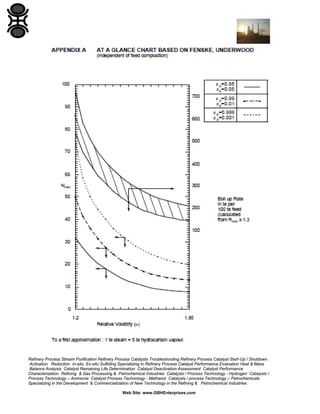
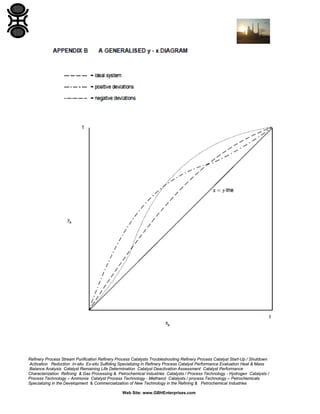
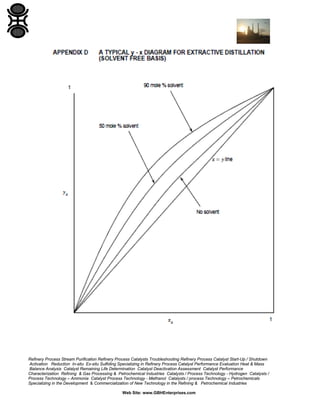
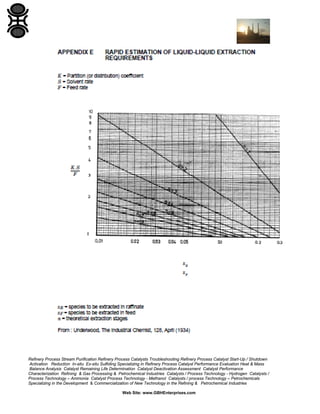
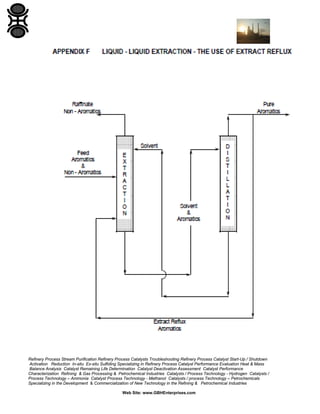
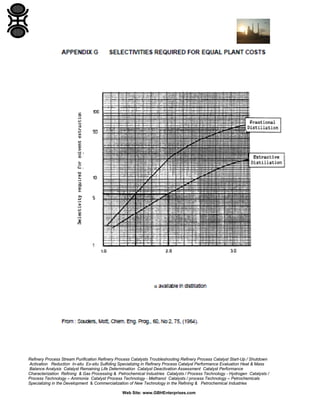
The GBH Enterprises, Ltd. Process Engineering Guide focuses on fluid separation methods, primarily addressing distillation techniques and liquid-liquid extraction, while providing insights into various separation technologies applicable in refining and petrochemical industries. It emphasizes the importance of selecting appropriate methods based on feasibility, cost, and operational requirements, and includes a stepwise approach for method selection through a separation logic tree. Additionally, the guide offers details on catalyst performance evaluation, troubleshooting, and the design considerations for effective fluid separation systems.