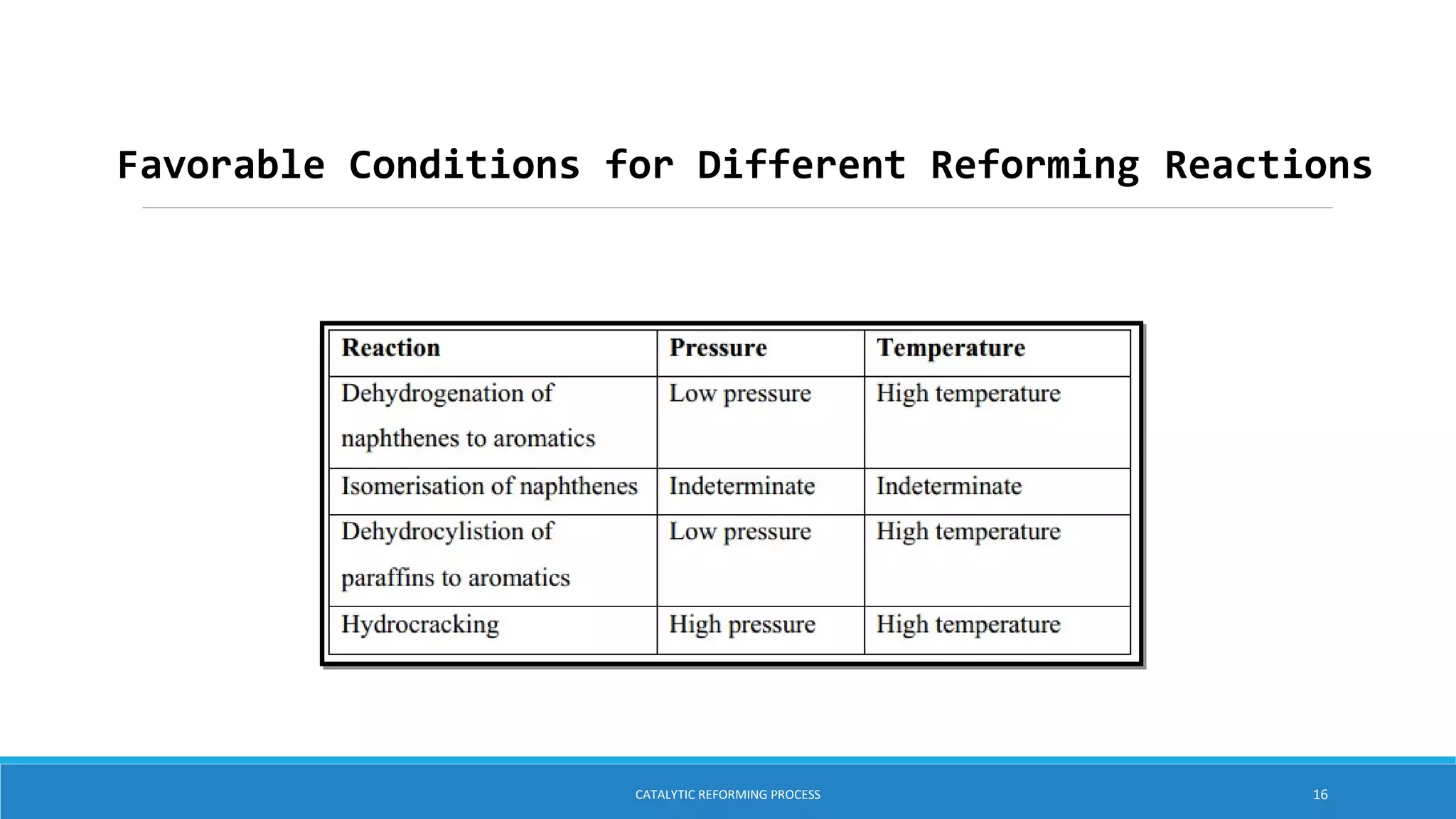
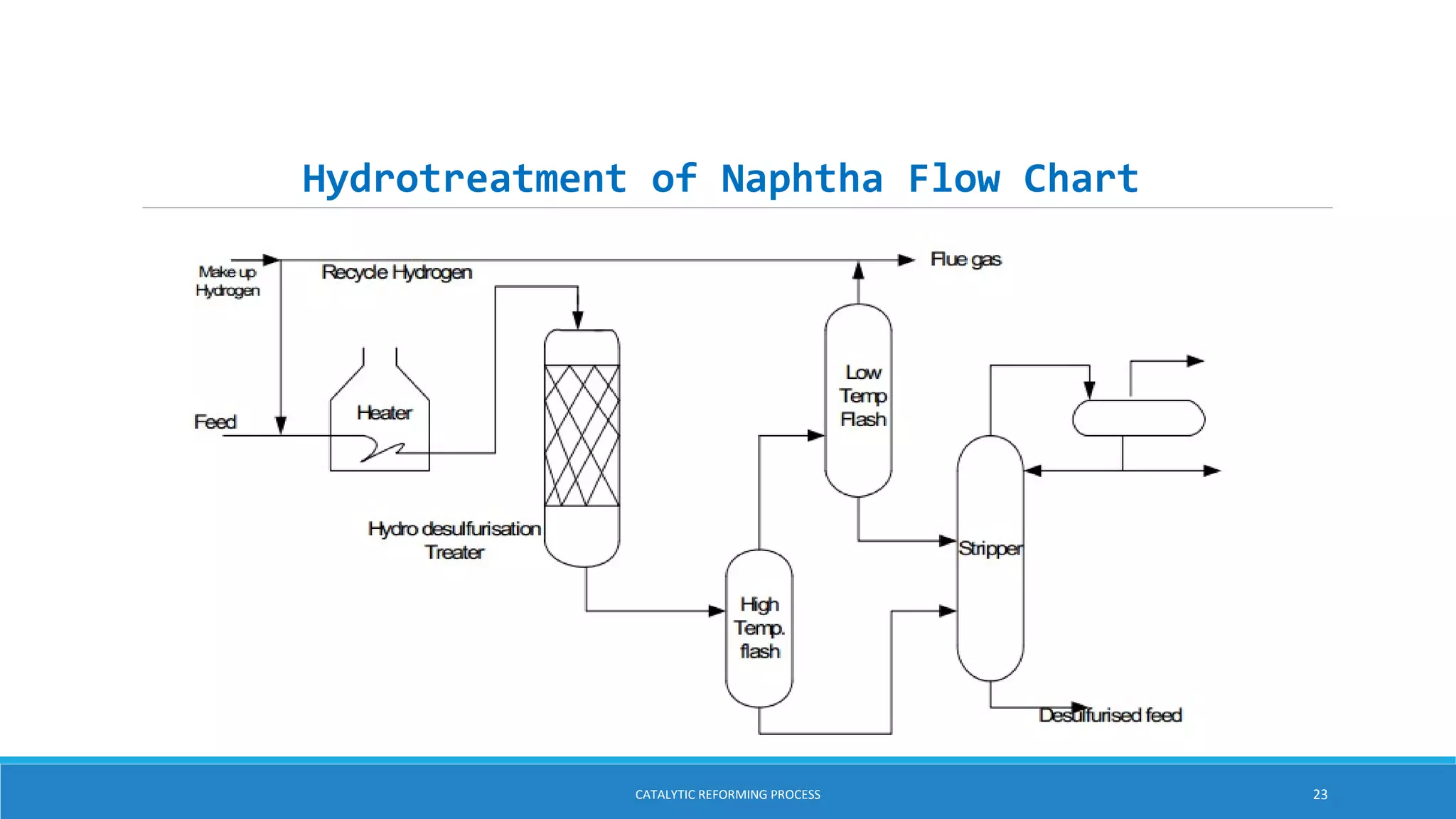
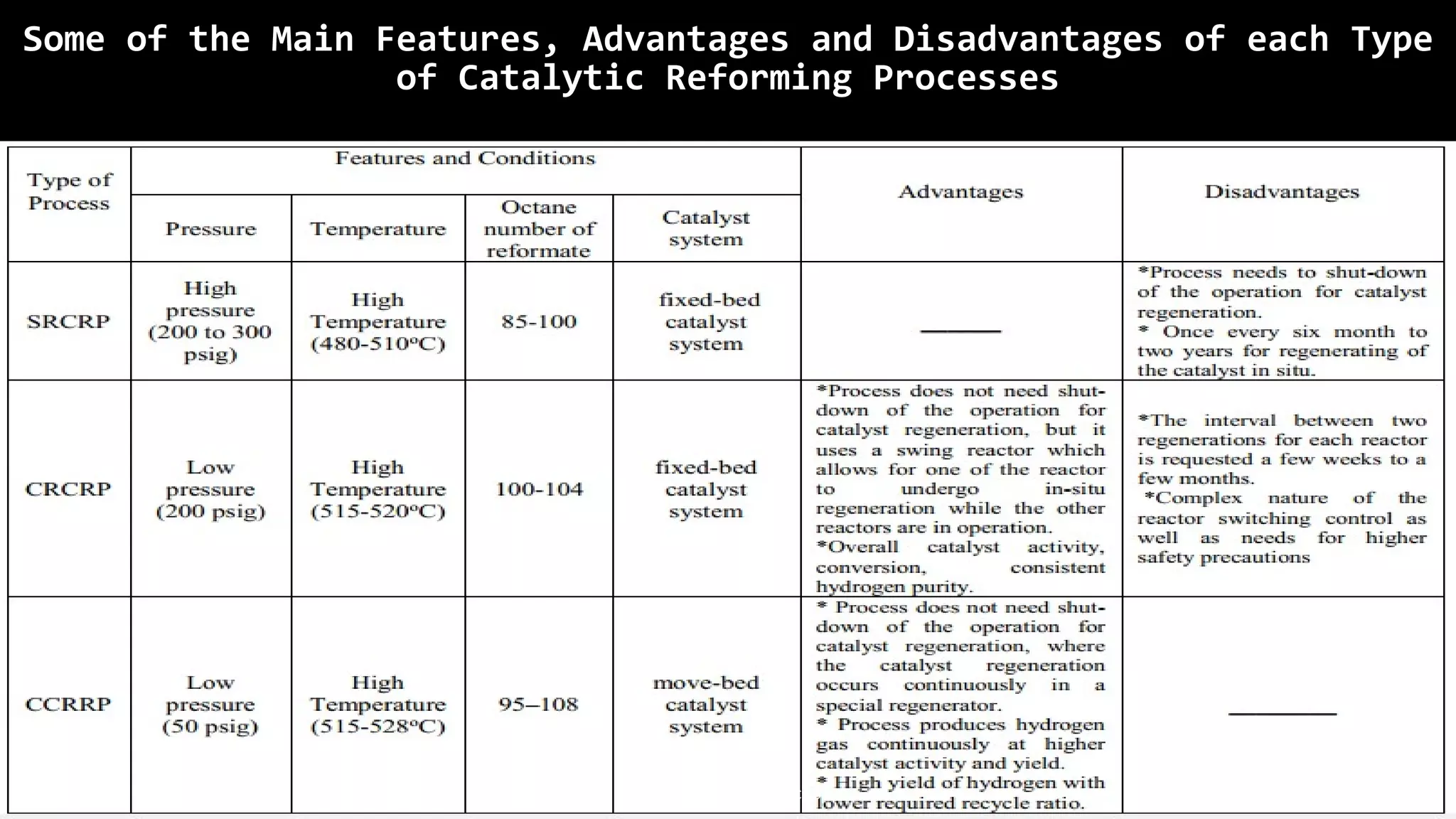
The catalytic reforming process transforms low-octane naphtha from crude oil into high-octane gasoline blending stocks and aromatics through various chemical reactions, including dehydrogenation and isomerization. Developed in the 1940s by Vladimir Haensel, the process utilizes catalysts like platinum on aluminum support to improve octane ratings and remove impurities. Current processes are classified into semi-regenerative, cyclic regenerative, and continuous catalytic regeneration systems, with the overall aim of producing high-quality fuel for the petroleum and petrochemical industries.