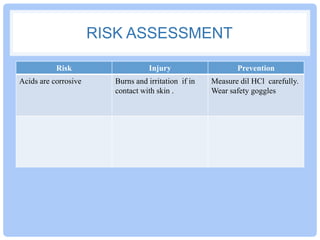
This document describes a laboratory experiment to measure the amount of antacid needed to neutralize hydrochloric acid. Students will add increasing amounts of antacid powder to 50mL of dilute hydrochloric acid while monitoring the color change of an indicator from red to orange, which indicates neutralization. They will record the mass of antacid used and compare results with other groups. Questions address calculating the amount of antacid needed for larger volumes of acid. The aim is to observe the neutralization reaction between an acid and base.