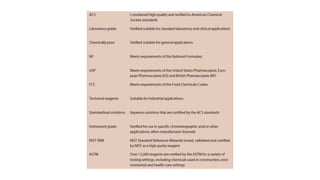
1. The document defines reagents as substances added to cause or test for chemical reactions. It describes analytical reagents which are used for qualitative or quantitative analysis and lists organizations that establish purity levels.
2. Types of analytical reagents are described including those for chemical analysis, indicators, and instrumentation. Laboratory reagents are compounds used to detect or measure other substances through color changes.
3. The key difference between primary and secondary standards is that primary standards are more pure and less reactive while secondary standards are less pure and more reactive. Primary standards are used to standardize secondary standards.