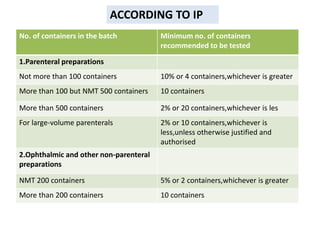
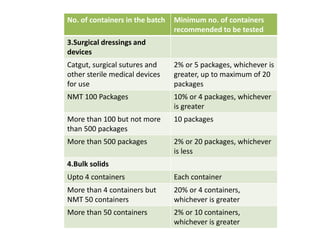
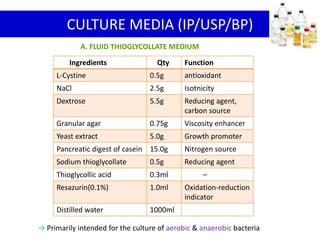
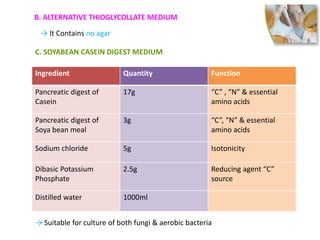
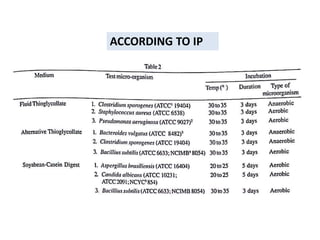
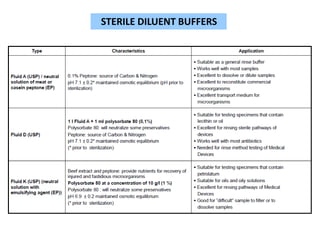
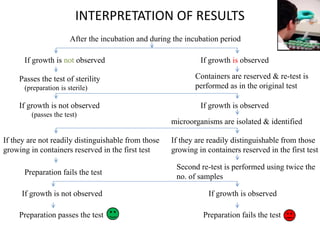
This document discusses sterility testing methods according to various pharmacopoeias. It provides details on membrane filtration and direct inoculation methods for testing sterility of pharmaceutical products like injections and ophthalmic preparations. These methods are based on incubating the product samples in fluid thioglycollate medium and soybean-casein digest medium to check for microbial growth. Validation of sterility testing methods and interpretation of results are also covered.