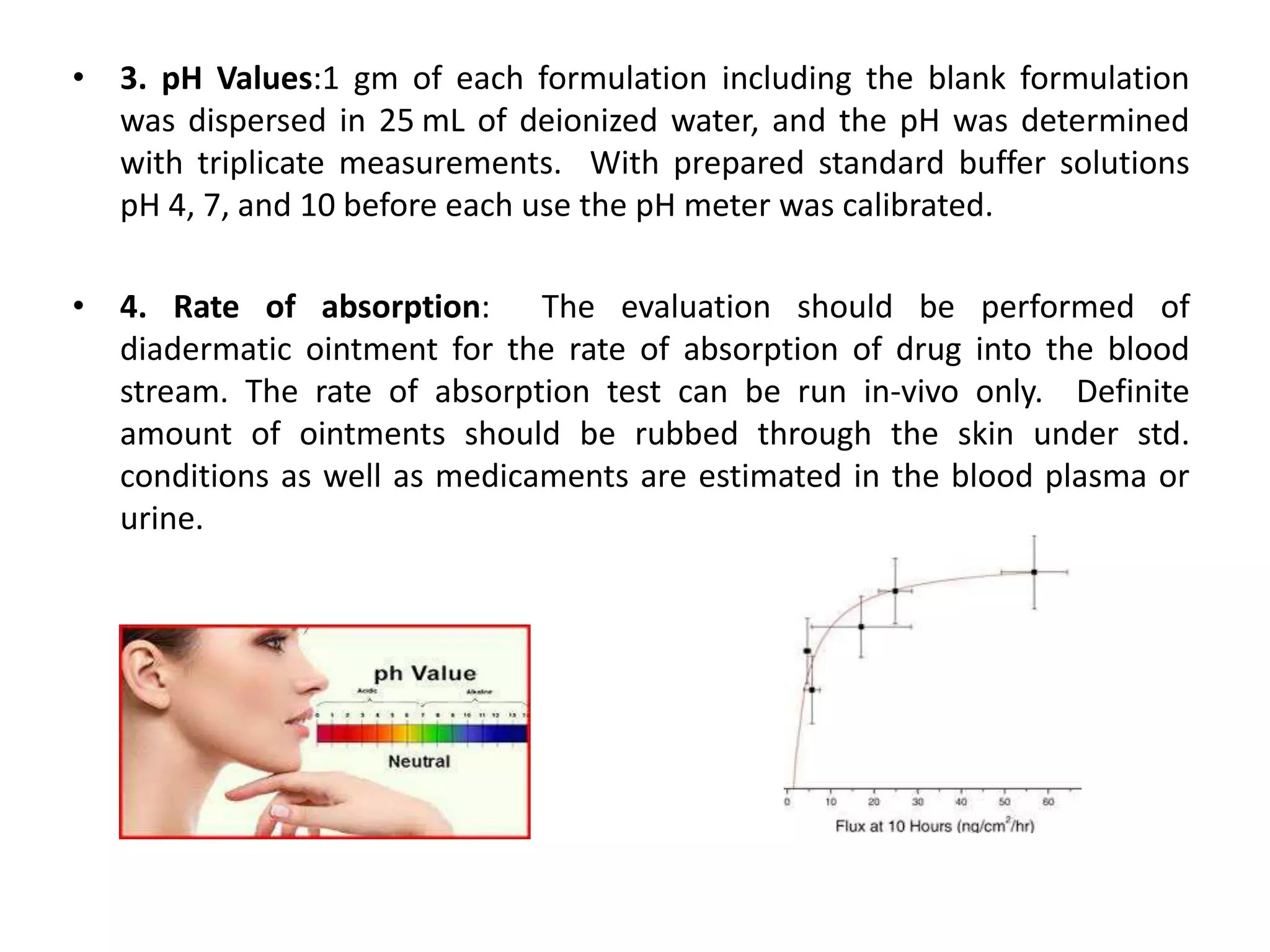
This document discusses quality control tests for semisolid dosage forms like creams, pastes, jellies and ointments. It outlines 11 tests: appearance, spreadability, pH, rate of absorption, irritancy, rate of penetration, consistency, sensitivity, drug content, rheology/viscosity, and rate of release of the active medication. These tests evaluate various physical properties and ensure safety, stability and appropriate release of the drug.