Group 18 of the periodic table contains the noble gases: helium, neon, argon, krypton, xenon, and radon. All noble gases are monoatomic gases that are chemically unreactive due to their stable electronic configurations. Xenon can form some compounds with highly electronegative elements like fluorine and oxygen. Xenon fluorides like XeF2 and XeF6 are powerful fluorinating agents. While most noble gases have no significant uses, argon is used for welding and as an inert atmosphere, and neon is used in signs and greenhouses.









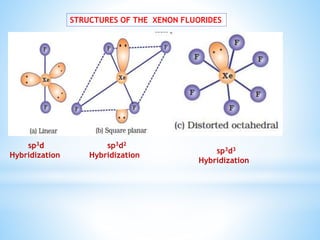
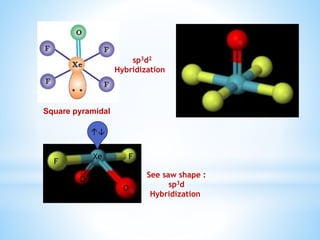
![ Xenon fluorides react with fluoride ion acceptors to
form cationic species and fluoride ion donors to form
fluoroanions.
XeF2 + PF5 → [XeF]+ [PF6]–
XeF4 + SbF5 → [XeF3]+ [SbF6]–
XeF6 + MF → M+ [XeF7]– (M = Na, K, Rb or Cs)](https://image.slidesharecdn.com/18elementszzznobelgas-150714171514-lva1-app6891/85/Amazing-facts-about-noble-gases-12-320.jpg)