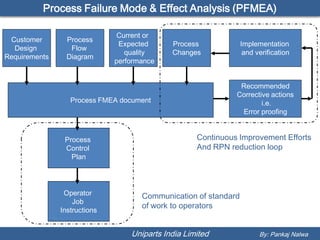
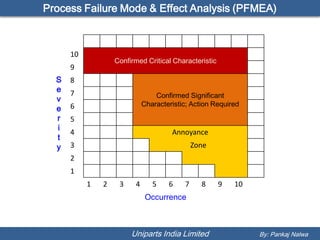
The document discusses Process Failure Mode and Effect Analysis (PFMEA). It explains that every product or process can have failure modes, even established ones, and that effective FMEAs require a team effort and should be done early in the design process. It also outlines the basic steps for a process FMEA, which involves identifying potential failures, effects, risks, and taking actions to reduce high-risk failures. The objective is to uncover process problems and reduce the risk of failures affecting products, efficiency or safety.