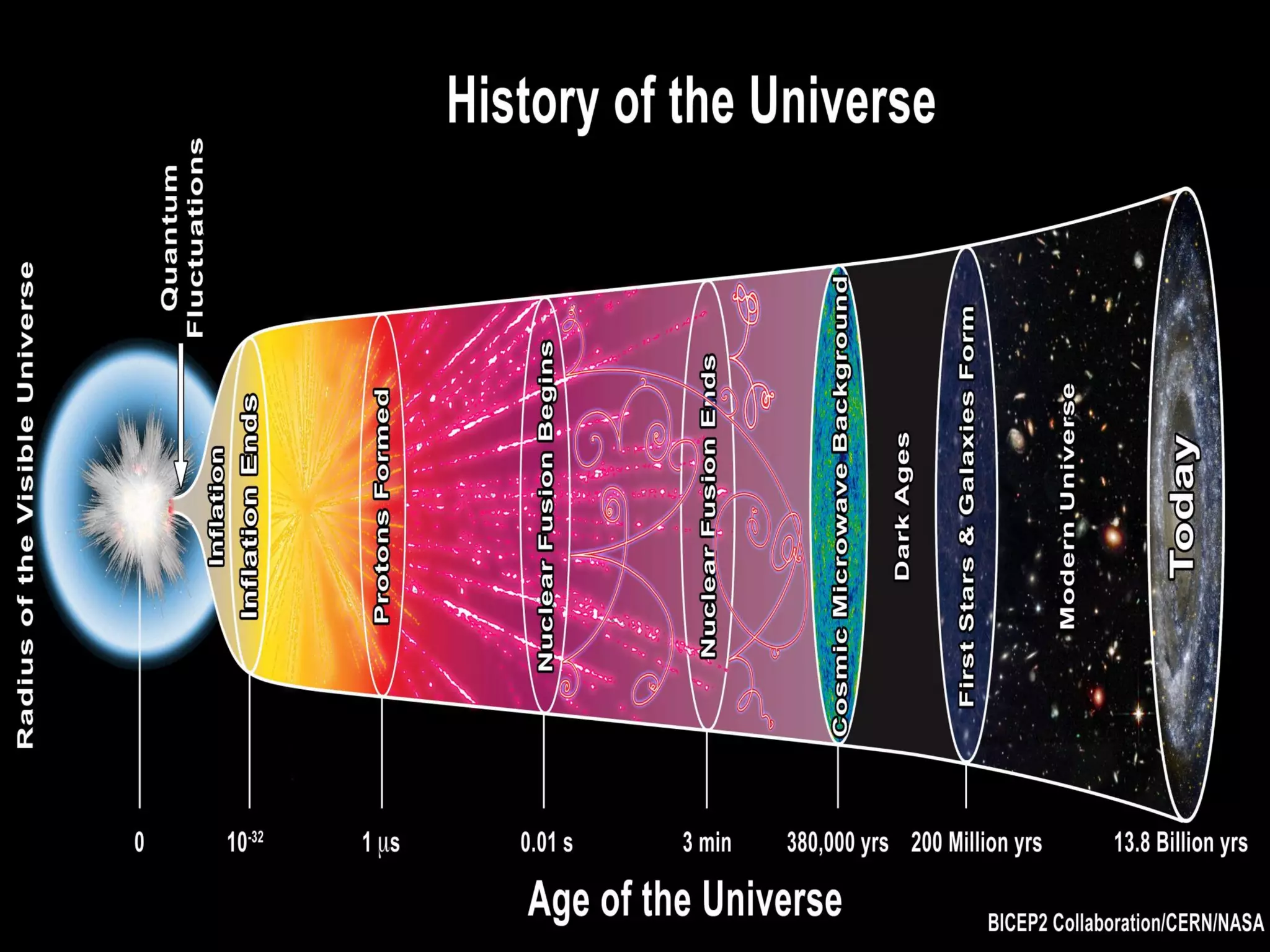
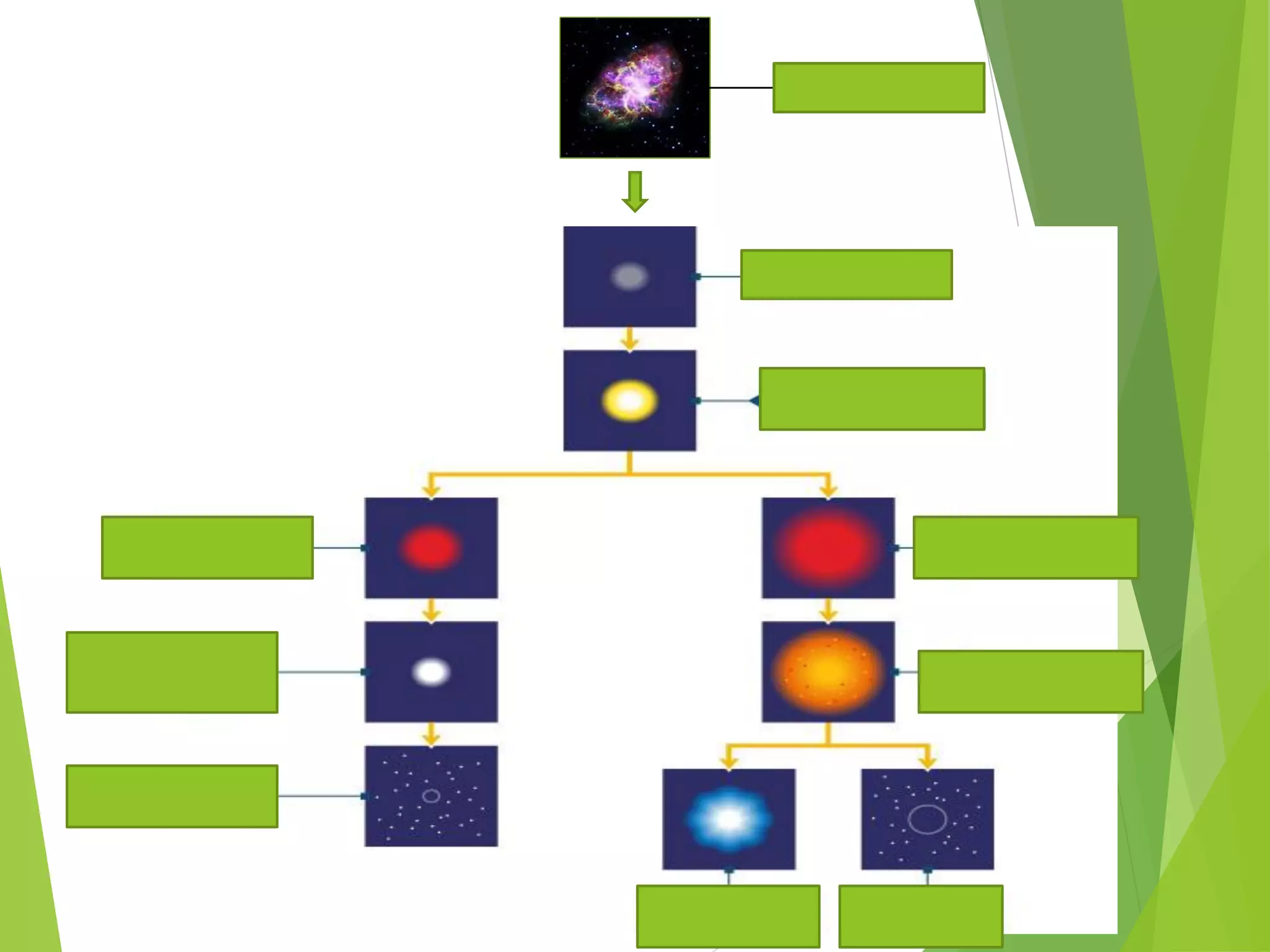
This document provides information about stars and their formation. It begins by discussing how the universe originated from the Big Bang approximately 13 billion years ago. It then explains that early in the universe's formation, light elements like hydrogen and helium were formed through nuclear fusion. The document goes on to state that stars are formed from clouds of dust and gas in nebulae, and that nuclear fusion in the cores of stars is responsible for forming heavier elements.