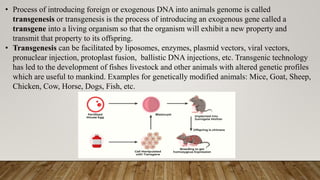
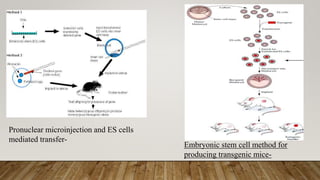
The document presents a detailed overview of transgenic animals, which are organisms that have been genetically modified to carry foreign genes for various purposes, including increased productivity in agriculture and advancements in medical research. It outlines methods for creating transgenic animals, historical milestones, advantages and disadvantages of specific species, and applications such as xenotransplantation and gene therapy. The document emphasizes the potential for transgenic technology to enhance traits in livestock and serve as models for human diseases, while also addressing challenges and ethical considerations.