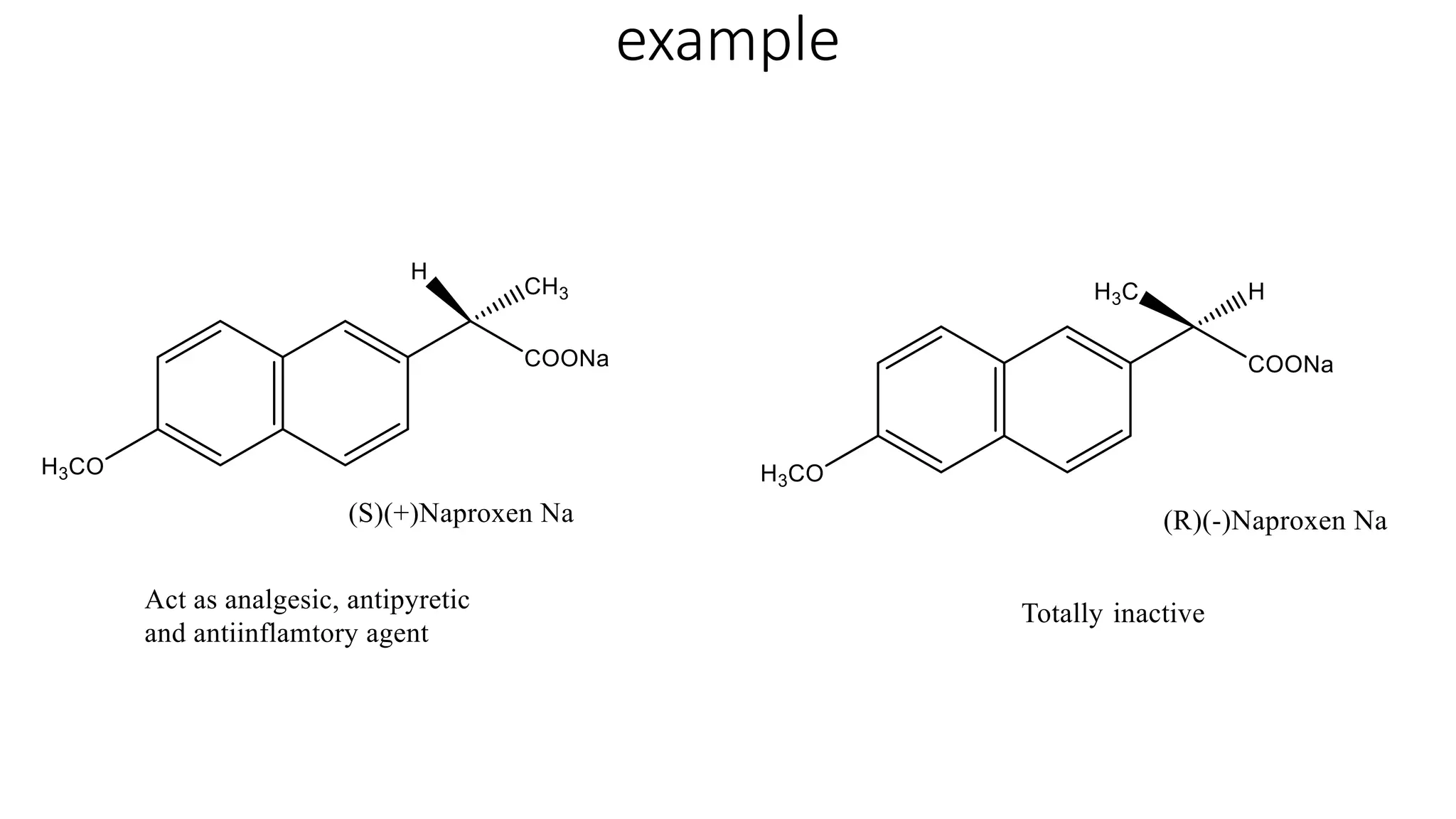
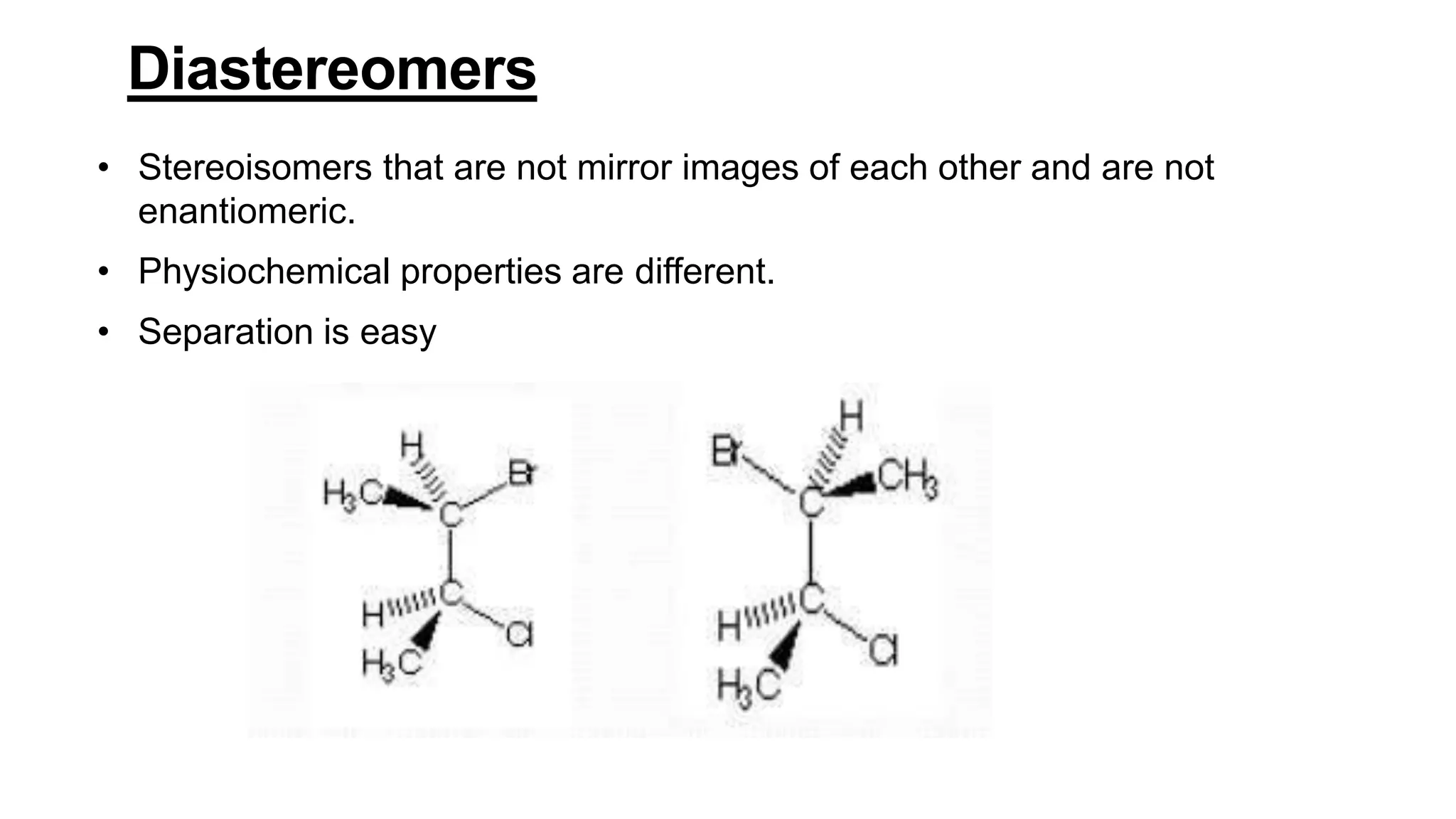
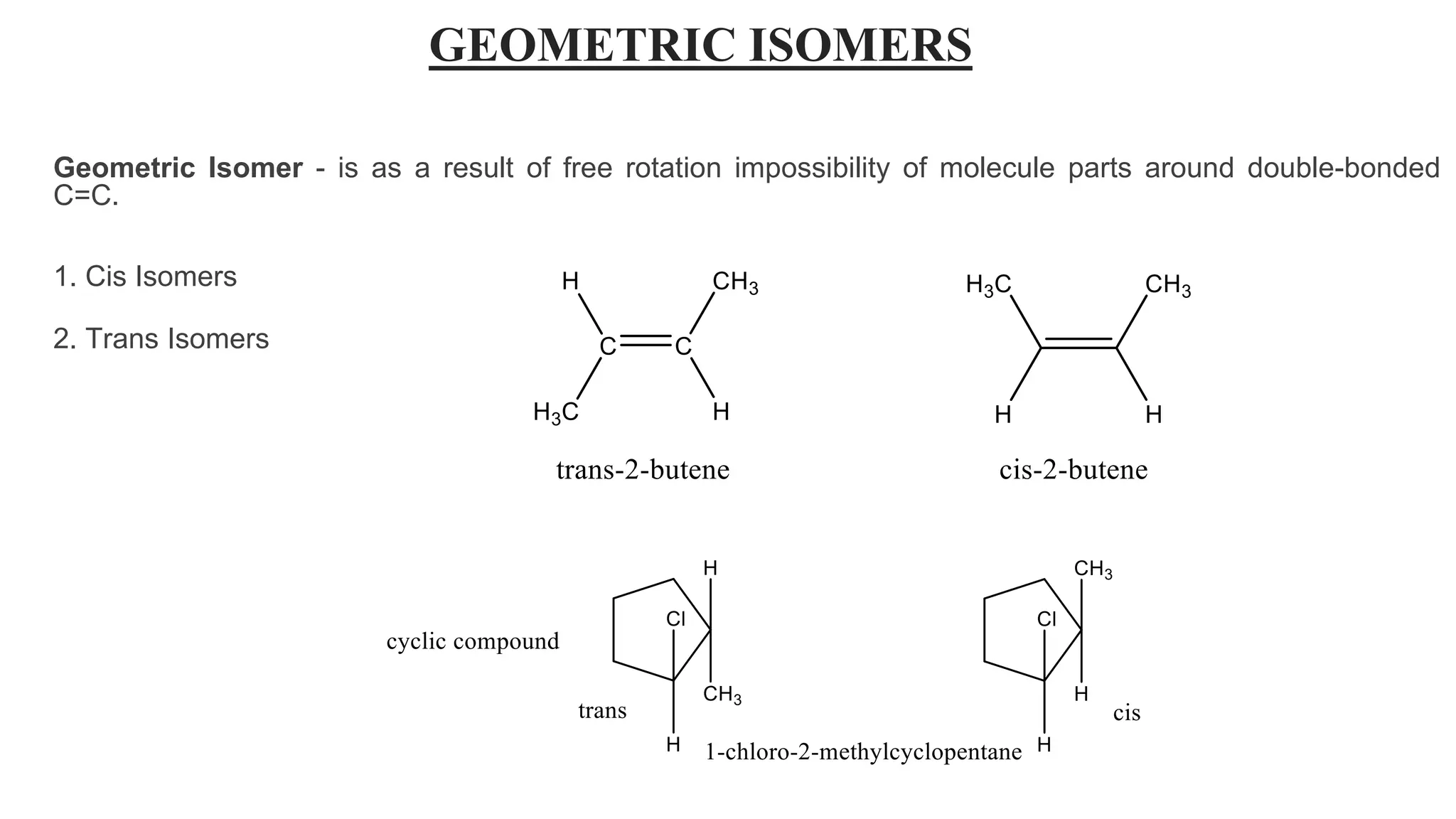
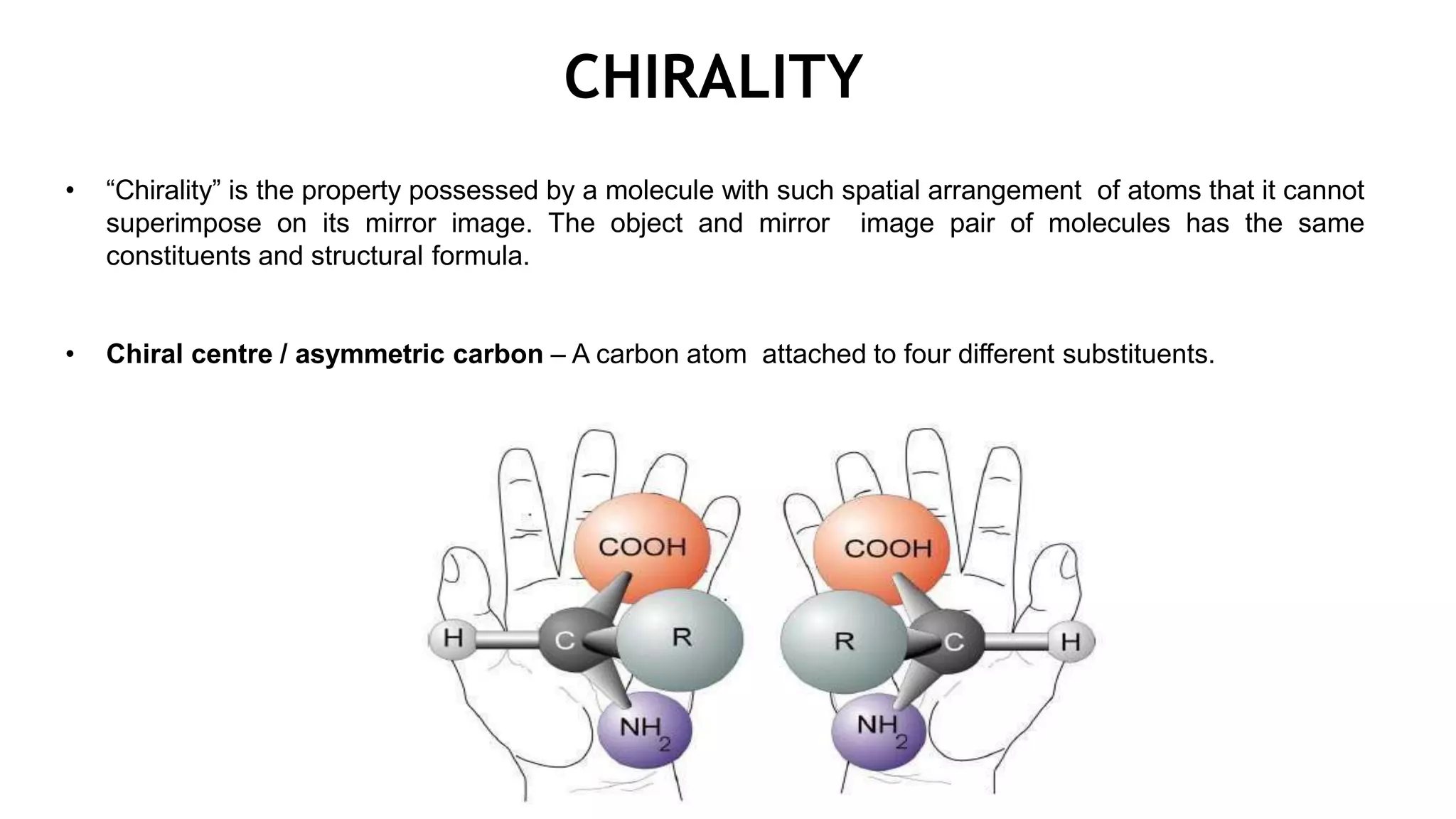
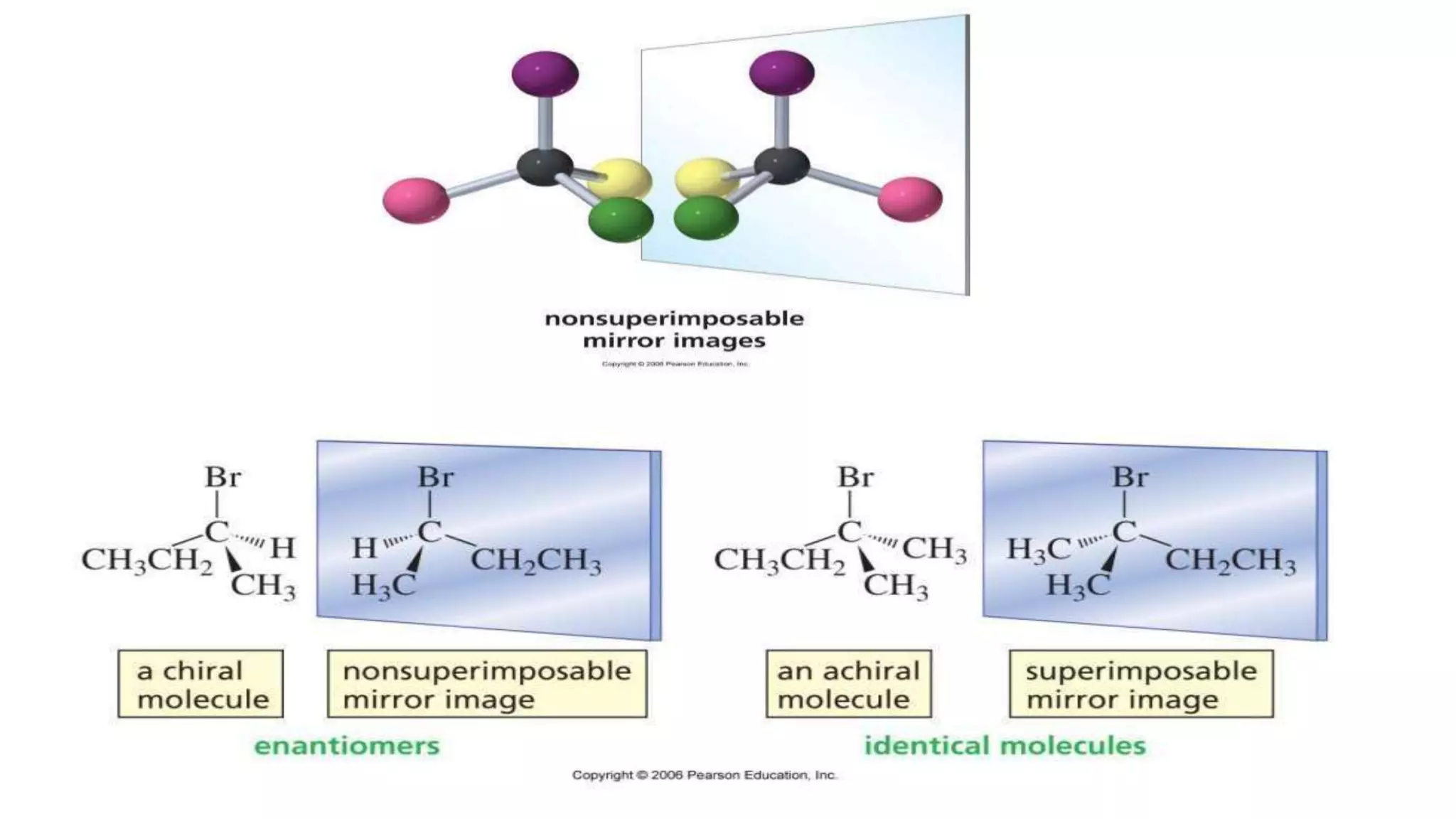
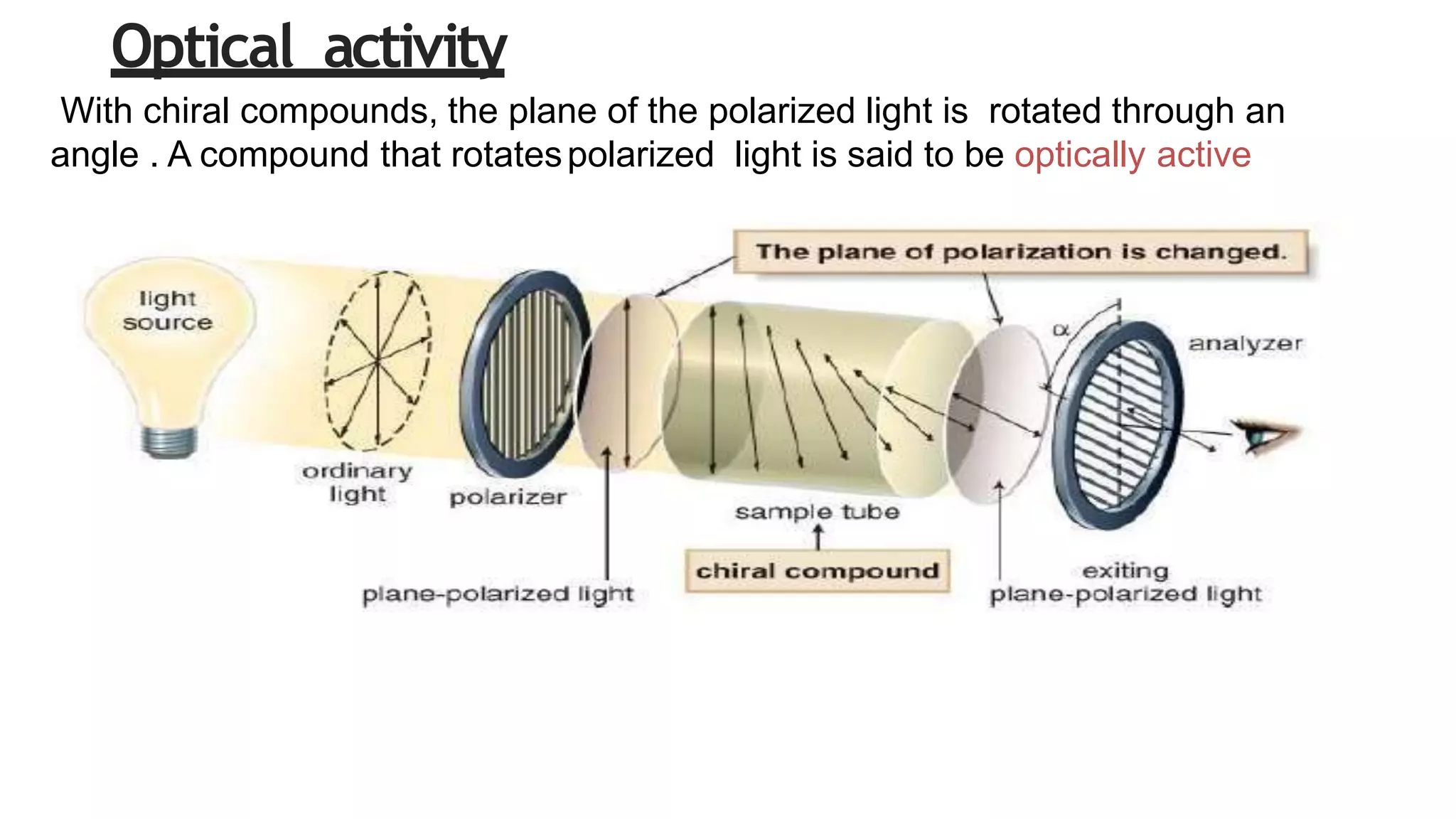
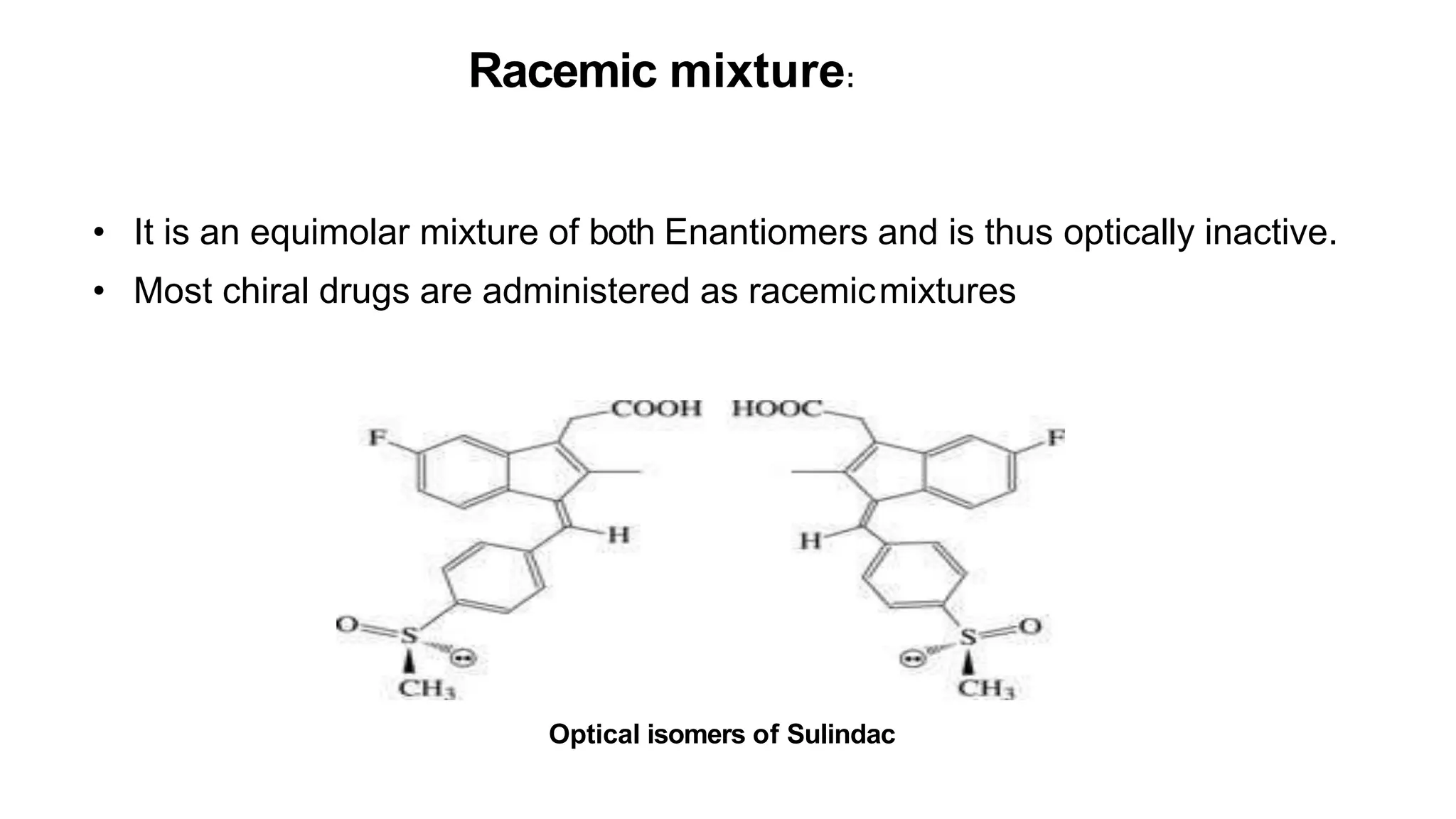
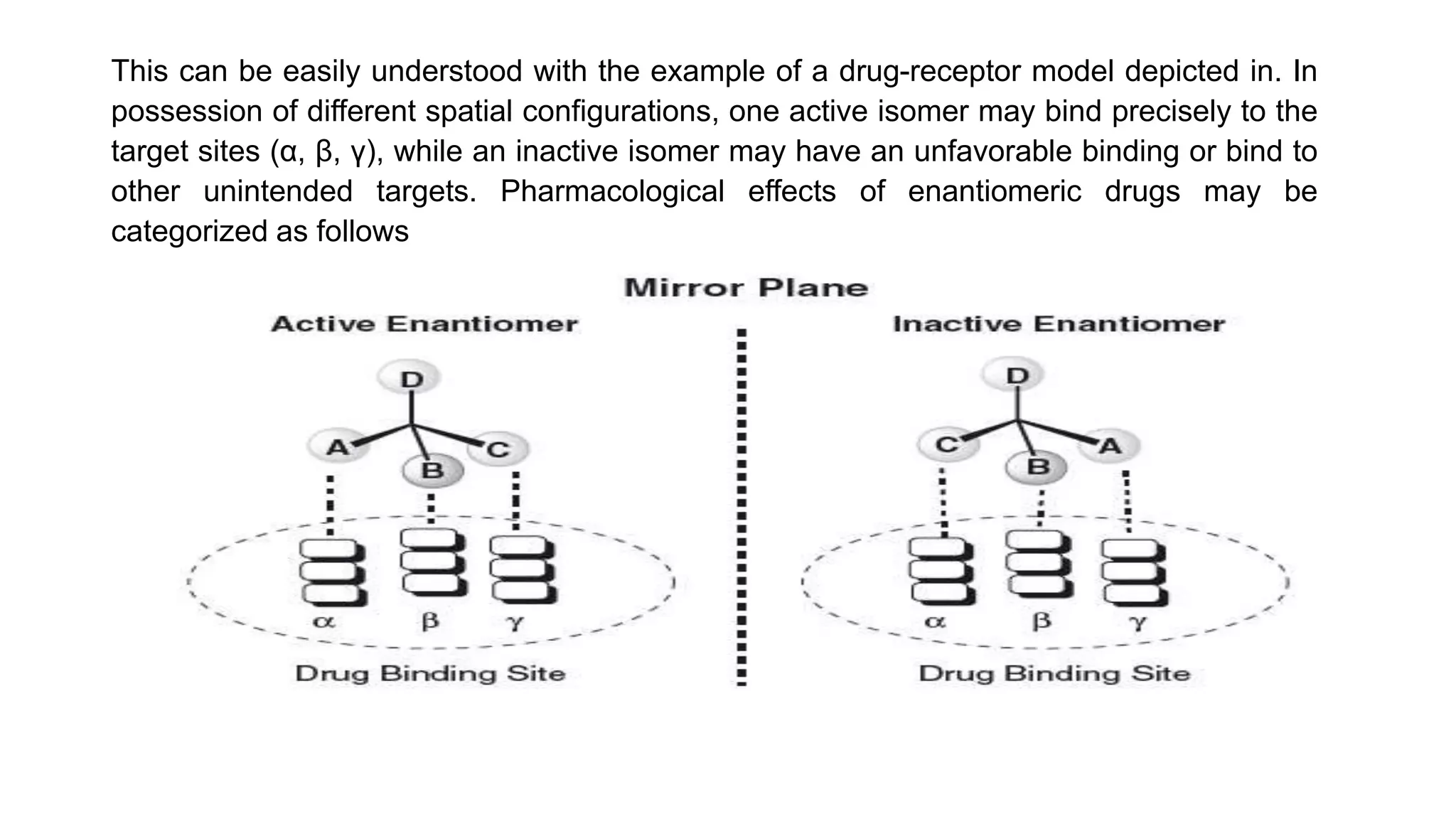
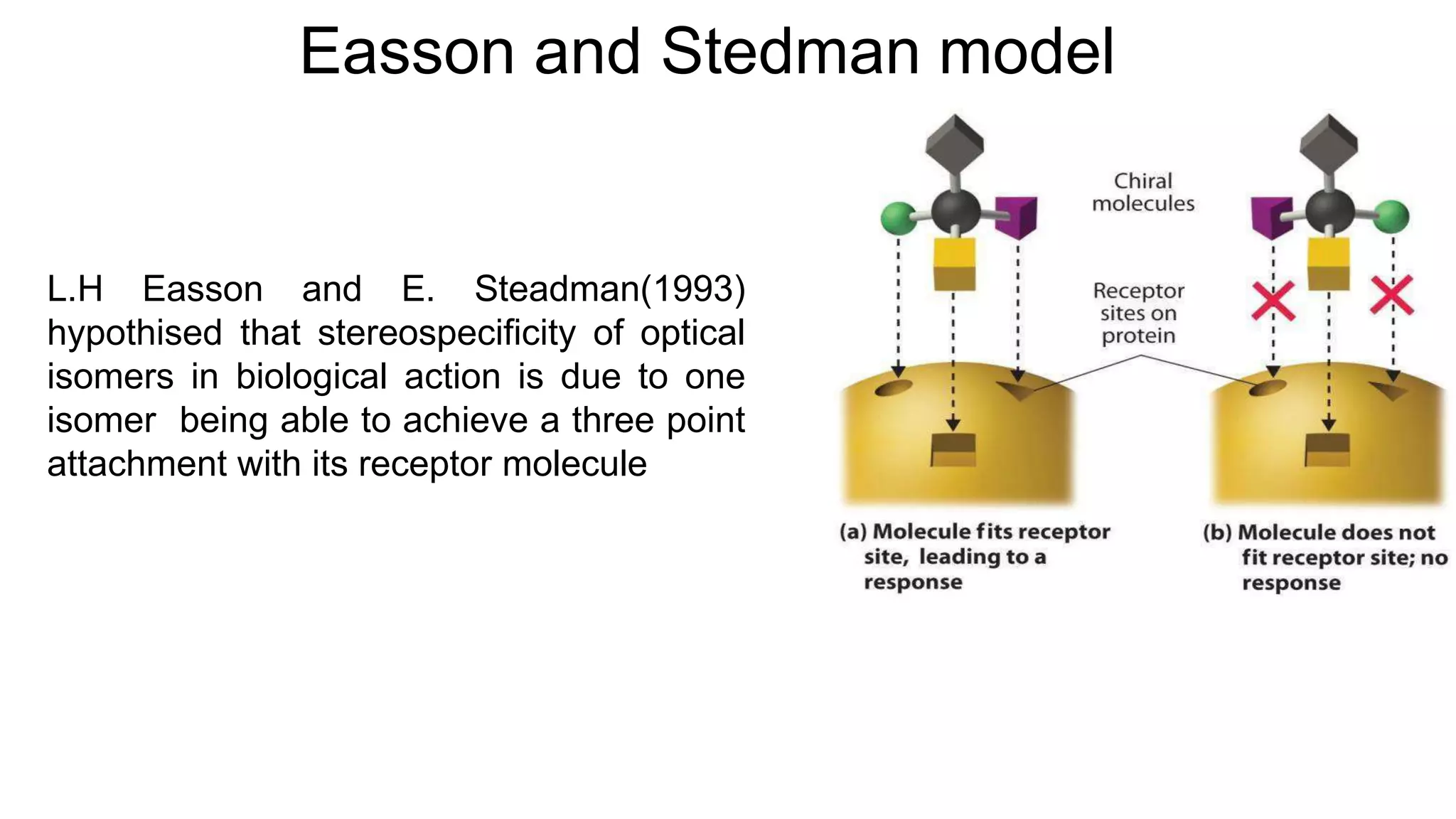
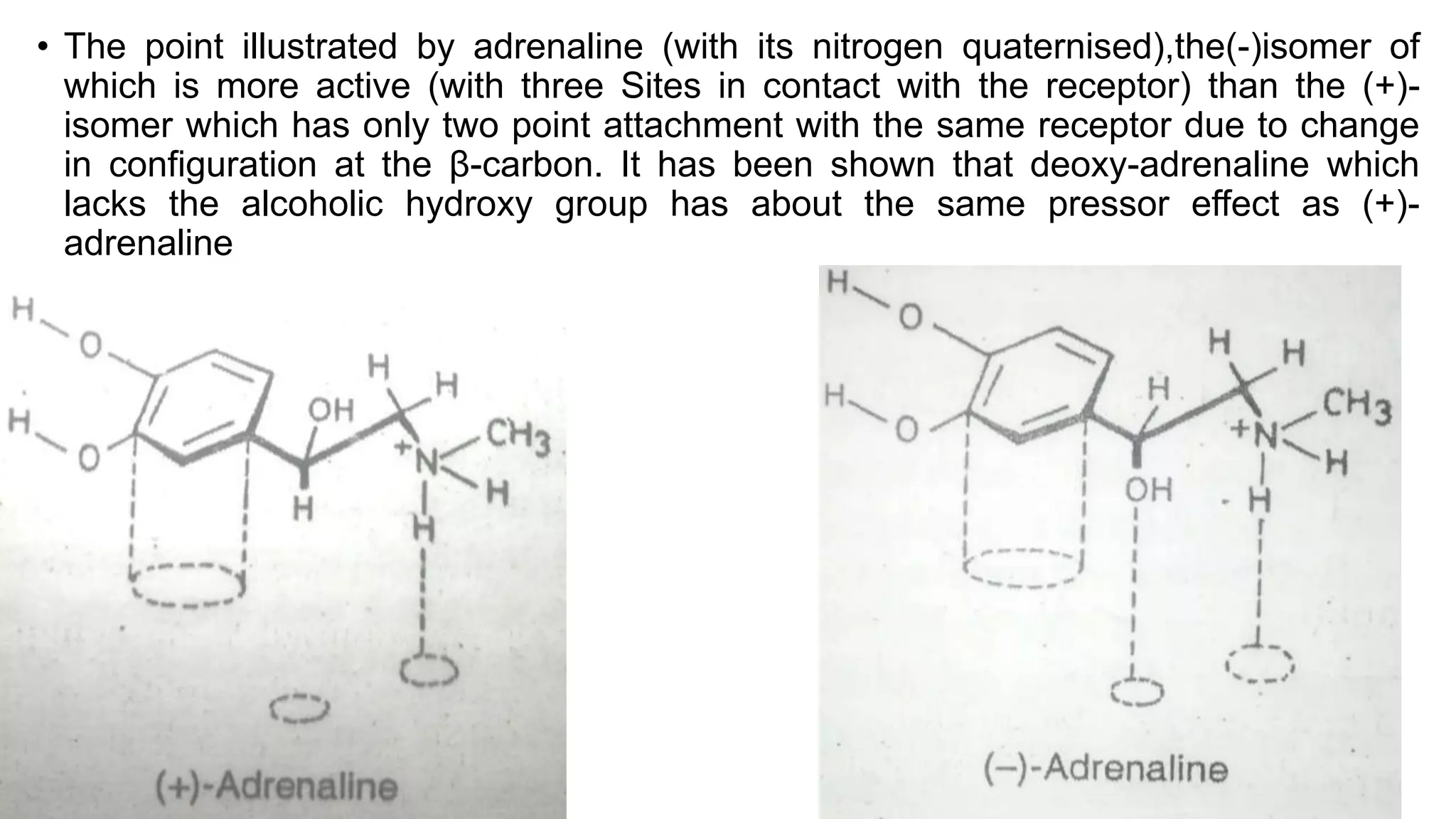
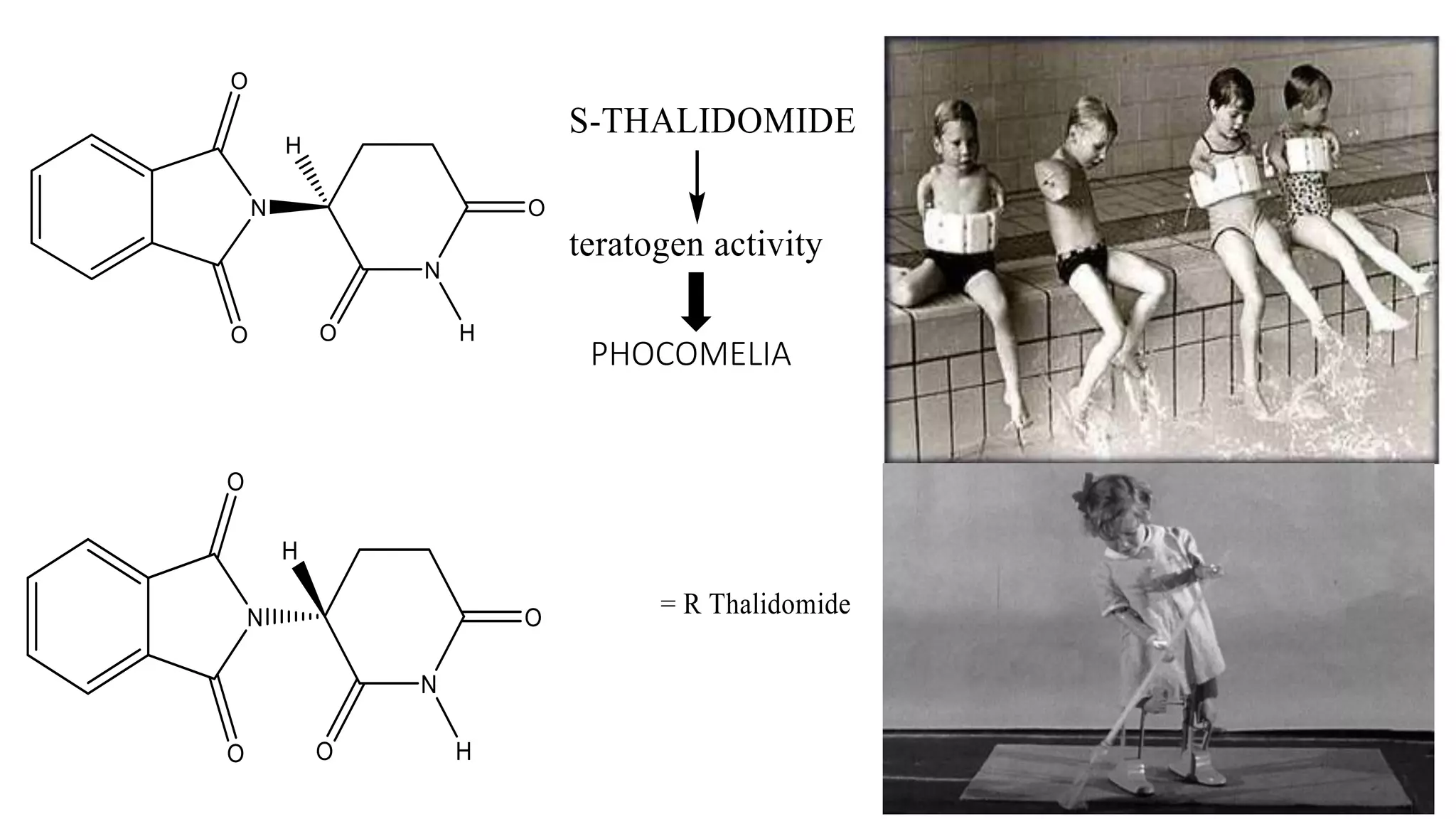
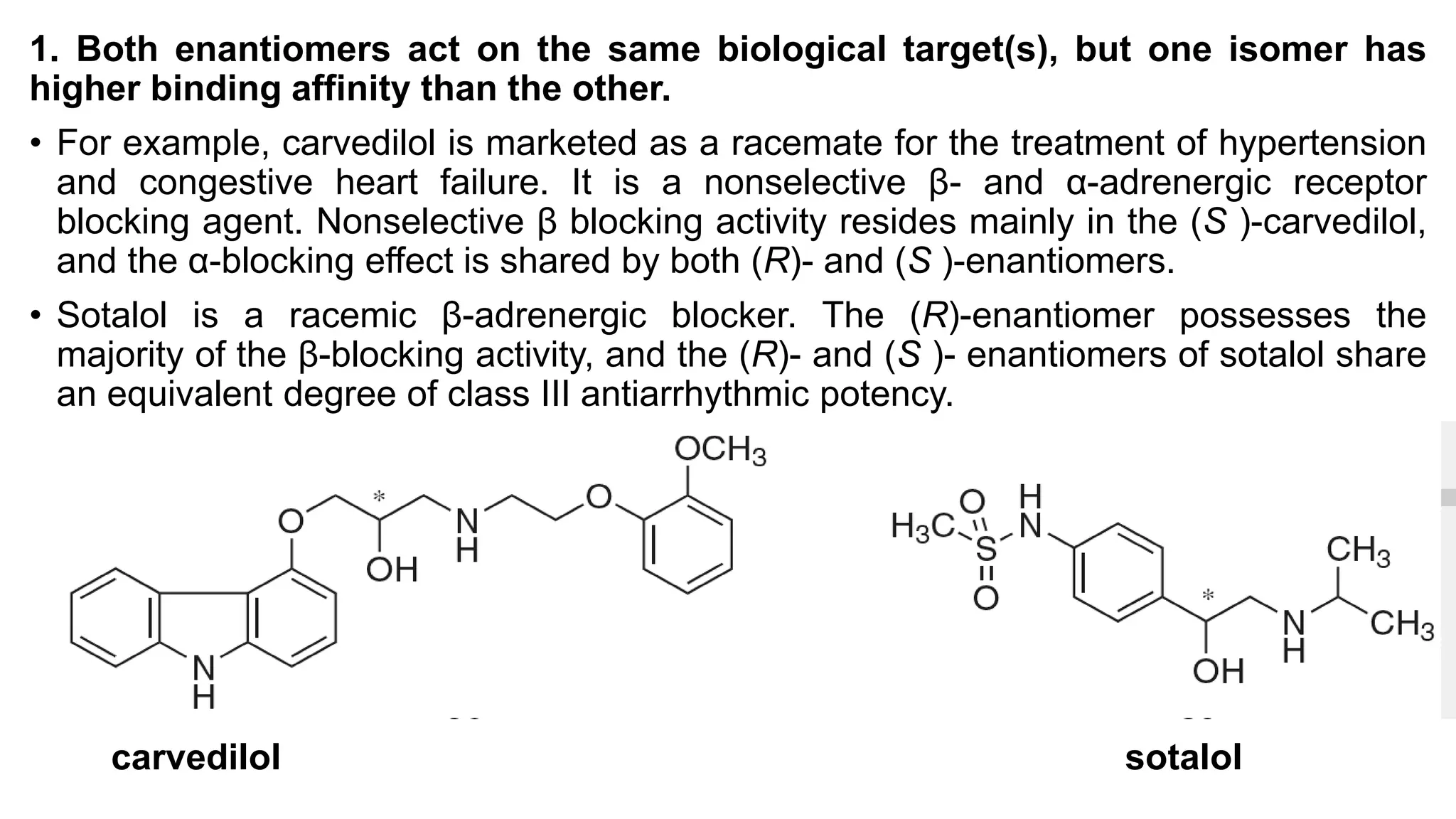
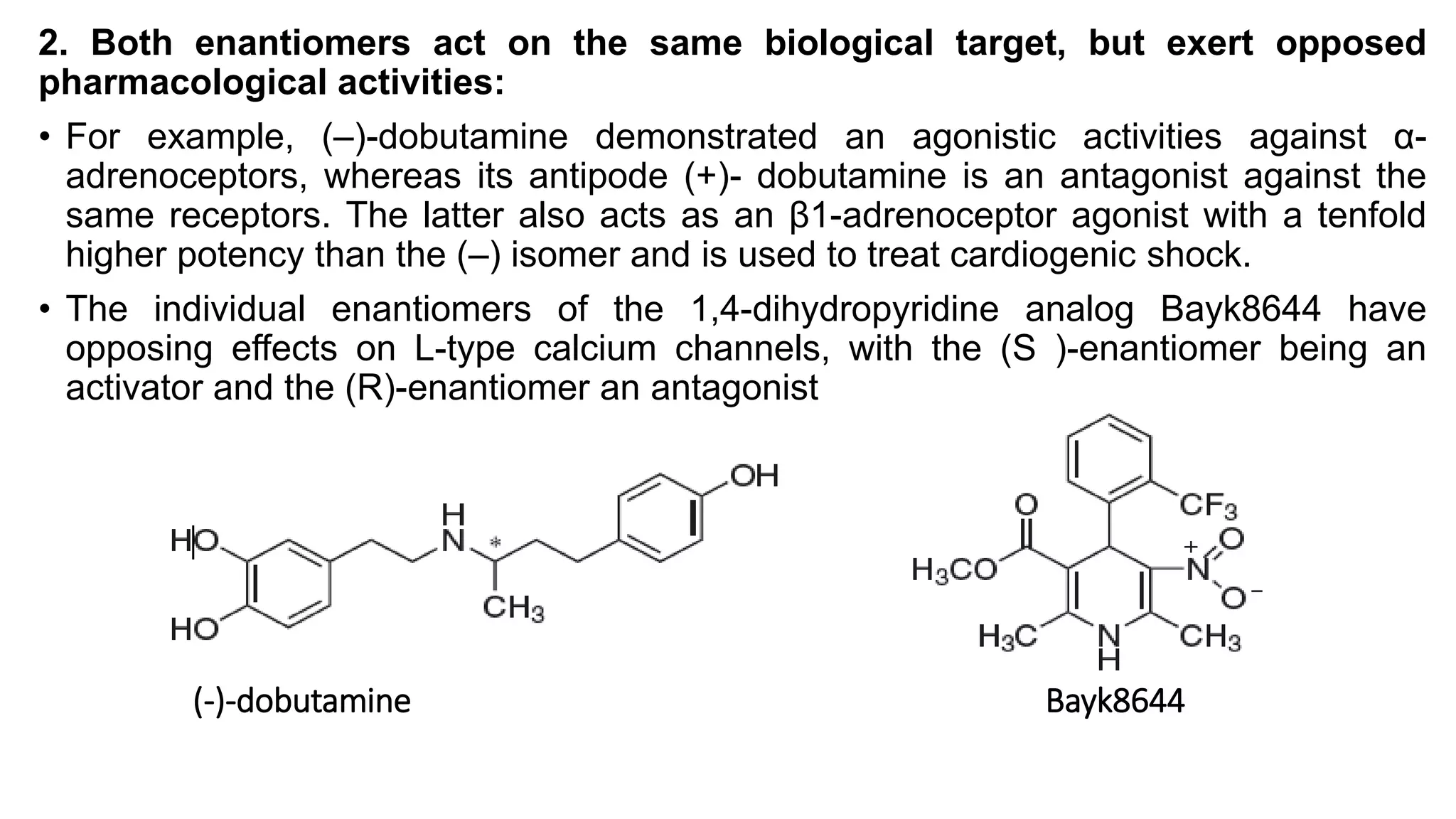
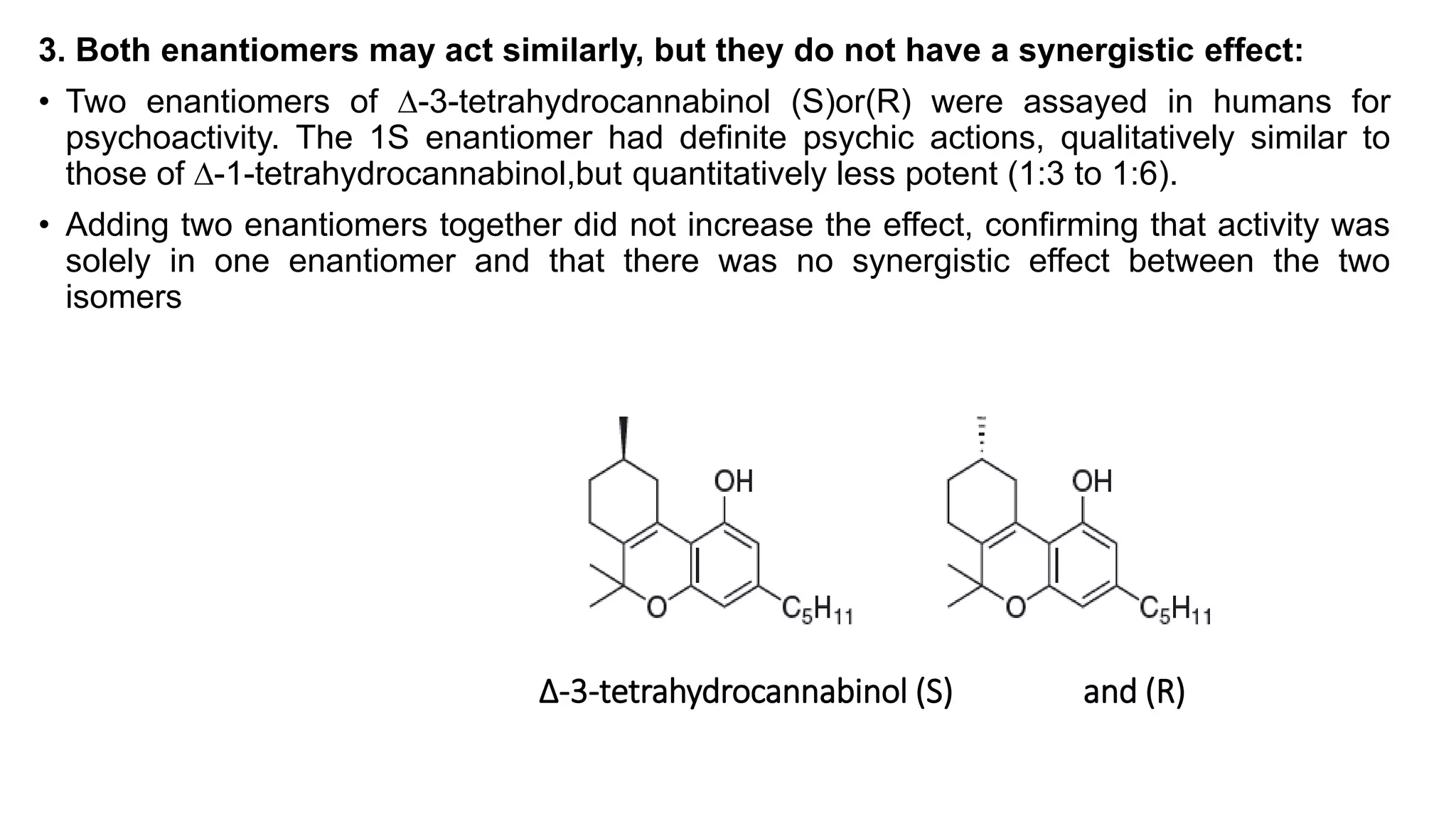
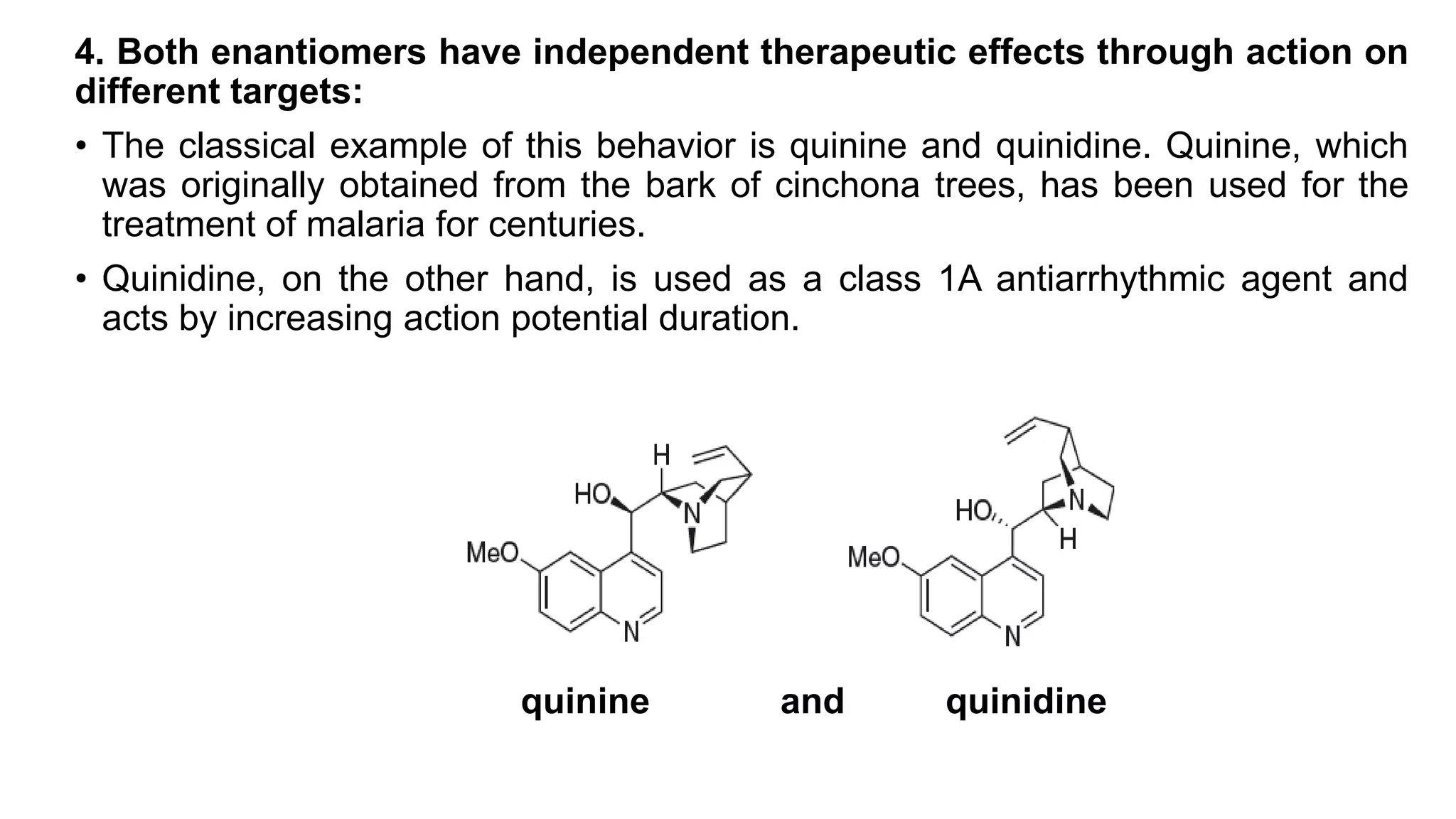
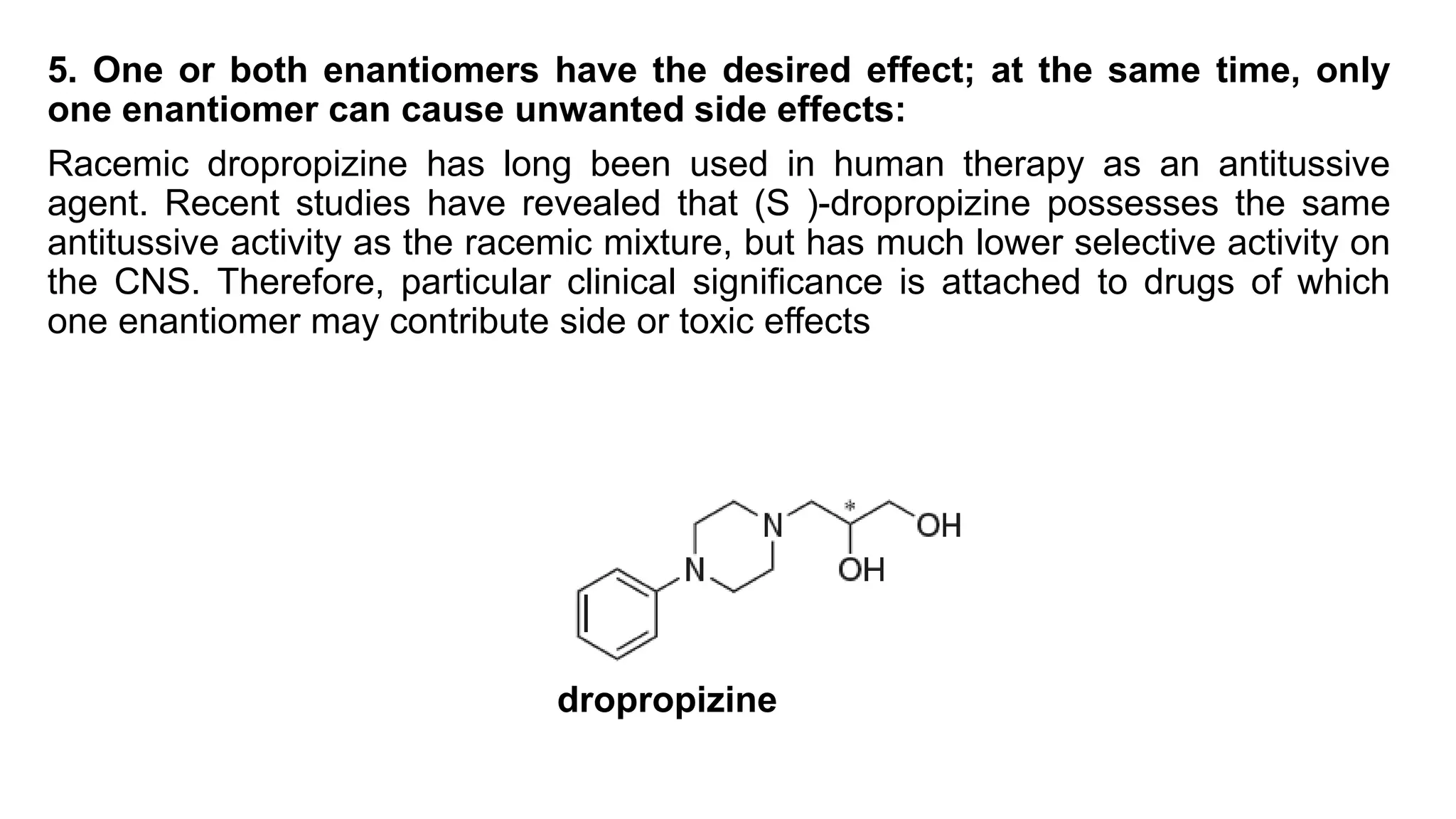
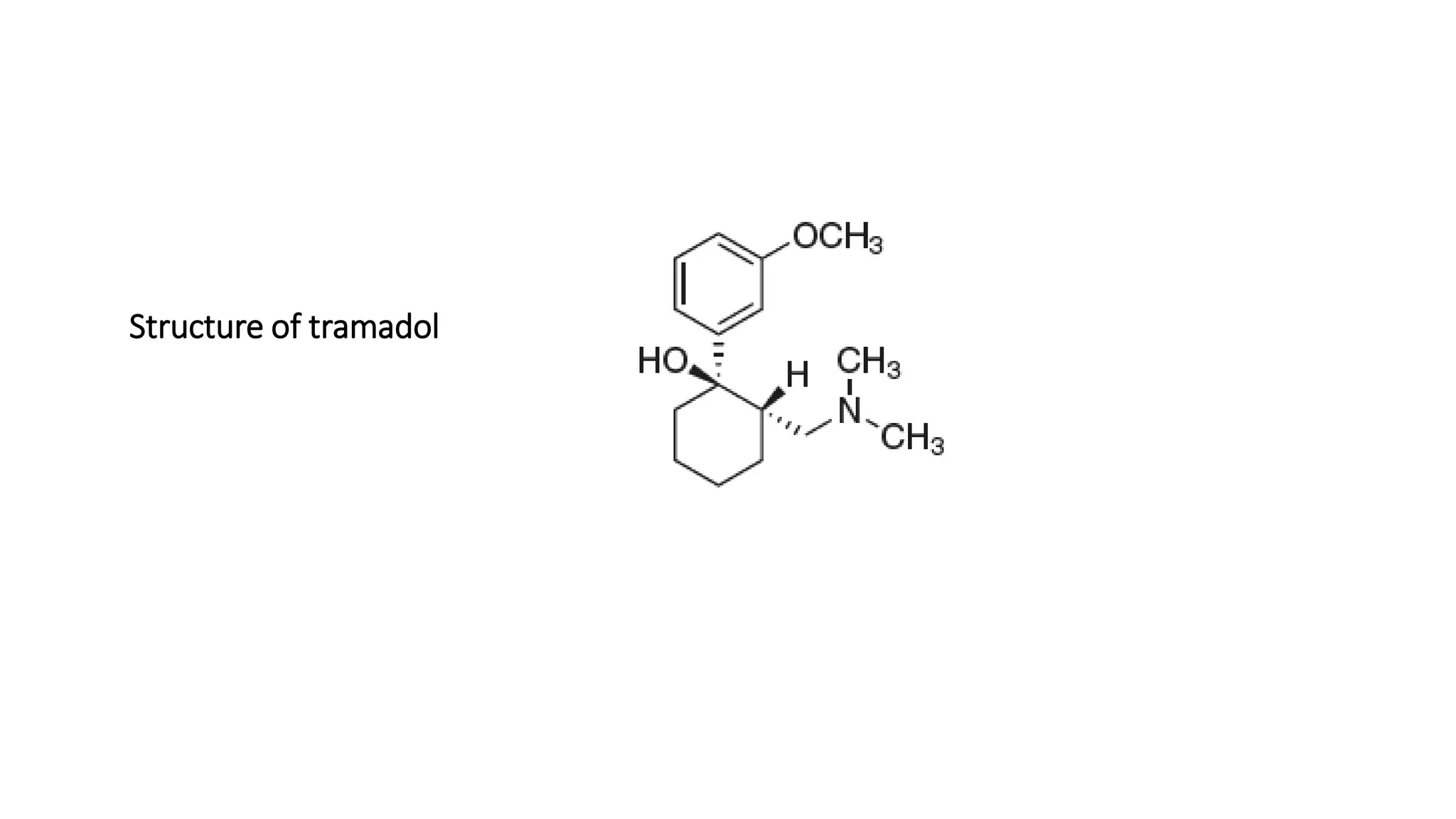
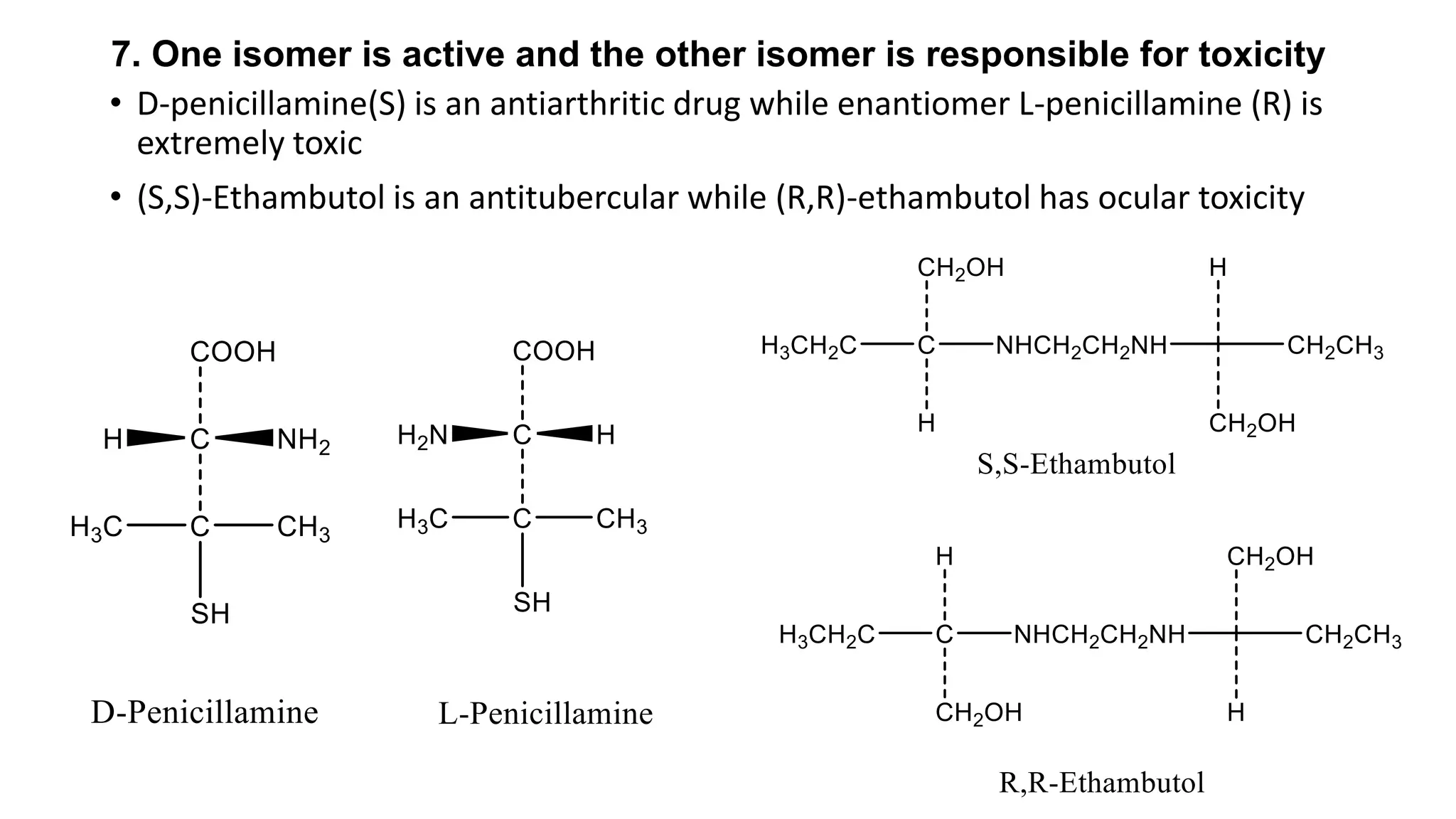
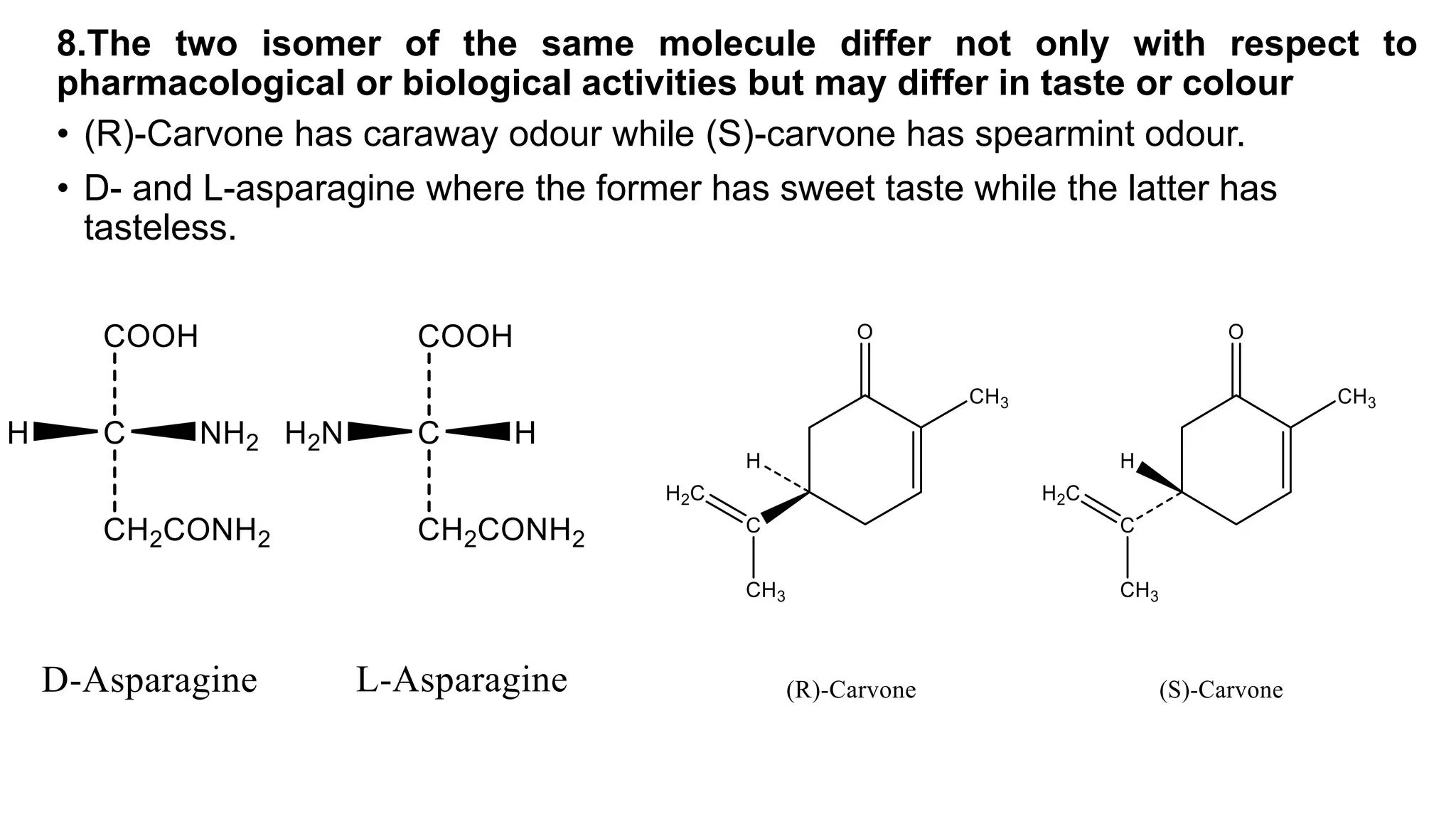
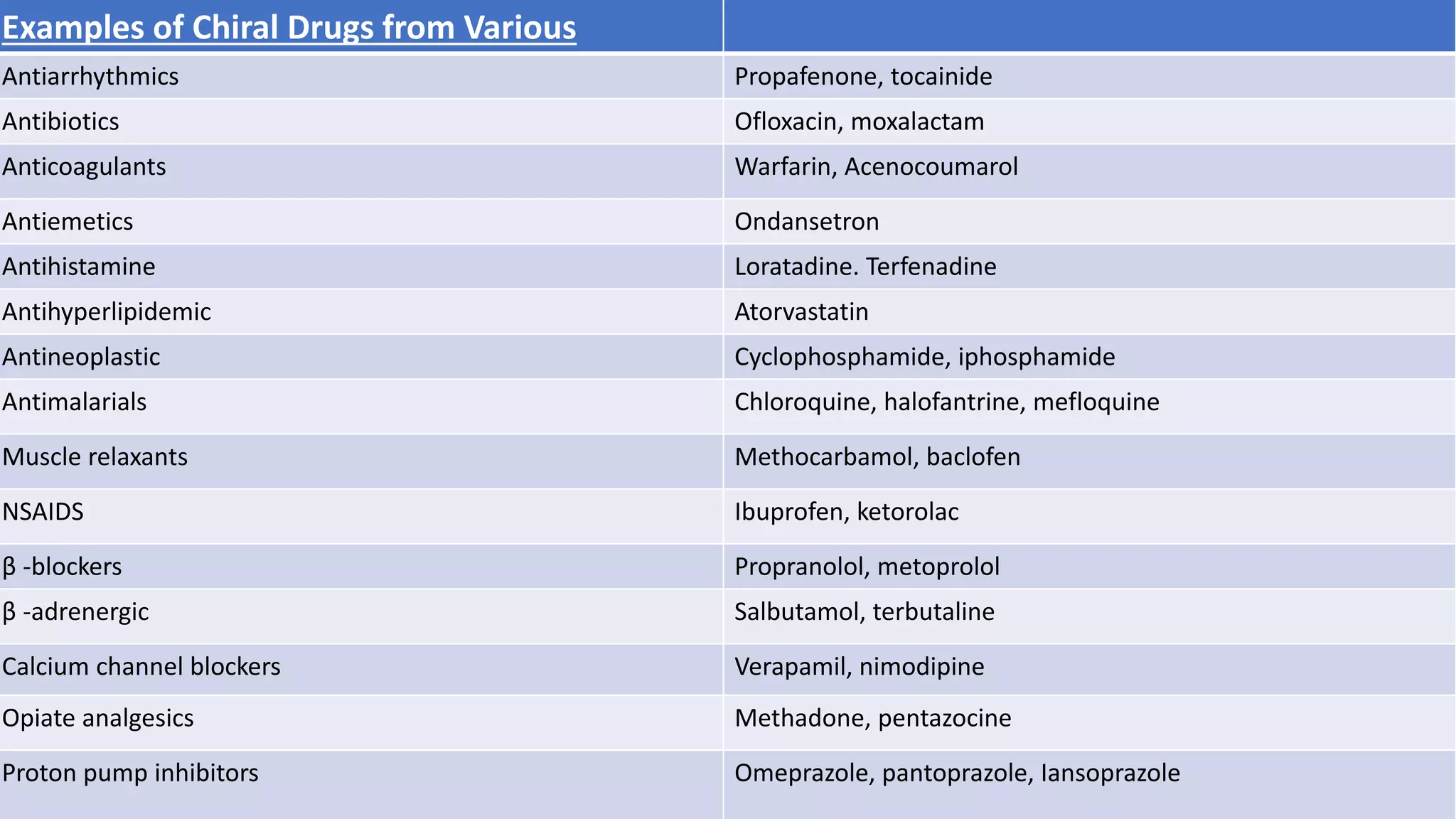
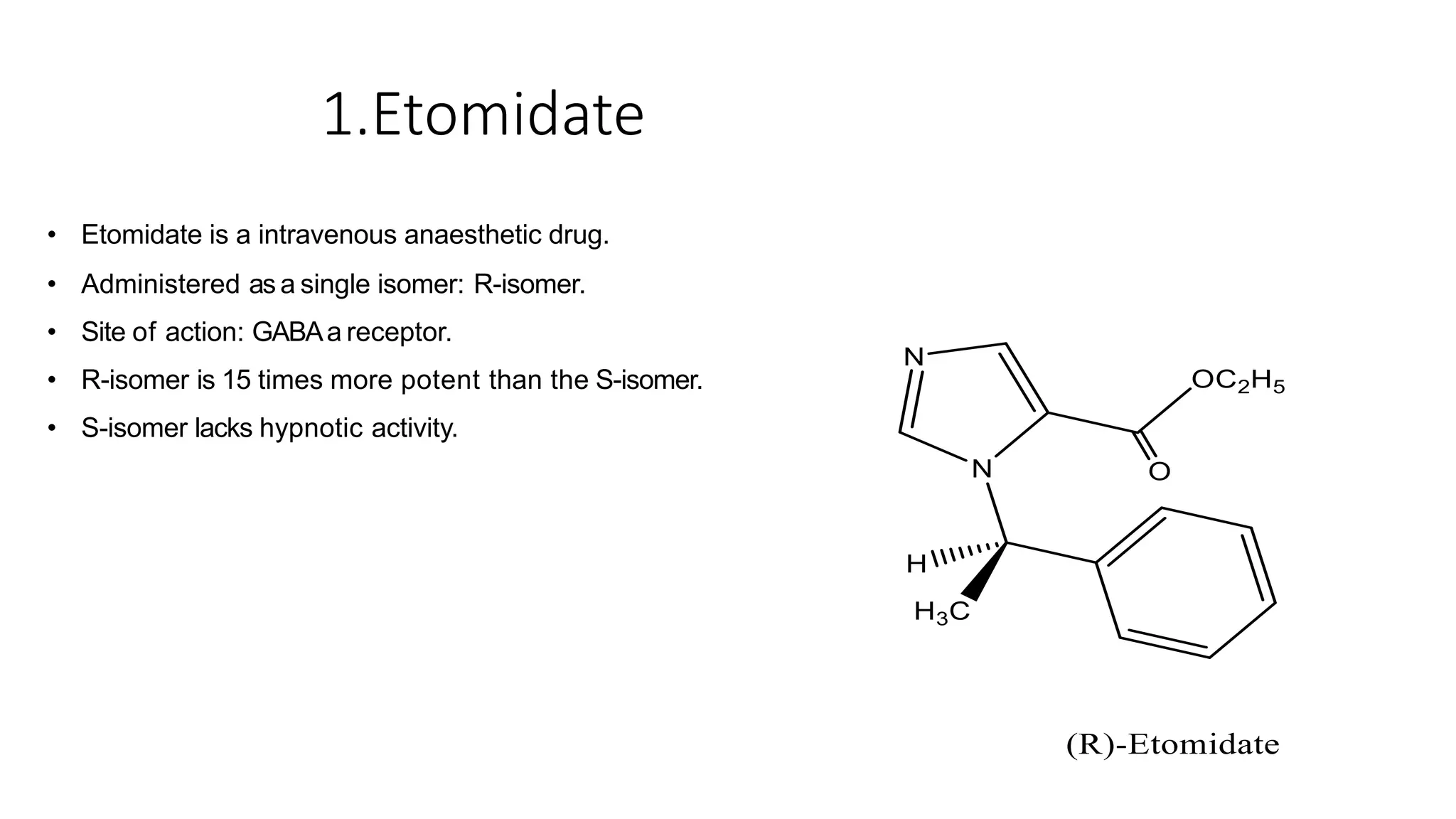
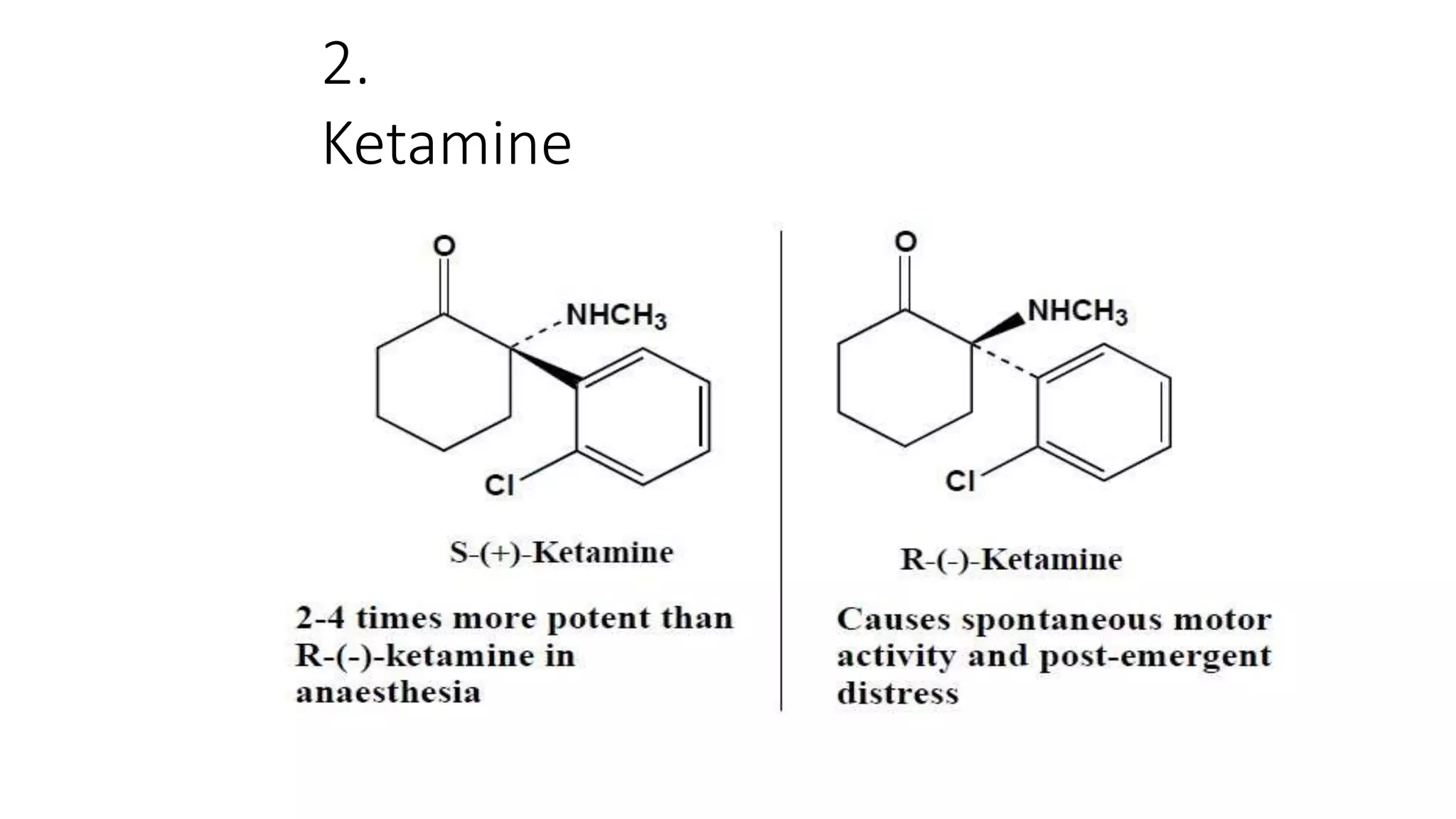
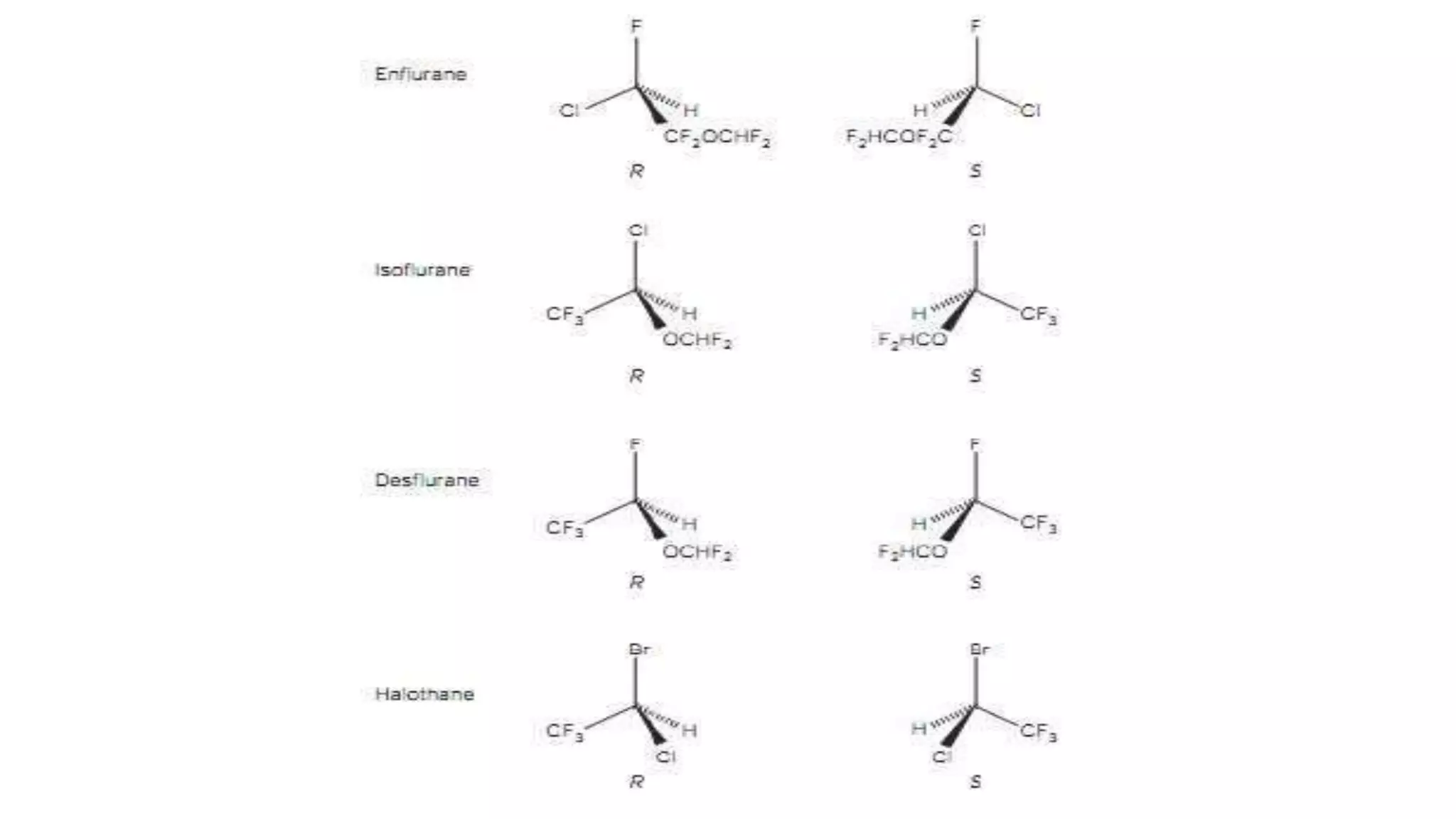
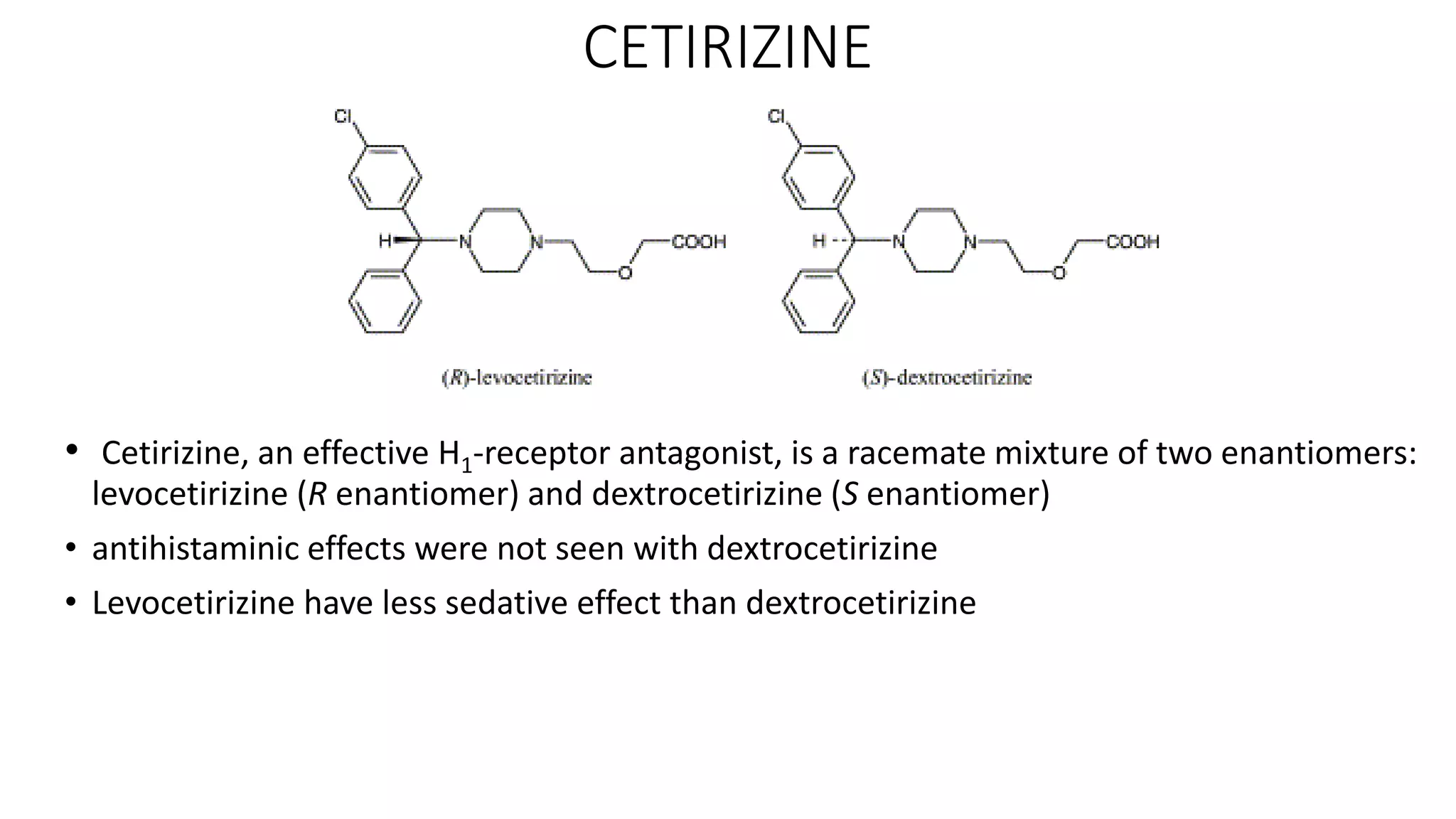
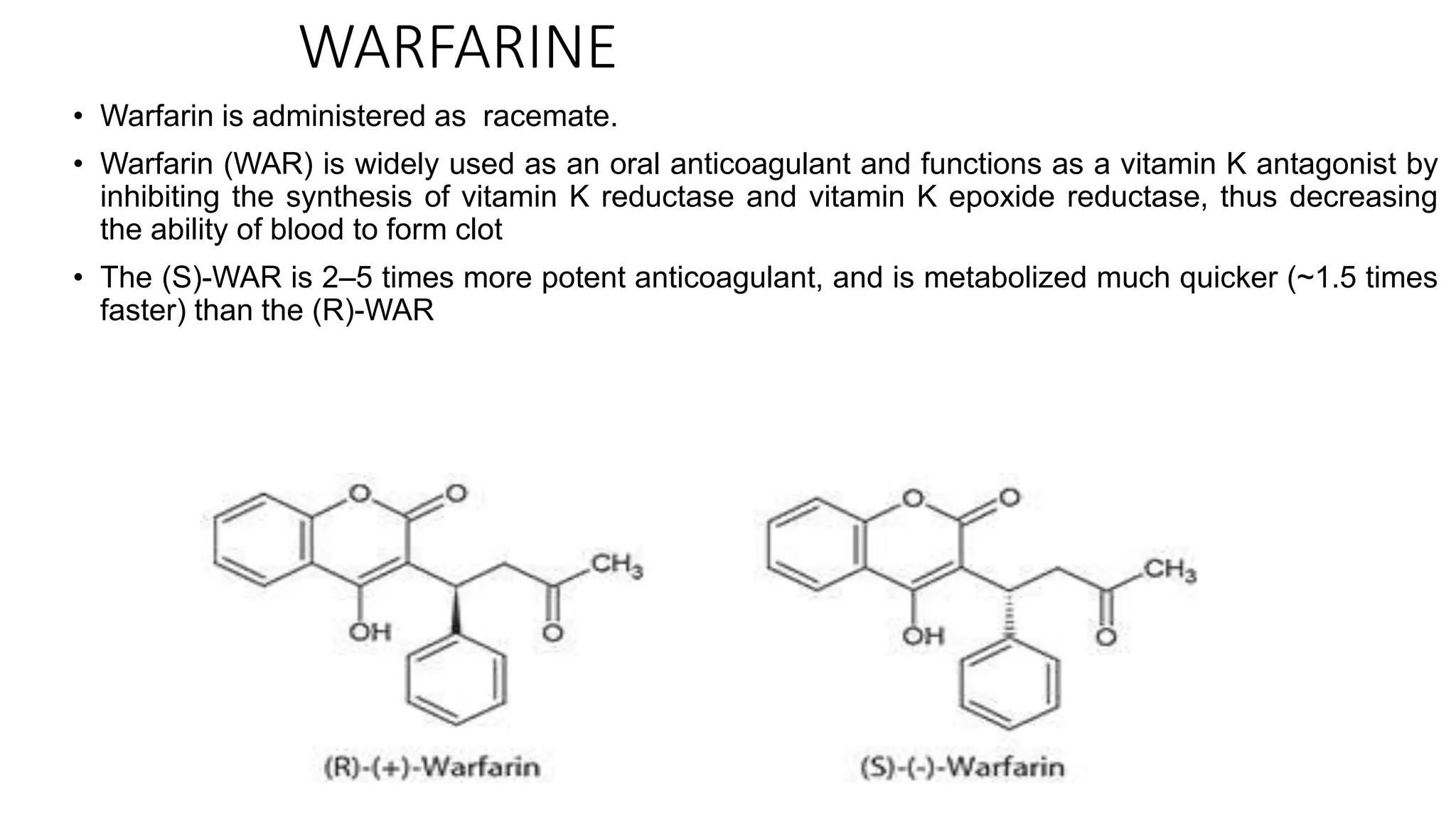
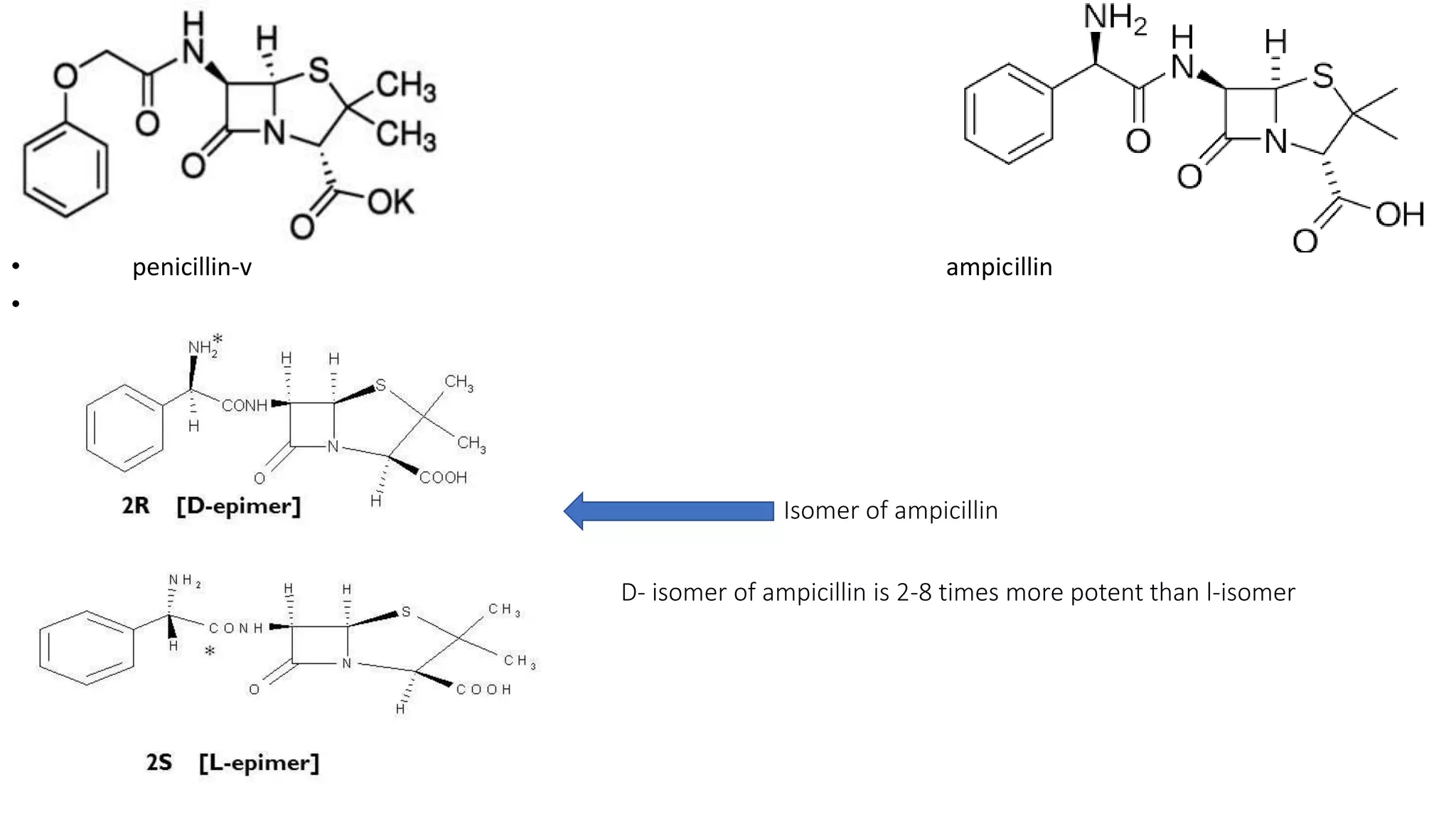
This document discusses the role of chirality in selective therapeutic agents. It begins by defining isomerism and the different types of isomers including constitutional, stereoisomers, optical isomers, enantiomers, and diastereomers. It then discusses the discovery of optical activity and chirality. The key points are that humans are chiral beings and the enantiomers of chiral drugs may have different biological effects. Several examples are given to illustrate how the biological activity of enantiomers can differ, including some being more active, having opposing effects, or one causing toxicity. The importance of understanding chirality in drug development and safety is emphasized.