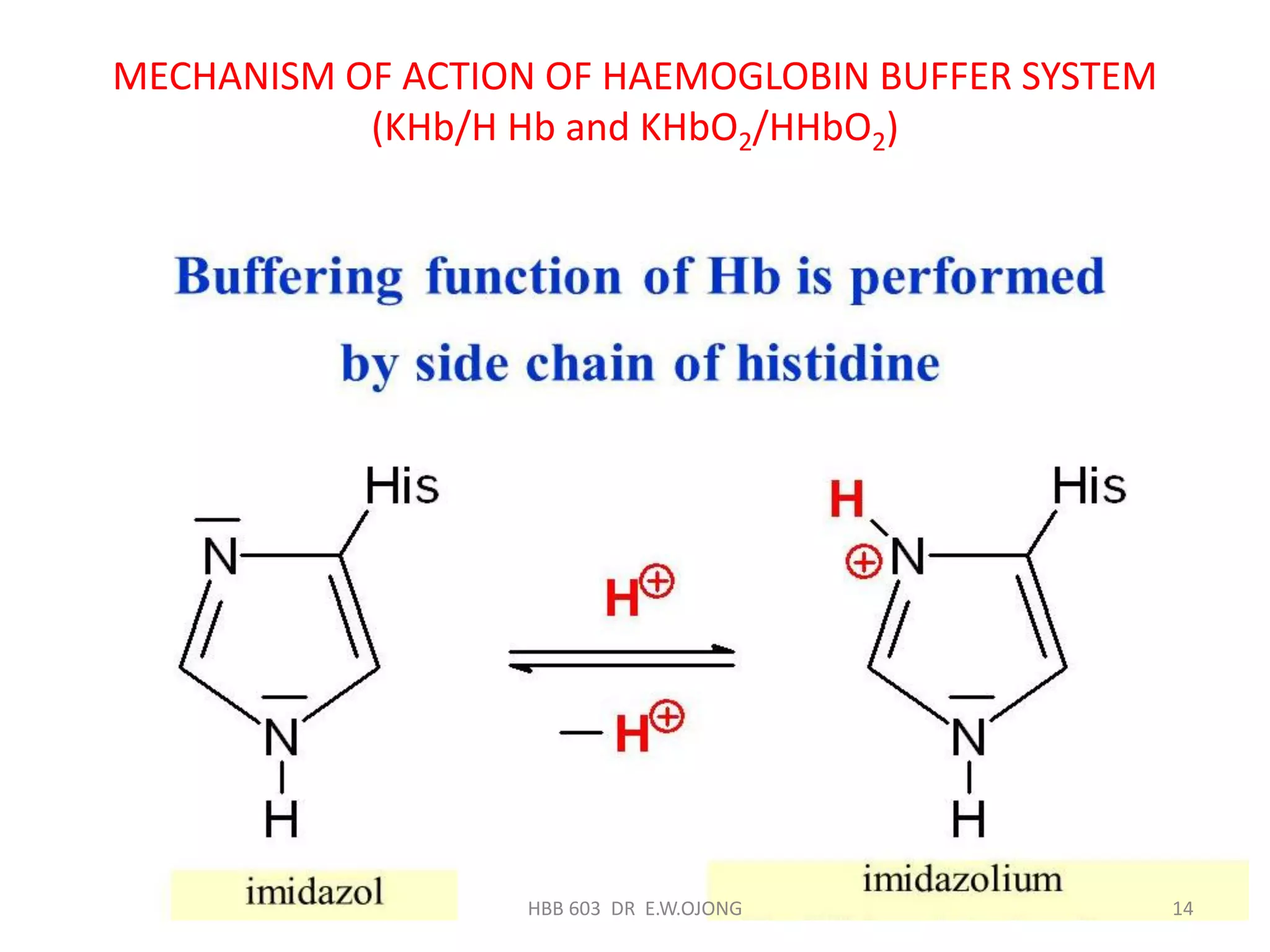
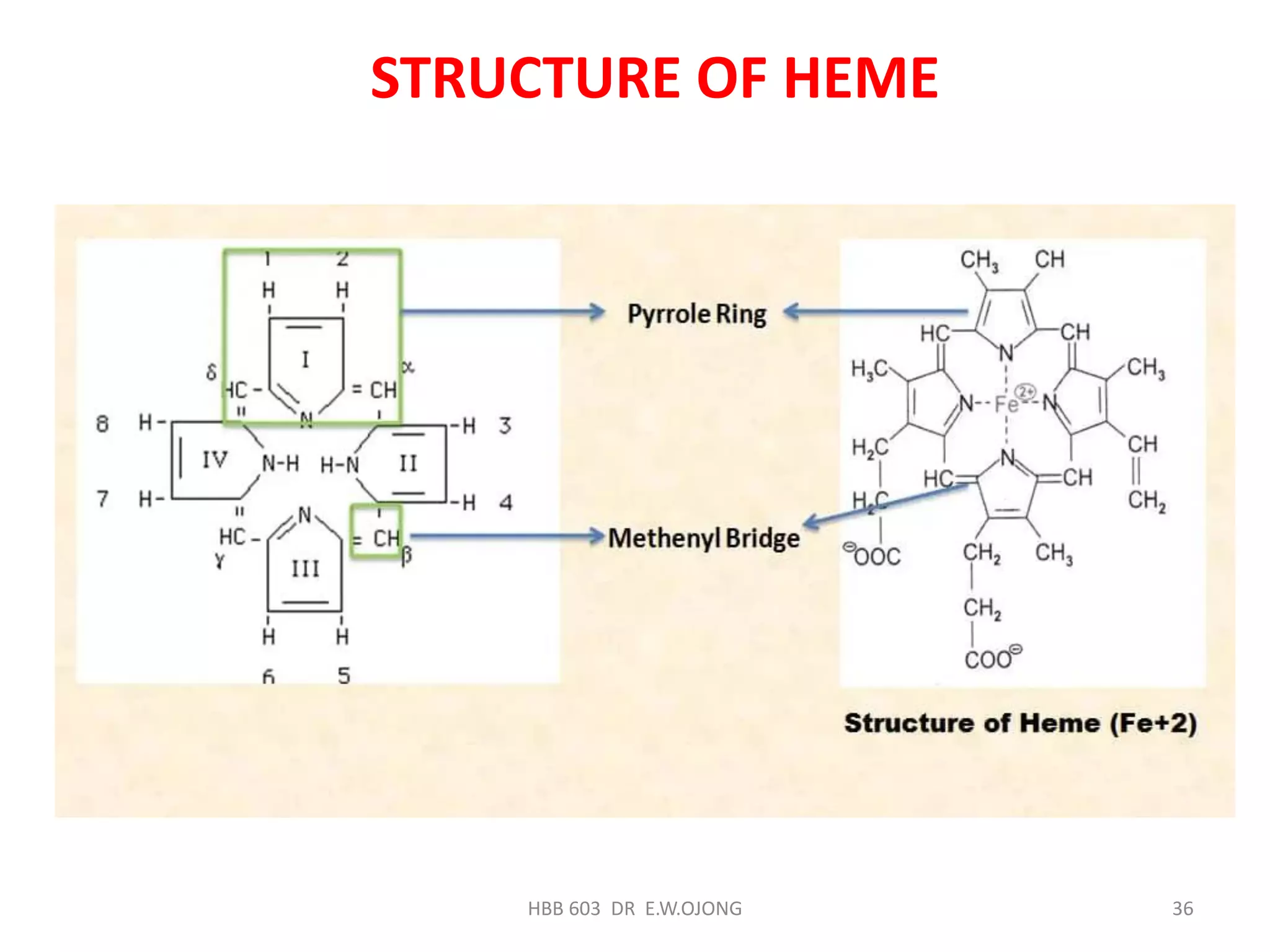
The document discusses hemoglobin synthesis, which consists of two parts: globin synthesis and heme synthesis. Globin is synthesized through transcription and translation of genes on chromosomes 16 and 11. Heme is synthesized through the Shemin cycle, where glycine and succinyl-CoA are condensed to form delta-aminolevulinic acid, which undergoes further modifications to ultimately form protoporphyrin IX. Protoporphyrin then binds iron to form heme. Heme synthesis is regulated by feedback inhibition of delta-aminolevulinic acid synthase, and both heme and globin are required for the formation of functional hemoglobin.