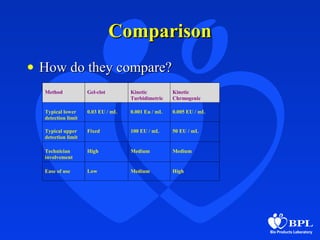
The document discusses three main methods for the bacterial endotoxin test - gel clot, turbidimetric, and chromogenic. The gel clot method is the simplest but least quantitative, while turbidimetric and chromogenic methods allow for more automation and precision using spectrophotometry. All three methods use Limulus amebocyte lysate and detect endotoxins through coagulation reactions. The choice of method depends on factors like testing volumes, sample properties, required sensitivity, and compliance needs. Photometric methods have advantages of automation and precision but higher costs, while gel clot is inexpensive but less quantitative.