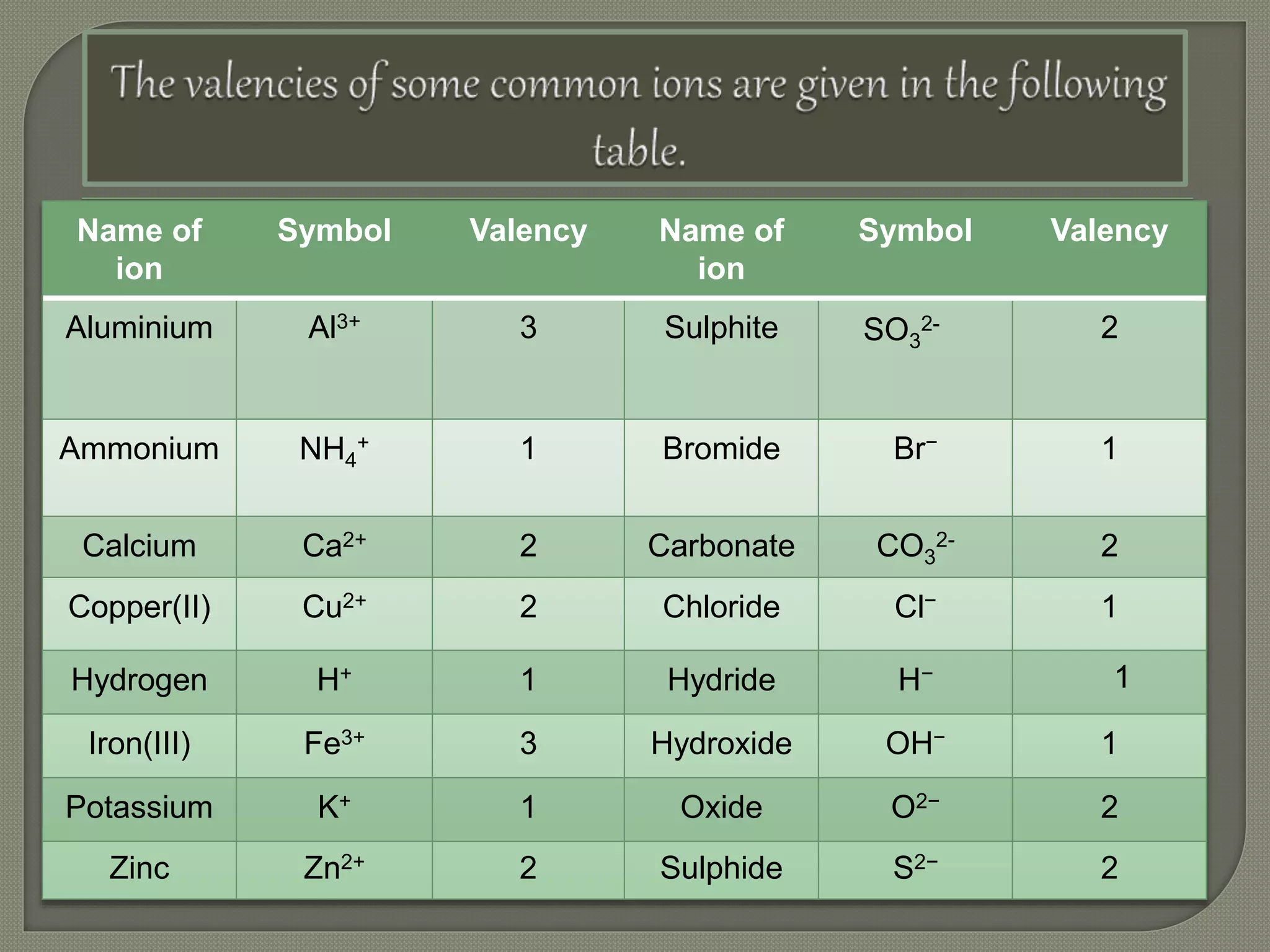
This document discusses radicals, ions, and how to write chemical formulas. Radicals are species with unpaired electrons that can exist independently, and there are two main types - acid and basic radicals. Ions are charged species formed when atoms gain or lose electrons to become cations (positively charged) or anions (negatively charged). To write a chemical formula, you need the symbols of the elements and their valencies (combining capacities), then cross-multiply the valencies to get the formula. Examples of common cation and anion symbols and their valencies are provided.