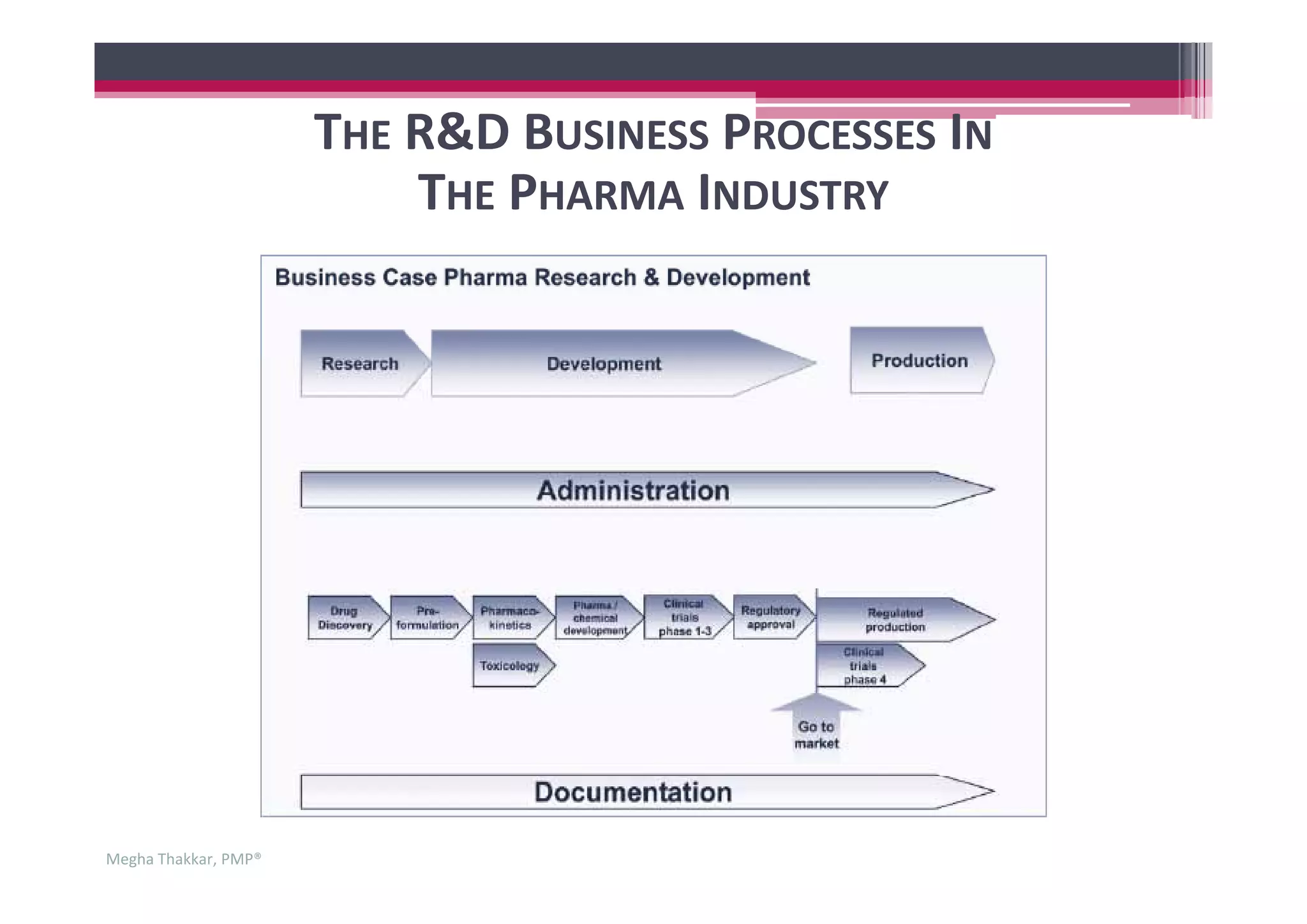
The document outlines the project management process in the pharmaceutical industry, detailing the stages of research and development (R&D) from drug discovery to market launch. It emphasizes the complexity and unpredictability of drug development, highlighting the importance of project management in coordinating cross-functional teams and optimizing resources. Key phases such as clinical trials, regulatory approval, and post-launch activities are discussed, along with the essential skills required for effective project management in this sector.