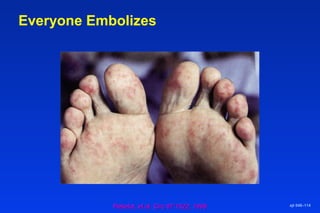
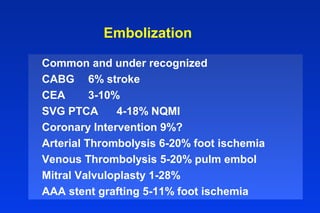
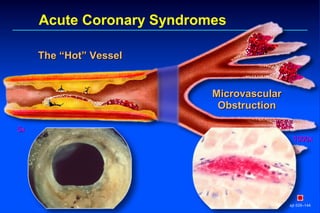
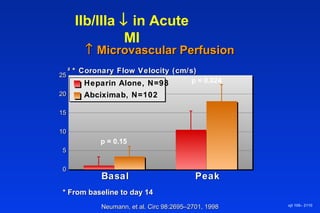
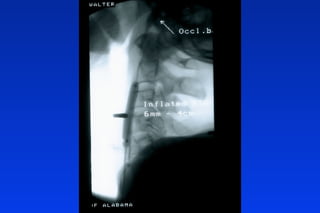
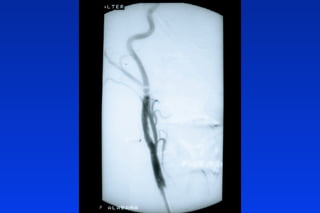
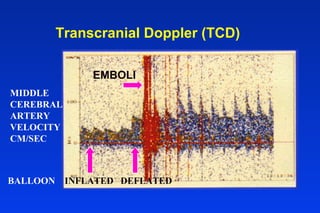
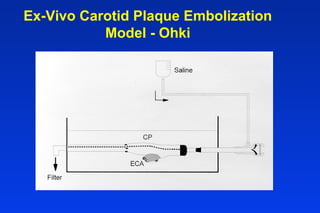
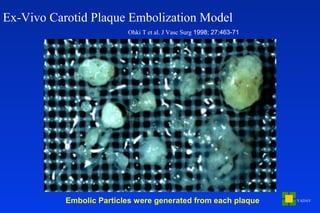
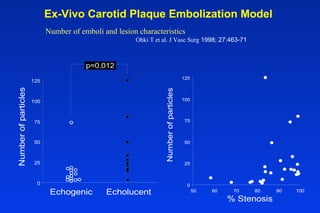
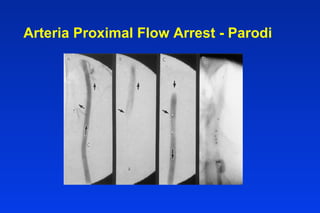
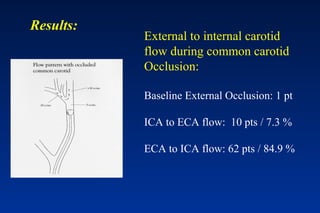
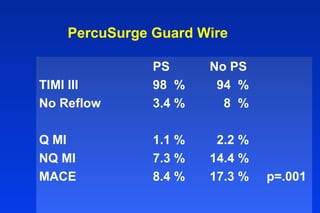
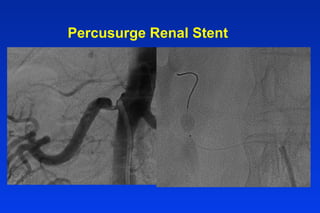
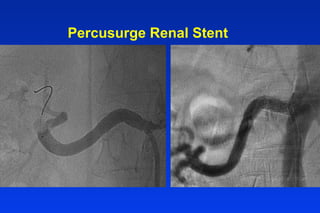
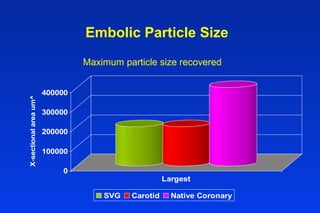
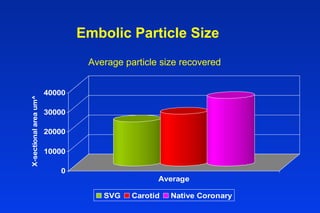
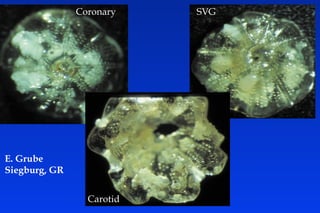
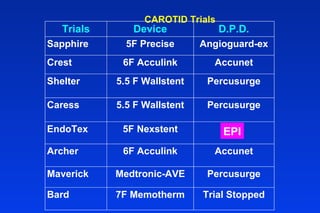
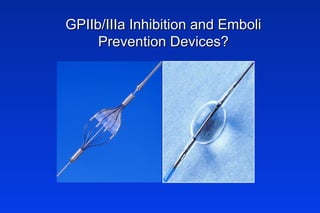
Embolic protection devices aim to prevent stroke and other complications during carotid artery procedures by capturing debris released during stenting or angioplasty. Several filter-based and occlusion-based devices have been developed, with filter designs including supported nitinol frames and unsupported mesh. Clinical trials show filter devices can retrieve numerous embolic particles from procedures without interrupting blood flow. Larger trials are currently underway to determine if combination of stenting and embolic protection reduces stroke risk compared to carotid endarterectomy. Embolic protection may benefit high-risk patients and procedures like SVGs, acute MI, and renal angioplasty.