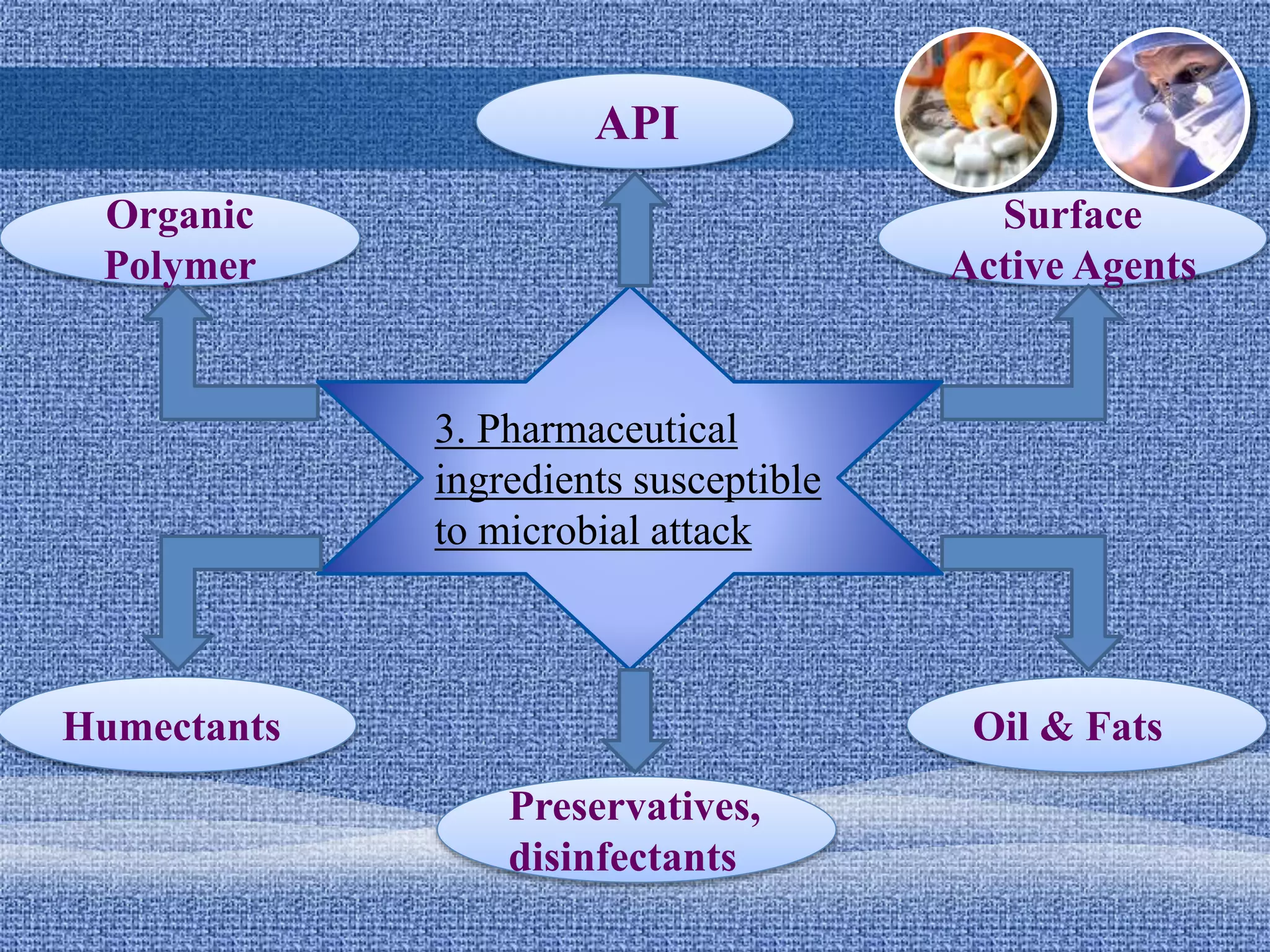
Microbial contamination of pharmaceutical products can lead to spoilage or health hazards. Contamination can occur during manufacturing from water, the environment or packaging. It can be prevented through good manufacturing practices and controlling moisture, temperature, pH and packaging design. Spoilage results from microbial growth and causes product deterioration or infection. Factors influencing spoilage include the contaminant type/level, nutrients, moisture, temperature, pH and packaging. Strict control of these factors is needed to prevent issues.