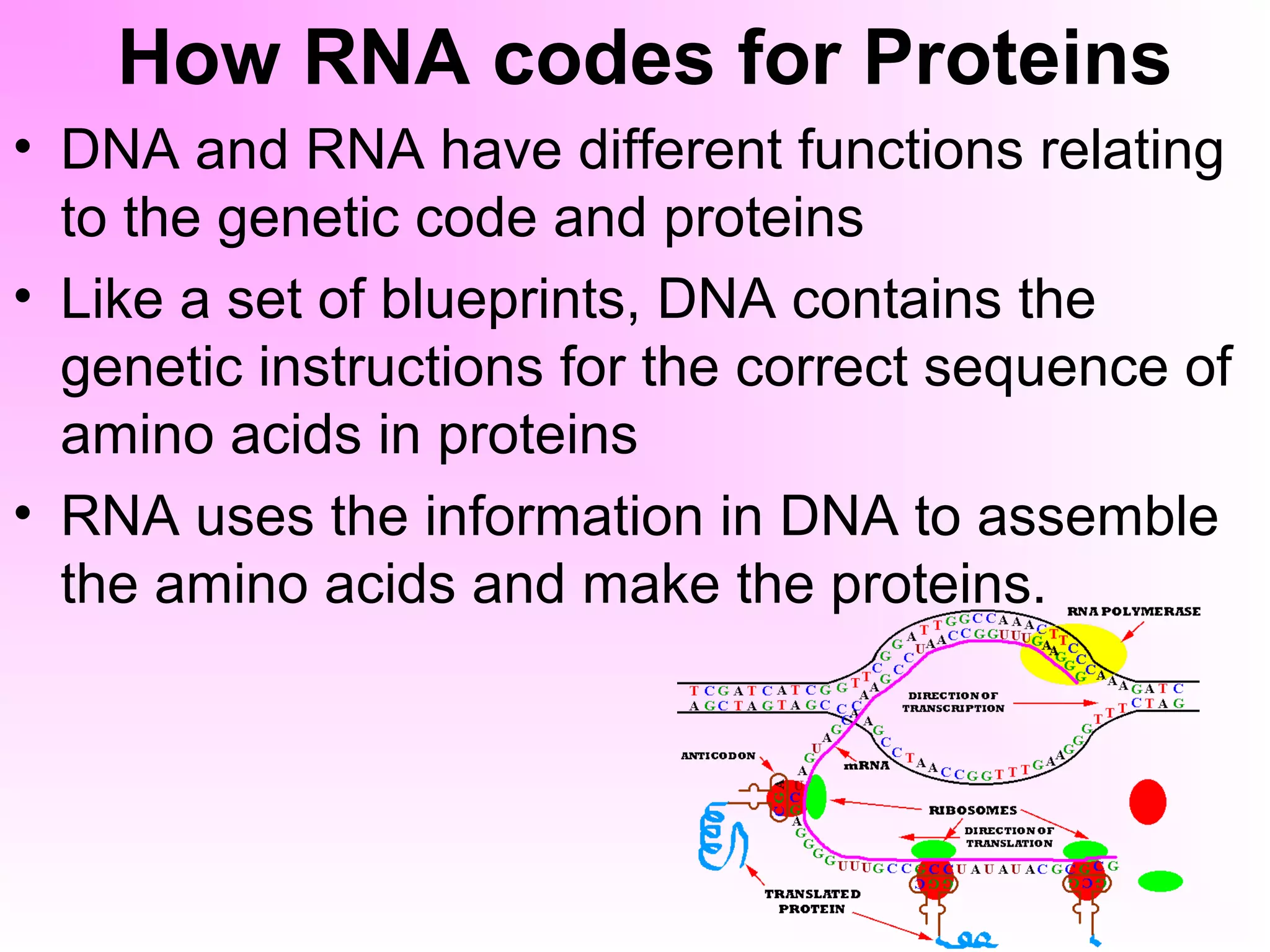
This document discusses the four major types of organic compounds found in living things: carbohydrates, lipids, proteins, and nucleic acids. It describes the structure and functions of each compound. Carbohydrates such as glucose provide energy, while lipids store energy and form cell membranes. Proteins have a variety of functions including maintaining cell shape and catalyzing reactions. Nucleic acids like DNA and RNA pass on traits by coding for amino acids and assembling proteins. Carbon is essential to life because it can form the large complex molecules that make up living organisms.