revision on chapter periodic table, chemical bonding and electrolysis with answers
•Download as PPT, PDF•
2 likes•1,674 views
The document contains information about the periodic table including: 1) It lists the elements hydrogen through argon and their symbols in the periodic table. 2) It explains that noble gases are chemically non-reactive because they have a stable electron configuration with a full outer shell. 3) Sodium undergoes reactions with oxygen, water, and chlorine that produce sodium oxide, sodium hydroxide, and sodium chloride, respectively.
Report
Share
Report
Share
Recommended
INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH AN EXPERIMENT

CHEMISTRY FORM 4 KSSM
CHAPTER 5 : CHEMICAL BONDS (IONIC BOND AND COVALENT BOND)
EXPERIMENT 5.1 TO INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH EXPERIMENT
F.sc.Part.2.Chemistry.(Chapter Wise Tests& Their Solution) - Malik Xufyan

F.sc.part.2.chemistry.(Chapter wise Tests& Their Solution)-Malik Xufyan
Cell # 03137355727
F.sc.2.Chemistry.Ch.05 Solved Test (Malik Xufyan)

F.sc.2.Chemistry.Ch.05 Solved Test (Malik Xufyan)
Cell # 0313-7355727
F.sc. Chemistry Part 2. (inorganic portion tests & solved - Malik Xufyan

F.sc. Chemistry Part 2. (inorganic portion tests & solved - Malik Xufyan
Recommended
INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH AN EXPERIMENT

CHEMISTRY FORM 4 KSSM
CHAPTER 5 : CHEMICAL BONDS (IONIC BOND AND COVALENT BOND)
EXPERIMENT 5.1 TO INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH EXPERIMENT
F.sc.Part.2.Chemistry.(Chapter Wise Tests& Their Solution) - Malik Xufyan

F.sc.part.2.chemistry.(Chapter wise Tests& Their Solution)-Malik Xufyan
Cell # 03137355727
F.sc.2.Chemistry.Ch.05 Solved Test (Malik Xufyan)

F.sc.2.Chemistry.Ch.05 Solved Test (Malik Xufyan)
Cell # 0313-7355727
F.sc. Chemistry Part 2. (inorganic portion tests & solved - Malik Xufyan

F.sc. Chemistry Part 2. (inorganic portion tests & solved - Malik Xufyan
F.Sc.2.Chemistry.Ch.6.Test Solved - Malik Xufyan

F.Sc.2.Chemistry.Ch.6.Test Solved - Malik Xufyan
Cell # 0313-7355727
Inorganic Chemistry : Electrochemistry

Deep explanation on electrochemistry :)
This is actually Malaysian Higher studies syllabus :D
Good Luck
F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan ...

F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan
Cell # 03137355727
Mcqs in inorganic and physical chemistry By Malik Xufyan

Mcqs in inorganic and physical chemistry By Malik Xufyan
strong your chemistry
I.S ACADEMY
03137355727
F.Sc. Part 1 Chemistry.Ch.04.Test (Malik Xufyan)

F.Sc. Part 1 Chemistry.Ch.04.Test (Malik Xufyan)
Cell # 0313-7355727
F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan

F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan
Amazing facts about noble gases...........

this will give you amazing knowledge regarding the nobel gases.................
F.sc.2.Chemistry.Ch.01 Solved Test (Malik Xufyan)

F.sc.2.Chemistry.Ch.01 Solved Test (Malik Xufyan)
Cell#03137355727
Ionization Energy By Malik Xufyan

Ionization Energy By Malik Xufyan
M.Sc Chemistry
Whatsapp # 03137355727
Elements examples

here are some elements their boiling point,melting point,physical properties and chemical properties.
More Related Content
What's hot
F.Sc.2.Chemistry.Ch.6.Test Solved - Malik Xufyan

F.Sc.2.Chemistry.Ch.6.Test Solved - Malik Xufyan
Cell # 0313-7355727
Inorganic Chemistry : Electrochemistry

Deep explanation on electrochemistry :)
This is actually Malaysian Higher studies syllabus :D
Good Luck
F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan ...

F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan
Cell # 03137355727
Mcqs in inorganic and physical chemistry By Malik Xufyan

Mcqs in inorganic and physical chemistry By Malik Xufyan
strong your chemistry
I.S ACADEMY
03137355727
F.Sc. Part 1 Chemistry.Ch.04.Test (Malik Xufyan)

F.Sc. Part 1 Chemistry.Ch.04.Test (Malik Xufyan)
Cell # 0313-7355727
F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan

F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan
Amazing facts about noble gases...........

this will give you amazing knowledge regarding the nobel gases.................
F.sc.2.Chemistry.Ch.01 Solved Test (Malik Xufyan)

F.sc.2.Chemistry.Ch.01 Solved Test (Malik Xufyan)
Cell#03137355727
Ionization Energy By Malik Xufyan

Ionization Energy By Malik Xufyan
M.Sc Chemistry
Whatsapp # 03137355727
What's hot (20)
F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan ...

F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan ...
Mcqs in inorganic and physical chemistry By Malik Xufyan

Mcqs in inorganic and physical chemistry By Malik Xufyan
F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan

F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan
Viewers also liked
Elements examples

here are some elements their boiling point,melting point,physical properties and chemical properties.
Fluoride as pollutant in air, water and its impacts on plants

Fluoride as pollutant in air, water and its impacts on plants
transition metal and halogen

if you are searching about transition metals and halogen stop searching its in front of you if you liked it press like i hope you liked it
Chemistry form 4 ionic bond

It's very good for SPM students . You have to learn the ionic bond thoroughly. If you understand well you can explain it vividly. For other chemistry notes can email me puterizamrud@gmail.com or facebook Pusat Tuisyen Zamrud .
Viewers also liked (20)
Fluoride as pollutant in air, water and its impacts on plants

Fluoride as pollutant in air, water and its impacts on plants
Chapter 9: Contact Process, Haber Process and Alloy

Chapter 9: Contact Process, Haber Process and Alloy
Similar to revision on chapter periodic table, chemical bonding and electrolysis with answers
Chemistry zimsec chapter 9 chemical periodicity

Chemistry zimsec chapter 9 chemical periodicity
Advanced Level
Chemistry
Zimsec
Alpro
Elearning
Chemistry Note Form 4 & 5

Nota ini diharap dapat membantu para pelajar membuat ulangkaji sebelum menghadapi peperiksaan SPM.
Seminar revision on chapter electrchemistry, carbon compound and thermo che...

Basic concepts in chemistry
Seminar revision on chapter electrchemistry, carbon compound and thermo che...

revision on basic concepts in chemistry
Similar to revision on chapter periodic table, chemical bonding and electrolysis with answers (20)
Seminar april revision form 5== chapter electrchemistry, carbon compound and ...

Seminar april revision form 5== chapter electrchemistry, carbon compound and ...
Seminar revision on chapter electrchemistry, carbon compound and thermo che...

Seminar revision on chapter electrchemistry, carbon compound and thermo che...
Seminar revision on chapter electrchemistry, carbon compound and thermo che...

Seminar revision on chapter electrchemistry, carbon compound and thermo che...
More from MRSMPC
Redox part 3= rusting - reactivity series and diff between electrolytic cell...

questions and answers on redox reactions for rusting, reactivity series and diff between electrolytic cell and electrolysis
Soap and detergent, medicine , food additives consumer 2011-edited-2

basic chemistry knowledge on soap, dtergent, food additives, medicines
fats and natural rubber module 3 carbon compounds with answers

basic concept in fats and natural rubber
More from MRSMPC (20)
Redox part 3= rusting - reactivity series and diff between electrolytic cell...

Redox part 3= rusting - reactivity series and diff between electrolytic cell...
Revision on consumer, r te, thermo and carbon compound

Revision on consumer, r te, thermo and carbon compound
Revision on consumer, r te, thermo and carbon compound

Revision on consumer, r te, thermo and carbon compound
Revision for salt, electrochemistry, carbon compounds , thermochemistry

Revision for salt, electrochemistry, carbon compounds , thermochemistry
Soap and detergent, medicine , food additives consumer 2011-edited-2

Soap and detergent, medicine , food additives consumer 2011-edited-2
fats and natural rubber module 3 carbon compounds with answers

fats and natural rubber module 3 carbon compounds with answers
Quiz 2( chemical bond, electrchemistry, acid base)

Quiz 2( chemical bond, electrchemistry, acid base)
Quiz3with answers latest 1(salt, industry, rate of rex)

Quiz3with answers latest 1(salt, industry, rate of rex)
Quiz 2 (chemical bond, electrochemistry, acid base)

Quiz 2 (chemical bond, electrochemistry, acid base)
Recently uploaded
Thesis Statement for students diagnonsed withADHD.ppt

Presentation required for the master in Education.
Overview on Edible Vaccine: Pros & Cons with Mechanism

This ppt include the description of the edible vaccine i.e. a new concept over the traditional vaccine administered by injection.
678020731-Sumas-y-Restas-Para-Colorear.pdf

KKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKK
Unit 8 - Information and Communication Technology (Paper I).pdf

This slides describes the basic concepts of ICT, basics of Email, Emerging Technology and Digital Initiatives in Education. This presentations aligns with the UGC Paper I syllabus.
Francesca Gottschalk - How can education support child empowerment.pptx

Francesca Gottschalk from the OECD’s Centre for Educational Research and Innovation presents at the Ask an Expert Webinar: How can education support child empowerment?
1.4 modern child centered education - mahatma gandhi-2.pptx

Child centred education is an educational approach that priorities the interest, needs and abilities of the child in the learning process.
Home assignment II on Spectroscopy 2024 Answers.pdf

Answers to Home assignment on UV-Visible spectroscopy: Calculation of wavelength of UV-Visible absorption
Digital Tools and AI for Teaching Learning and Research

This Presentation in details discusses on Digital Tools and AI for Teaching Learning and Research
Chapter 3 - Islamic Banking Products and Services.pptx

Chapter 3 - Islamic Banking Products and Services.pptxMohd Adib Abd Muin, Senior Lecturer at Universiti Utara Malaysia
This slide is prepared for master's students (MIFB & MIBS) UUM. May it be useful to all.Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46

Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46
Event Link:-
https://meetups.mulesoft.com/events/details/mulesoft-mysore-presents-exploring-gemini-ai-and-integration-with-mulesoft/
Agenda
● Java 17 Upgrade Overview
● Why and by when do customers need to upgrade to Java 17?
● Is there any immediate impact to upgrading to Mule Runtime 4.6 and beyond?
● Which MuleSoft products are in scope?
For Upcoming Meetups Join Mysore Meetup Group - https://meetups.mulesoft.com/mysore/
YouTube:- youtube.com/@mulesoftmysore
Mysore WhatsApp group:- https://chat.whatsapp.com/EhqtHtCC75vCAX7gaO842N
Speaker:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Priya Shaw - https://www.linkedin.com/in/priya-shaw
Organizers:-
Shubham Chaurasia - https://www.linkedin.com/in/shubhamchaurasia1/
Giridhar Meka - https://www.linkedin.com/in/giridharmeka
Priya Shaw - https://www.linkedin.com/in/priya-shaw
Shyam Raj Prasad-
https://www.linkedin.com/in/shyam-raj-prasad/
Biological Screening of Herbal Drugs in detailed.

Biological screening of herbal drugs: Introduction and Need for
Phyto-Pharmacological Screening, New Strategies for evaluating
Natural Products, In vitro evaluation techniques for Antioxidants, Antimicrobial and Anticancer drugs. In vivo evaluation techniques
for Anti-inflammatory, Antiulcer, Anticancer, Wound healing, Antidiabetic, Hepatoprotective, Cardio protective, Diuretics and
Antifertility, Toxicity studies as per OECD guidelines
special B.ed 2nd year old paper_20240531.pdf

Instagram:-
https://instagram.com/special_education_needs_01?igshid=YmMyMTA2M2Y=
WhatsApp:-
https://chat.whatsapp.com/JVakNIYlSV94x7bwunO3Dc
YouTube:-
https://youtube.com/@special_education_needs
Teligram :- https://t.me/special_education_needs
Slide Shere :-
https://www.slideshare.net/shabnambano20?utm_campaign=profiletracking&utm_medium=sssite&utm_source=ssslideview
Polish students' mobility in the Czech Republic

Polish students mobility to the Czech Republic within eTwinning project "Medieval adventures with Marco Polo"
Synthetic Fiber Construction in lab .pptx

Synthetic fiber production is a fascinating and complex field that blends chemistry, engineering, and environmental science. By understanding these aspects, students can gain a comprehensive view of synthetic fiber production, its impact on society and the environment, and the potential for future innovations. Synthetic fibers play a crucial role in modern society, impacting various aspects of daily life, industry, and the environment. ynthetic fibers are integral to modern life, offering a range of benefits from cost-effectiveness and versatility to innovative applications and performance characteristics. While they pose environmental challenges, ongoing research and development aim to create more sustainable and eco-friendly alternatives. Understanding the importance of synthetic fibers helps in appreciating their role in the economy, industry, and daily life, while also emphasizing the need for sustainable practices and innovation.
CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE

Class 11 CBSE Business Studies Project ( AIDS TO TRADE - INSURANCE)
Acetabularia Information For Class 9 .docx

Acetabularia acetabulum is a single-celled green alga that in its vegetative state is morphologically differentiated into a basal rhizoid and an axially elongated stalk, which bears whorls of branching hairs. The single diploid nucleus resides in the rhizoid.
The approach at University of Liverpool.pptx

How libraries can support authors with open access requirements for UKRI funded books
Wednesday 22 May 2024, 14:00-15:00.
Recently uploaded (20)
Thesis Statement for students diagnonsed withADHD.ppt

Thesis Statement for students diagnonsed withADHD.ppt
Overview on Edible Vaccine: Pros & Cons with Mechanism

Overview on Edible Vaccine: Pros & Cons with Mechanism
Unit 8 - Information and Communication Technology (Paper I).pdf

Unit 8 - Information and Communication Technology (Paper I).pdf
Francesca Gottschalk - How can education support child empowerment.pptx

Francesca Gottschalk - How can education support child empowerment.pptx
1.4 modern child centered education - mahatma gandhi-2.pptx

1.4 modern child centered education - mahatma gandhi-2.pptx
Home assignment II on Spectroscopy 2024 Answers.pdf

Home assignment II on Spectroscopy 2024 Answers.pdf
Digital Tools and AI for Teaching Learning and Research

Digital Tools and AI for Teaching Learning and Research
Chapter 3 - Islamic Banking Products and Services.pptx

Chapter 3 - Islamic Banking Products and Services.pptx
Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46

Mule 4.6 & Java 17 Upgrade | MuleSoft Mysore Meetup #46
CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE

CLASS 11 CBSE B.St Project AIDS TO TRADE - INSURANCE
revision on chapter periodic table, chemical bonding and electrolysis with answers
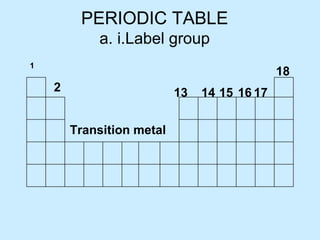
- 1. PERIODIC TABLE a. i.Label group 1 17 Transition metal 13 14 15 16 18 2
- 2. ii.Label period 1 to 5 Period 1 Period 2 Period 3 Period 4 Period 5 1 2 Transition metal 13 14 15 16 17 18
- 3. iii.Place proton number of elements 1 to 20 in the periodic table above 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
- 4. iv.Place all the elements given in the periodic table above with their symbols Transition metal H He F Ne Na Cu Cl Br Ar K Mn Li
- 7. Sodium undergoes the following reactions. Write chemical equation: i) With oxygen: ii) With water: iii) With chlorine: 4Na + O 2 2Na 2 O 2Na + 2H 2 O 2NaOH + H 2 2Na + Cl 2 2NaCl
- 18. Draw electron arrangement for compounds formed between a) Sodium and oxygen ( given proton no. Na, 11: O, 8)
- 20. b) For water molecule
- 22. c) Carbon and chlorine
- 26. 1. MOLTEN LEAD(II)IODIDE WITH CARBON ELECTRODE
- 29. ANODE CATHODE OBSER A PURPLE GAS EVOLVED GREY AND SHINY SOLID DEPOSITED
- 30. 2. HYDROCHLORIC ACID 0.0001 moldm -3 WITH CARBON ELECTRODE ALL IONS PRESENT H + , Cl - & OH -
- 33. ANODE CATHODE OBS COLOURLESS GAS THAT REKINDLES GLOWING WOODEN SPLINTER COLOURLESS GAS THAT GIVES ‘POP’ SOUND -TESTED USING BURNING WOODEN SPLINTER
