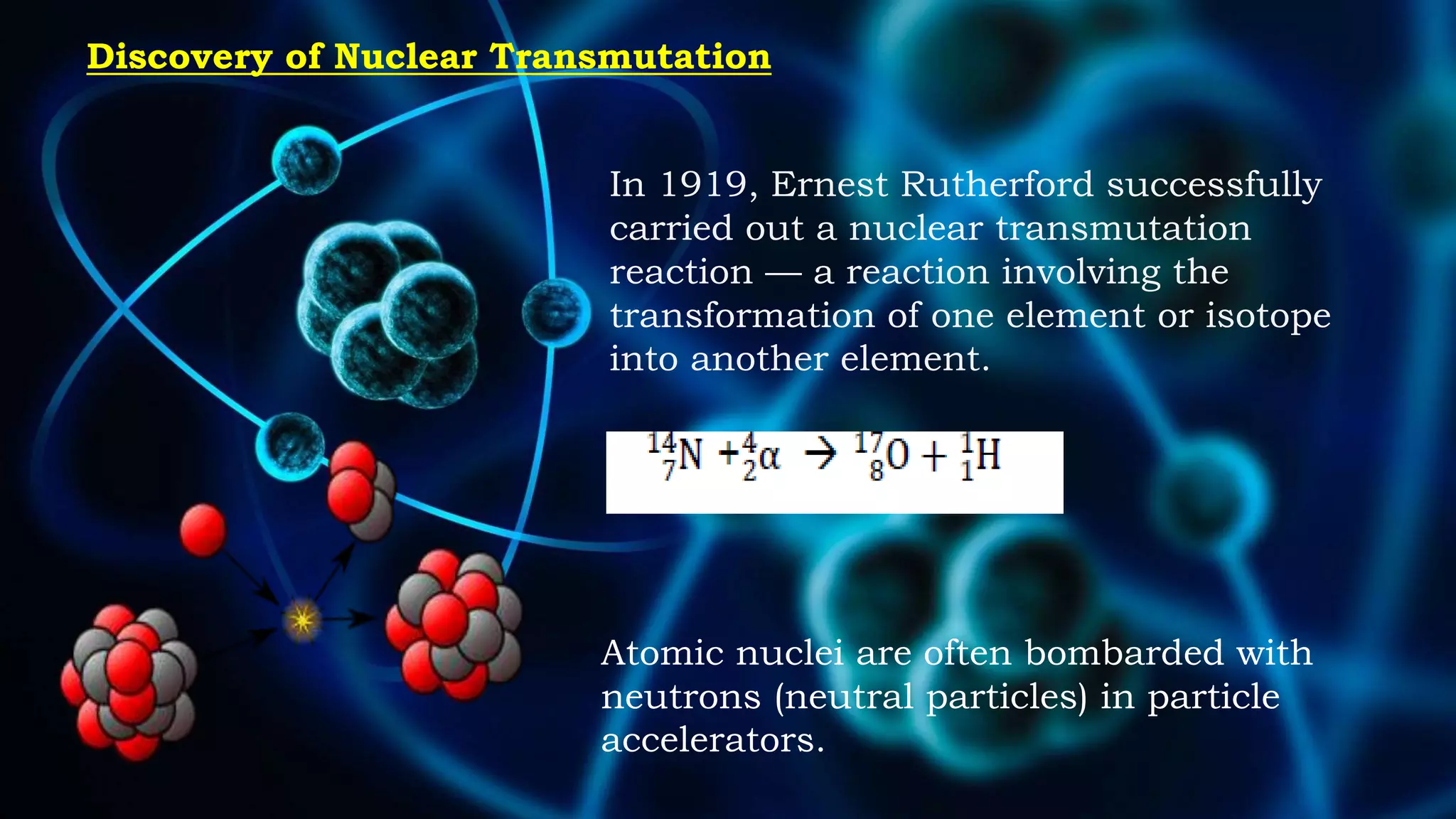
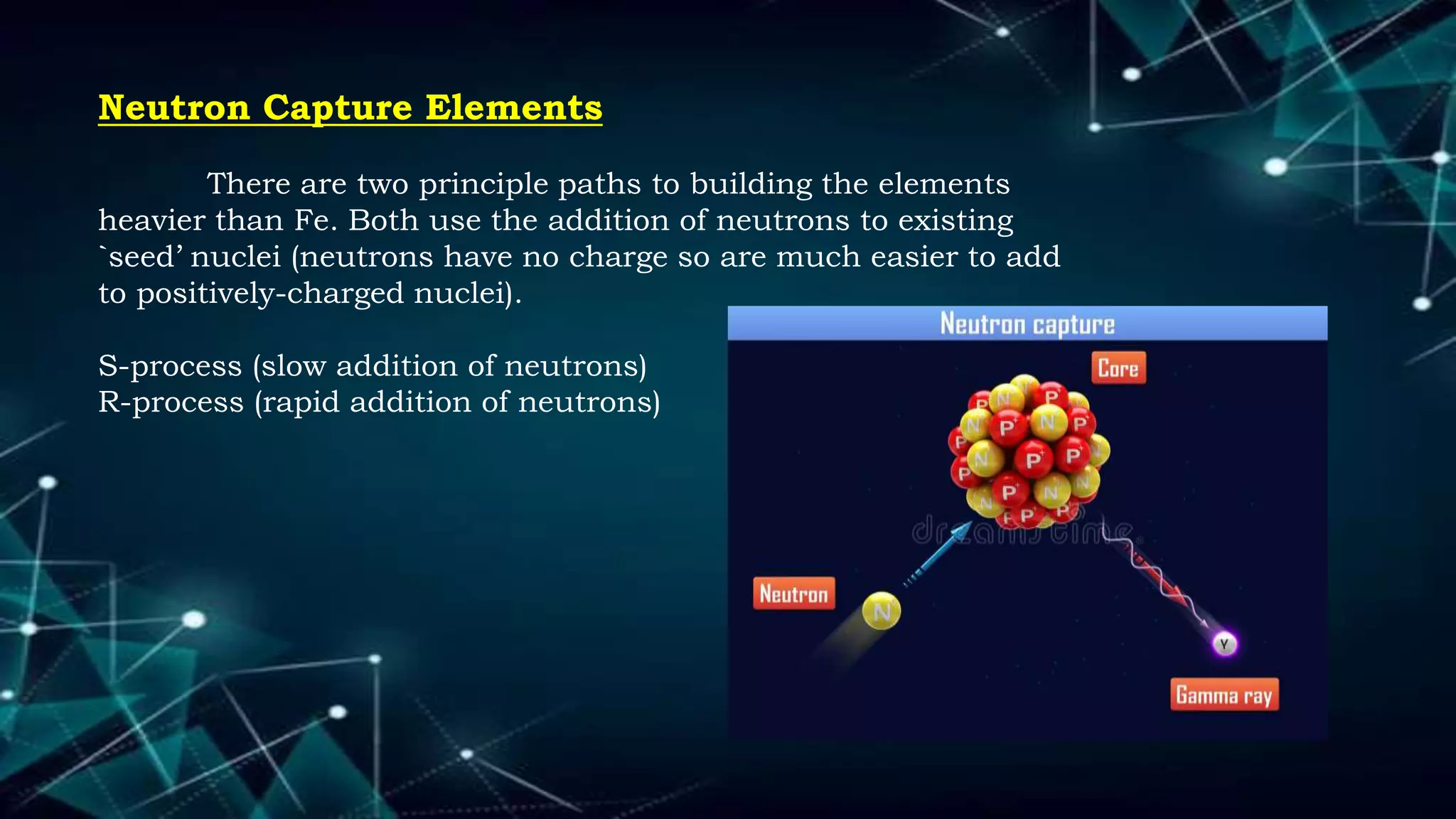
1. The concept of atomic number led scientists to synthesize new elements in the laboratory by bombarding target elements with particles using particle accelerators.
2. Early synthesized elements included technetium with atomic number 43 and elements with atomic numbers 43 and 85.
3. The atomic number determines an element's properties and identity on the periodic table, allowing scientists to identify newly synthesized elements.