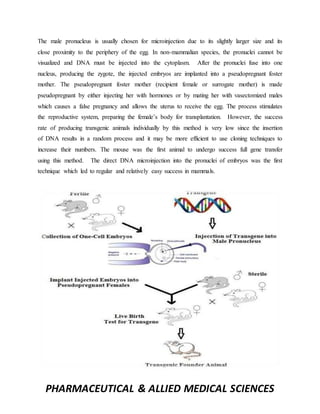
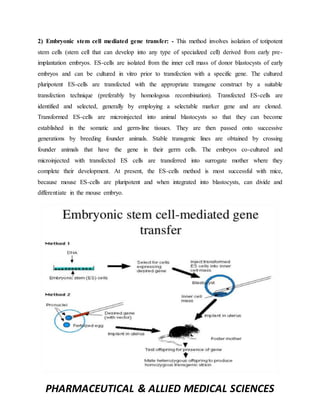
This document discusses methods for creating transgenic animals. It defines transgenic animals as those with recombinant DNA introduced through human intervention. The major methods described are DNA microinjection, embryonic stem cell mediated gene transfer, retrovirus mediated gene transfer, use of transposons, sperm mediated gene transfer, and nuclear transfer. Applications mentioned include using transgenic animals as models for studying oncogenesis, diseases, and producing therapeutic proteins.