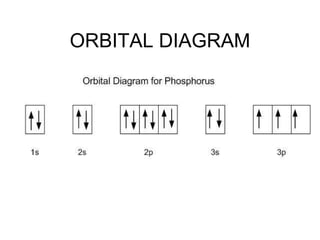
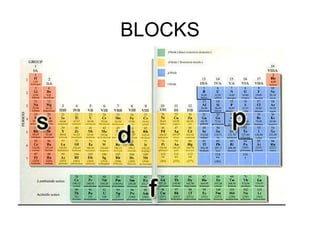
Electron configurations provide information about the location of electrons in an atom's orbitals. There are three main rules for determining electron configurations: 1) electrons fill orbitals starting with the lowest energy levels and moving upwards, 2) the Pauli exclusion principle states that no two electrons can occupy the same orbital with the same spin, and 3) Hund's rule states that electrons will occupy orbitals singly before pairing up. Writing out electron configurations involves determining the element's number of electrons and following the Aufbau principle of filling orbitals in order of increasing energy.








![Noble Gas Electron Configurations Recall, the electron configuration for Na is: Na: 1 s 2 2 s 2 2 p 6 3 s 1 We can abbreviate the electron configuration by indicating the innermost electrons with the symbol of the preceding noble gas. The preceding noble gas with an atomic number less than sodium is neon, Ne. We rewrite the electron configuration: Na: [Ne] 3 s 1](https://image.slidesharecdn.com/electronconfiguration-111130184447-phpapp02/85/Electron-configuration-11-320.jpg)
![NOBLE GAS CONFIGURATION The electron configuration for gold (Au) is: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 10 5p 6 6s 2 4f 14 5d 9 Gold (Au) is in the 6 th row of the periodic table : This gives us: [Xe] 6s 2 4f 14 5d 9](https://image.slidesharecdn.com/electronconfiguration-111130184447-phpapp02/85/Electron-configuration-12-320.jpg)

![MORE EXAMPLES Noble Gas Notation: C = [He]2s 2 2p 2 Te = [Kr]5s 2 4d 10 5p 4 I = [Kr]5s 2 4d 10 5p 5](https://image.slidesharecdn.com/electronconfiguration-111130184447-phpapp02/85/Electron-configuration-14-320.jpg)






