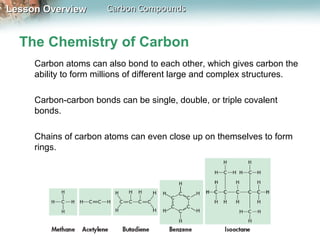
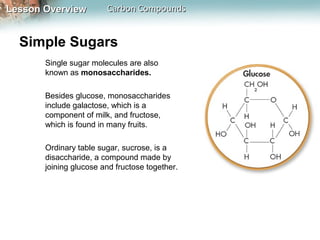
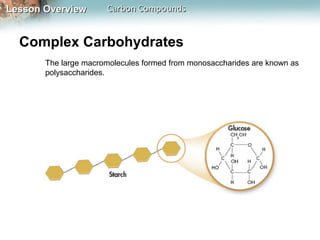
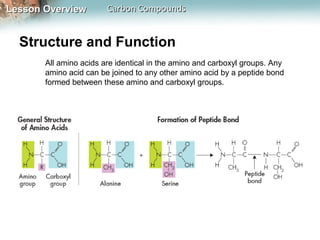
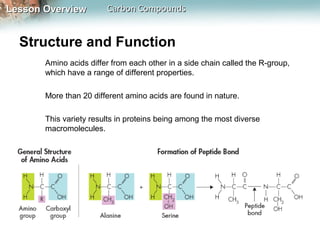
1) Carbon atoms can form strong bonds with many other elements due to having four valence electrons. This allows carbon to form the complex structures that make up living organisms.
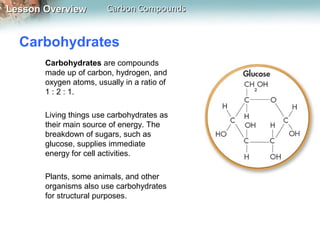
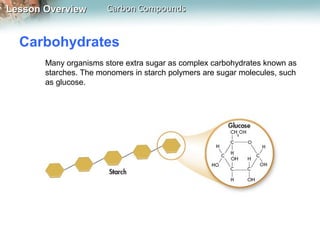
2) The four main macromolecules found in living things are carbohydrates, lipids, nucleic acids, and proteins. Carbohydrates, lipids, and proteins are made of carbon, hydrogen, and oxygen while nucleic acids also contain phosphorus and nitrogen.
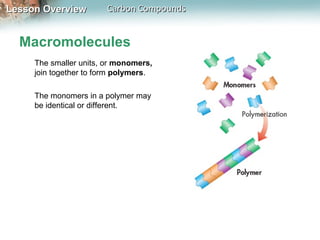
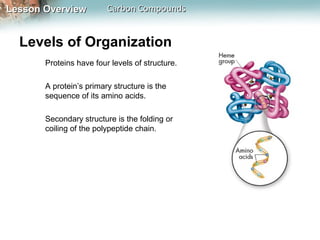
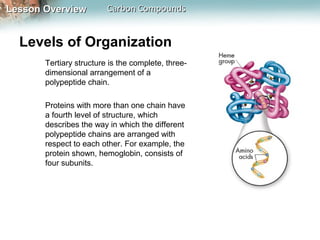
3) Macromolecules perform essential functions like energy storage, structure, heredity, catalysis, and transport. They are formed by polymerization of smaller monomers and have primary, secondary, tertiary, and quaternary levels of structure.