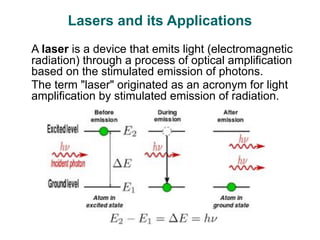
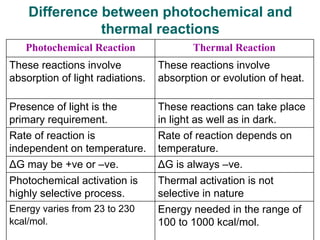
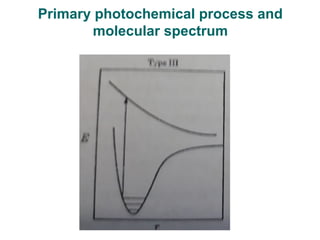
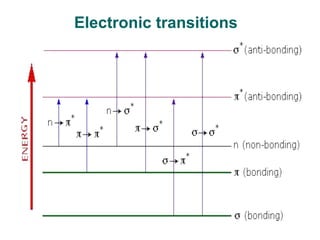
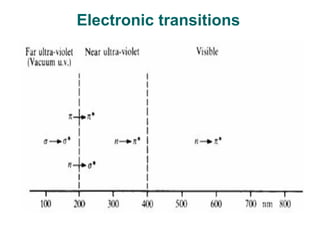
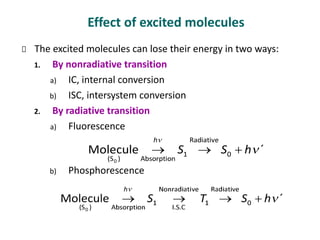
Photochemistry is the study of chemical reactions caused by the absorption of visible or ultraviolet light. There are two main types of reactions: primary processes where light is directly absorbed, and secondary processes involving excited states produced in primary processes. Factors like fluorescence, quantum yield, and chain reactions can affect the efficiency of photochemical reactions. Photochemistry plays roles in important natural and industrial processes.









































































![5. Uranyl ion as a photosensitizer
Uranyl ion acts as a photosensitizer in the
photolysis of formic acid
UO2
2+
+ hν → [UO2
2+
]*
[UO2
2+
]* + HOOC-COOH → CO2
+ CO + H2
O + UO2
2+
6. Chlorophyll as a photosensitizer
Chlorophyll acts as a photosensitizer in the
photosynthesis of carbohydrate from CO2
and H2
O.
Chlorophyll + hν → [Chlorophyll]*
6CO2
+ H2
O + [Chlorophyll]* → C6
H12
O6
+ O2
+
Chlorophyll
Photosensitization](https://image.slidesharecdn.com/chapter3-photochemistry-230412212355-3c67a85f/85/Photochemistry-pdf-81-320.jpg)