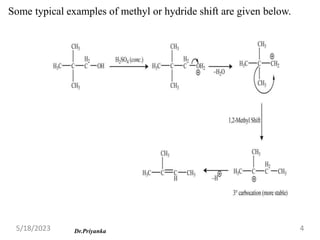
The document discusses elimination reactions, focusing on carbocation rearrangements and the differences between E1 and E2 mechanisms. It highlights Saytzeff's rule for predicting major products and factors influencing the rates of E1 and E2 reactions, such as substrate structure, base strength, and solvent effects. Additionally, the document notes the competitive nature of elimination and substitution reactions, with conditions favoring one over the other depending on various factors.